The Ten Simplest Probe Experiments
Chemical species can be probed by adding and removing: photons, electrons, protons, hydride ions and hydrogen radicals.
Introduction
Chemical species can be studied by using other chemical species as probes. The five simplest probes are:
Photon, hv
Electron, e–
Proton (hydrogen cation), H+
Hydride ion (hydrogen anion), H–
Hydrogen radical (hydrogen atom), H•
Each of these five probe species can – in principle – be added or removed form a species, which results in 10 possible probe experiments, however, any single species not be able to partake in all 10 of the probe experiments.
We will be using methane as our subject molecule:
Add A Photon to Methane
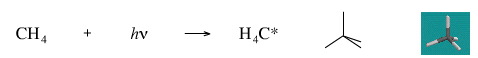
Adding a photon gives an excited state methane, which may relax in a number of ways. The effect will depend upon the wavelength (energy) of the photon. Infrared radiation of the correct wavelength will cause the methane molecule to vibrate. Ultra violet radiation of the correct wavelenth will result in electronic excitation, or possibly ionisation.
Geometry optimisation using density function theory at the pBP/DN** level shows excited state methane to be tetrahedral.
The study of electromagnetic radiation interacting with matter is the science of spectroscopy and is outside the scope of this web book.
Remove A Photon from Methane
A photon can only be removed from an excited species. Thus, the above process can be deemed to occur when an excited methane loses a photon (ie, the reverse of the methane plus photon reaction).
Add An Electron to Methane
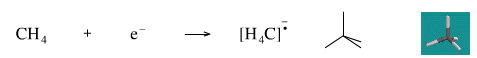
Adding an electron to methane gives the methane radical anion. The species is an anion because it has an excess electron and it is a radical because the extra electron is unpaired.
The methane radical anion is known in the gas phase. Radical anions are rouienly generated and studied in the technique of negative ion mass spectrometry.
Geometry optimisation using density function theory at the pBP/DN** level shows the methane radical anion to be tetrahedral.
Remove An Electron from Methane
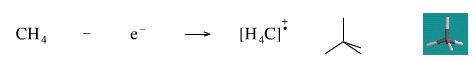
Removing an electron from methane gives the methane radical cation. The species is a cation because is has one electron less that is required for neutrality, and it is a radical because it has an unpaired electron.
Radical cations are routienly generated in the gas phase and studied in the technique of mass spectrometry.
Geometry optimisation using density function theory at the pBP/DN** level shows the methane radical cation to be tetrahedral.
Add A Proton to Methane
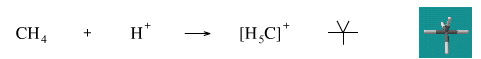
Methane (and other alkanes) can be protonated in solution by George Olah type "super-acids", for example H+[SbF6]–. Protonated methane is called the carbonium ion.
Geometry optimisation using density function theory at the pBP/DN** level shows the carbonium ion to be trigonal bipyramidal.
Remove A Proton from Methane
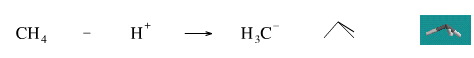
While it is not possible to remove a proton from methane to generate the methyl anion, H3C–, the reverse reaction is well know. Methyl lithium, CH3Li, is a well known strong base which violently reacts with water to give methane.
Geometry optimisation using density function theory at the pBP/DN** level shows the carbanion to be trigonal pyramidal. The same geometry is predicted with VSEPR.
Add A Hydride Ion to Methane
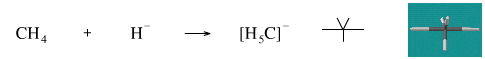
This reaction is not known for methane. However borane, BH3, reacts with hydride ion to give the tetrahydroborate ion, [BH4]–.
Geometry optimisation using density function theory at the pBP/DN** level shows the [H5C]– ion to be trigonal bipyramidal.
Remove A Hydride Ion from Methane
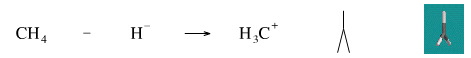
While it is not possible to remove a hydride ion from methane to generate the methyl cation (the carbenium ion), the reverse reaction is well know. Methyl iodide reacts with lithium anuminium hydride [a source of hydride ion] to give methane.
Geometry optimisation using density function theory at the pBP/DN** level shows the carbenium ion to be planar. The same geometry is predicted with VSEPR.
Add A Hydrogen Radical to Methane
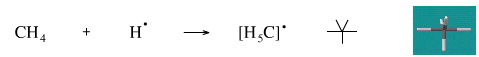
This reaction is not known for methane.
Geometry optimisation using density function theory at the pBP/DN** level shows the H5C• radical to be trigonal bipyramidal.
Remove A Hydrogen Radical from Methane
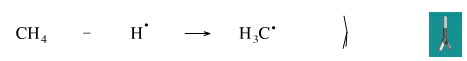
A hydrogen radical can be abstracted from methane by a chlorine radical to give a methyl radical, and this is one of the steps in the free radical halogenation of methane to methyl chloride.
Geometry optimisation using density function theory at the pBP/DN** level shows the carbenium ion to be planar. Experiment shown it to be slightly distorted to trigonal pyramidal.
© Mark R. Leach 1999-2009
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com
This free, open access web book is an ongoing project and your input is appreciated.



