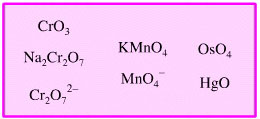
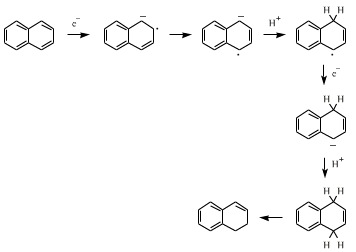The chemogenesis analysis identifies five distinct types of electronic reaction mechanism, here, and one of these is redox chemistry. This page explores the different types of oxidation and reduction reaction behaviour that lie behind the easily memorised: OIL RIG, Oxidation Is [electron] Loss and Reduction Is [electron] Gain.
Introduction to Redox Chemistry
Redox chemistry is concerned with net electron flow to and from a defined centre during a chemical reaction. A defined centre may be:
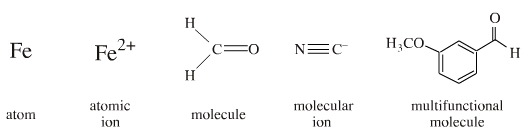
A defined centre is said to be oxidised if the electron density decreases, and reduced if electron density increases, during a reaction. The rule is:
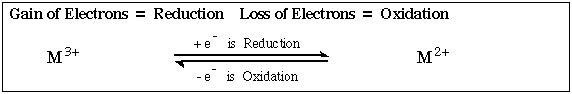
Loss of electrons equates with Oxidation
and
Gain of electrons equates with Reduction
The oxidation of a defined centre can be changed in two ways.
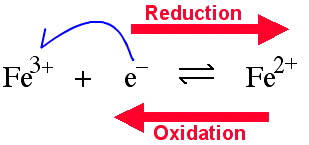
Firstly by Single Electron Transfer (SET) to the defined centre (reduction) or from the defined centre (oxidation). For example, the iron(III) ion, Fe3+, can be reduced to iron(II), Fe2+. The reaction can also occur in the oxidation direction.
The reduction electron can either be provided by a chemical reducing agent (often a metal) or electrochemically.
Electron flow by way of single electron transfer oxidation and reduction can be predicted using standard reduction potential data. A software gadget using has been built into The Chemical Thesaurus.
The second method of changing the oxidation number is by reversal of bond polarisation at the defined centre.
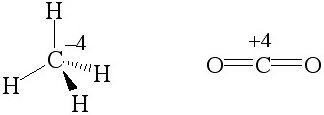
Hydrogen is electropositive and it renders the carbon of methane, CH4, electron rich and it is defined as having an oxidation number of -4. However, the carbon of carbon dioxide has an oxidation number of +4 because oxygen is more electronegative than carbon. (Each bond contributes once.)
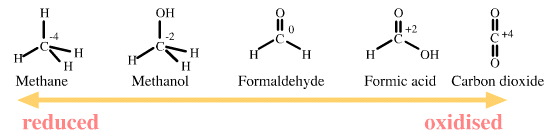
Carbon is able to exist in several oxidation states:
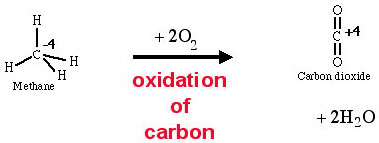
The combustion of methane to carbon dioxide is an oxidation of carbon because the oxidation number of carbon increases from -4 to +4
It follows that Redox Chemistry can proceed by three types of redox reaction:


Classification of Redox Types
A considerable number of oxidising agents and reducing agents have been added to The Chemical Thesaurus. These have been classified into six general types of reducing agent and six general types of oxidising agent. However, please note that the classification is NOT as clear cut or rigorous as that carried out for Lewis acid and Lewis base types.
Single Electron Transfer Electron Donor Reducing Agent
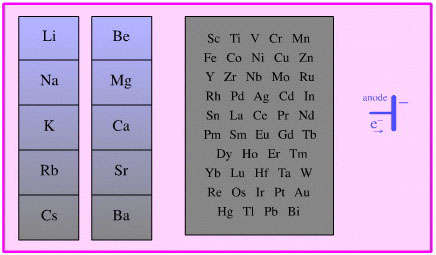
Species which act as donors of electrons, including all electropositive elements including all metals and anodes.
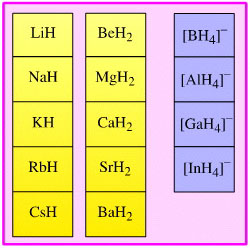
Hydride Complex Reducing Agent
Complexes which act as donors of basic, nucleophilic hydride ion.
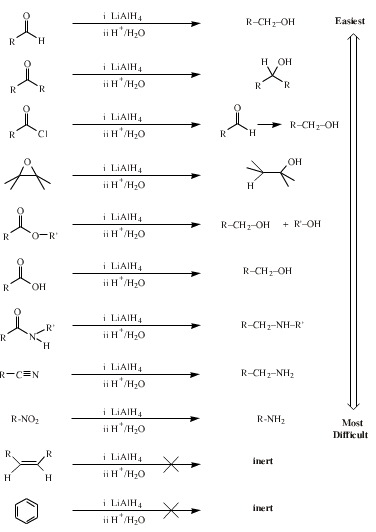
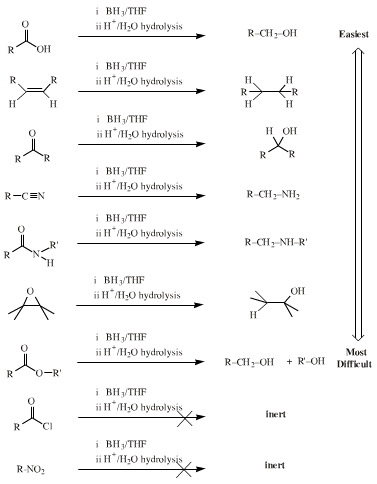
One of the most powerful hydride complex reducing agents is lithium aluminium hydride (lithal), LiAlH4. Lithal selectively reduces some functional groups more easily than others:
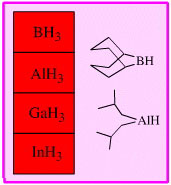
Lewis Acid Hydride Donor Reducing Agent
Hydrogen rich Lewis acids which complex with a Lewis base function and then transfer hydride ion to the function.
Borane, BH3, and its dimer, diborane, B2H6, exhibit a range of reducing ability which is complementary to lithal:
Hydrogen Reducing Agent
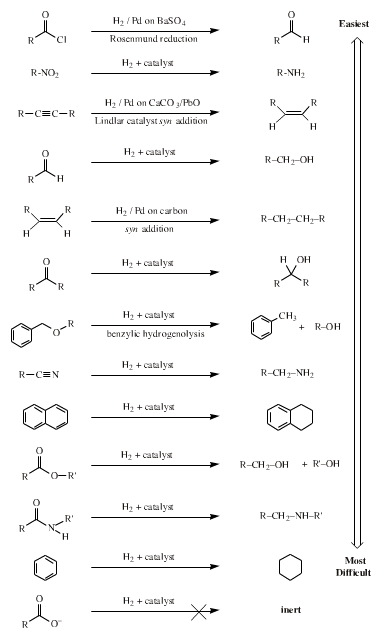
Hydrogen is a reducing agent, although it is nearly always used with a transition metal catalyst to aid addition of hydrogen to a function.
Many functional groups are reduced by hydrogen plus catalyst:
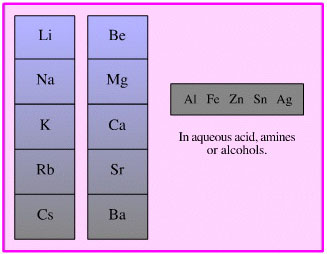
Dissolved Metal Reducing Agent
There are a number of reduction methodologies which use a metal dissolved in a polar solvent: aqueous acid, water, ammonia, alcohols, amines, etc. Careful choice of metal and solvent can result in very selective reduction.
An example of a metal, such as sodium, acting as a reducing agent in an organic reaction is the Birch reduction of aromatic compounds, such as naphthalene, in a weakly protic solvent. The metal provides electrons and the solvent provides protons:
Miscellaneous Reducing Agent
There are a number of reducing agents which act with mechanisms which do not fit into any general pattern.
Single Electron Transfer Electron Removal Oxidising Agent
Species which accept electrons, including: electronegative elements (halogens), metal cations and cathodes.
Hydrogen Removal Oxidising Agent
There are a number of reagents which remove hydrogen from hydrogen rich species. For example, sulfur and selenium can convert cyclohexane to benzene.
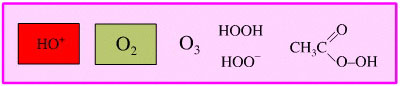
Per-Oxygen Oxidising Agent
As well as oxygen, there are oxygen rich species which are powerful oxidising agents, including: ozone, hydrogen peroxide and per-acids.
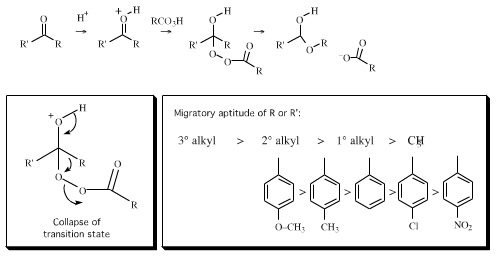
An example of such a reaction is the Baeyer-Villiger oxidation of ketones to esters:
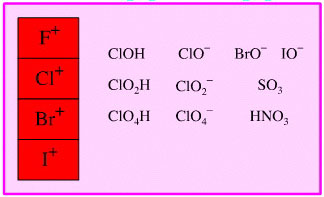
Oxidised Main Group Element Oxidising Agent
Highly oxidised main group elements, including the halogen (enium) cations can act as oxidising agents.
Oxidised Heavy Metal Oxidising Agent
Highly oxidised heavy metals and oxidised heavy metal ions can act as oxidising agents:
Miscellaneous Oxidising Agent
There are a number of oxidising agents which act with mechanisms which do not fit into any general pattern:
© Mark R. Leach 1999-2007
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com
This free, open access web book is an ongoing project and your input is appreciated.