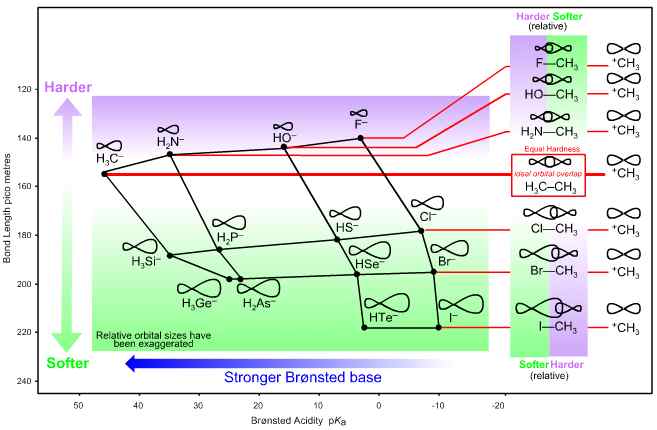
Figure 7
Lobe-HOMO Lewis base hardness and bond length.
The graph shows the ‘to carbenium ion’ bond length of the anionic Lobe-HOMO array plotted against pKa. The C—C bond length associated with ethane, formed when H3C+ complexes with –CH3 (a type 15 Lewis acid/base complexation), is of intermediate length, 154 pm.
If the carbenium ion, H3C+, is deemed to be of the same hardness as the carbanion, —CH3 then "harder-softer" arguments reverse between F—CH3 (hard/soft) and Cl—CH3 (soft/hard). Therefore, F– cannot be congeneric with Cl–, Br– and I– with respect to H3C+.
© Mark R. Leach 1999-2005
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mrl@meta-synthesis.com
This free, open access web book is an ongoing project and your input is appreciated.