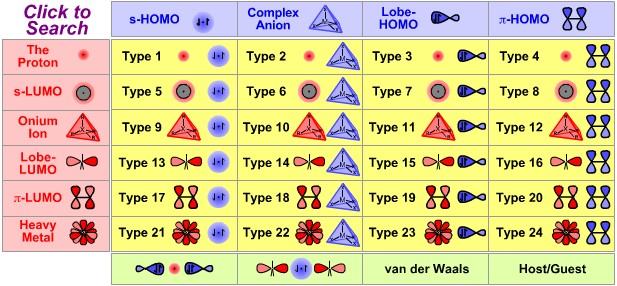
Periodic Table |
 |
 |
 |
 |
 |
 |
 |
| Poster | Nucleophiles & Bases |
Lewis Acid/Base Interaction Matrix Database
 |
Onium Ion Lewis Acids

| Hypervalent Molecular Cations | 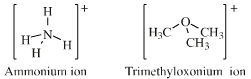
Search for onium ion Lewis acid species in The Chemical Thesaurus |
| FMO Topology: |
The onium ion Lewis acids have a central electronegative atom saturated with Lewis acid "ligands", usually H+ or alkyl+. Onium ion Lewis acids are all proton/X Lobe-HOMO or carbenium ion/X Lobe-HOMO complexes where: X = N, O, F, Ne, P, S, Cl, Ar, As, Se, Br, Kr, Sb, Te, I, Xe |
| Charge: | Positive |
| HSAB: | Intrinsically hard, but species behave as a source of hard H+ or the relatively soft Lobe-LUMO Lewis acid carbenium ion, H3C+. |
| Chemistry: |
Onium ions either form charge-controlled (ionic) complexes or they react by transferring a ligand to a nucleophilic /basic Lewis base. If the transferred ligand is H+, the onium ion acts as a Brønsted Acid. If the transferred ligand is a carbenium ion Lewis acid, the onium ion is said to be an alkylating agent. High symmetry tetraalkyl ammonium ions, such as [(CH3)4N]+, can act as spectator cations. Methane can be protonated to the five valent carbonium ion: [CH5]+ Second order nucleophilic substitution reactions at carbon pass through a five valent carbonium ion transition state: 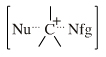 |
| Congeneric Series: |
There is one onium ion Lewis acid planar: 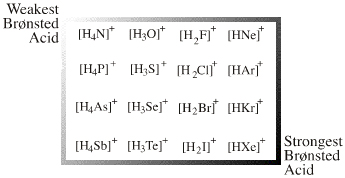
Ammonium, phosphonium, oxonium and sulfonium ions give rise to many ligand replacement congeneric series, for example: 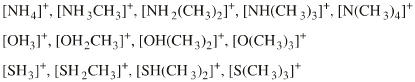
[R4N]+ [R3NR']+ [R2NR2]+ [RNR'3]+ [NR'4]+ where R and/or R' = H, CH3, alkyl, C6H5 etc. |
| Onium ion Lewis acid (generic) |
|
|
|
Aldehyde, protonated (generic) more here |
|
|
Alkyl alkoxy hydroxy carbenium ion (generic) more here |
|
|
Alkyl oxonium ion (generic) more here |
|
|
1° Alkyl oxonium ion (generic) more here |
|
|
Alkyl telluronium ion (generic) more here |
|
|
Amide, protonated (generic) more here |
|
|
Ammonium ion more here |
|
|
1° Ammonium ion (generic) more here |
|
|
2° Ammonium ion (generic) more here |
|
|
3° Ammonium ion (generic) more here |
|
|
4° Ammonium ion (generic) more here |
|
|
Aqueous mineral acid, 0.05 to 1.0 molar (generic) more here |
|
|
Argonium ion more here |
|
|
Aronium ion (generic) more here |
|
|
Arsonium ion more here |
|
|
Arsonium ion (generic) more here |
|
|
Benzene diazonium ion more here |
|
|
Benzonium ion more here |
|
|
Brønsted acid, oxonium (generic) more here |
|
|
Bromonium ion more here |
|
|
tertiary Butyl oxonium ion more here |
|
|
Carbenium ion, beta-amino (generic) more here |
|
|
Carbenium ion, beta-ether (generic) more here |
|
|
Carbenium ion, beta-thioether (generic) more here |
|
|
Carbonium ion more here |
|
|
Carboxylic acid, protonated (generic) more here |
|
|
Chloronium ion more here |
|
|
Chloronium ion, alkyl (generic) more here |
|
|
Dialkyl oxonium ion (generic) more here |
|
|
Dialkyl telluronium ion (generic) more here |
|
|
Dialkylhydroxy carbenium ion (generic) more here |
|
|
Diazonium ion (generic) more here |
|
|
Difluorochloronium ion more here |
|
|
Dihydroxymethyl carbenium ion more here |
|
|
Dimethyl sulfonium ion more here |
|
|
Dimethylammonium ion more here |
|
|
Dimethylformamide, protonated more here |
|
|
Dimethylhydroxy carbenium ion more here |
|
|
Dimethyloxonium ion more here |
|
|
Dimethylsulfoxide, protonated more here |
|
|
Diphenylammonium ion more here |
|
|
Duteronium ion more here |
|
|
Eigen Cation more here |
|
|
Ethenyl bromonium ion more here |
|
|
Ethylacetate, protonated more here |
|
|
Ethylbenzene SeAr intermediate more here |
|
|
Fluoronium ion more here |
|
|
Fluoronium ion, alkyl (generic) more here |
|
|
Guanidine, protonated more here |
|
|
Hydrazinium ion more here |
|
|
Hydronium ion, [H3]+ more here |
|
|
Hydroxy acid, protonated (generic) more here |
|
|
Hydroxy carbenium ion more here |
|
|
Hydroxyammonium ion more here |
|
|
Hydroxydialkoxy carbenium ion (generic) more here |
|
|
Hydroxymethyl carbenium ion more here |
|
|
Imidazonium ion more here |
|
|
Iodonium ion more here |
|
|
Iodonium ion, alkyl (generic) more here |
|
|
Isopropyl oxonium ion more here |
|
|
Kryptonium ion more here |
|
|
Methylammonium ion more here |
|
|
Methyloxonium ion more here |
|
|
Methylsulfonium ion more here |
|
|
Methyltriethylammonium ion more here |
|
|
Neonium ion more here |
|
|
Nitric acid, protonated more here |
|
|
Nitrilium ion (generic) more here |
|
|
Nitrobenzene SeAr intermediate more here |
|
|
Nitrobenzene, protonated more here |
|
|
Nitromethane, protonated more here |
|
|
Oxonium ion more here |
|
|
Phenyl ammonium ion (generic) more here |
|
|
Phenylammonium ion more here |
|
|
Phenylethanone SeAr intermediate more here |
|
|
Phenyloxonium ion more here |
|
|
Phosphonium ion more here |
|
|
Phosphonium ion (generic) more here |
|
|
Phosphonium ion, alkyl triphenyl (generic) more here |
|
|
Protonated base, [H-B]+ (generic) more here |
|
|
Pyridinium ion more here |
|
|
Pyrylium ion more here |
|
|
Selenonium ion more here |
|
|
Stibonium ion more here |
|
|
Sulfonium ion more here |
|
|
Sulfonium ion, alkyl (generic) more here |
|
|
Sulfonium ion, dialkyl (generic) more here |
|
|
Telluronium ion more here |
|
|
Tetrahydrofuran, protonated more here |
|
|
Tetramethylammonium ion more here |
|
|
Thiocarbenium ion (generic) more here |
|
|
Thiopyrylium ion more here |
|
|
Trialkyloxonium ion (generic) more here |
|
|
Trialkylsulfonium ion (generic) more here |
|
|
Triethylarsonium ion more here |
|
|
Trimethylammonium ion more here |
|
|
Trimethyloxonium ion more here |
|
|
Trimethylphosphonium ion more here |
|
|
Trimethylsulfonium ion more here |
|
|
Trimethylsulfoxonium iodide more here |
|
|
Trimethylsulfoxonium ion more here |
|
|
Triphenyloxonium ion more here |
|
|
Urea, protonated more here |
|
|
Xenon fluoride cation more here |
|
|
Xenon, protonated more here |
|
|
Ylid (generic) more here |
|
|
Zundel cation more here |
 |
 |
 |
| Poster | Nucleophiles & Bases |
© Mark R. Leach 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com
This free, open access web book is an ongoing project and your input is appreciated.