Periodic Table |
 |
 |
 |
 |
 |
 |
 |
| INTERNET Database of Periodic Tables | Binary Compounds |
Periodicity
A pattern of repeating order is called periodicity. Since the 1830s chemists explored the repeating patterns of properties associated with the chemical elements and their compounds, and from these studies the modern 'medium form' periodic table emerged.
The Periodic Law | Periodicity
The periodic table is a schema showing the elements with their positions emphasising the periodic law:
"The periodic law is the principle that certain properties of elements occur periodically when arranged by atomic number. These similarities can be reflected best by a table, so that commonalties between elements appear both in rows and in columns of the table." Wikipedia
However, it must be emphasised the the periodic "law" is NOT a physical law NOR is it a scientific principle. These terms have very precise meanings and well understood limits of applicability.
Newton's Laws of motion work extraordinary well, as long as the speed is not too fast (approaching the speed of light) or the gravitational potential too high. A Newtonian analysis will get a space craft to Mars, but will not explain the existence of black holes.
Periodicity is a "rule of thumb", a set of trends, a pragmatic analysis... with many exceptions.
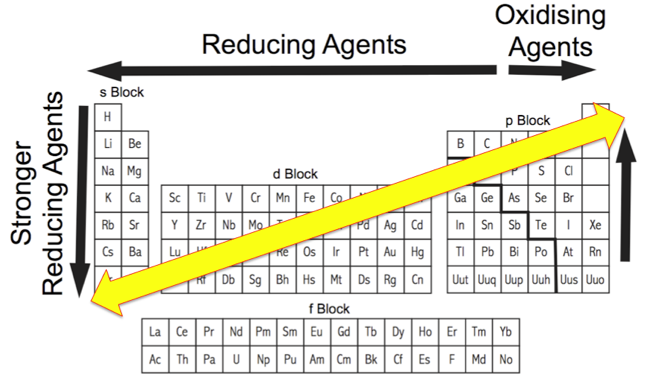
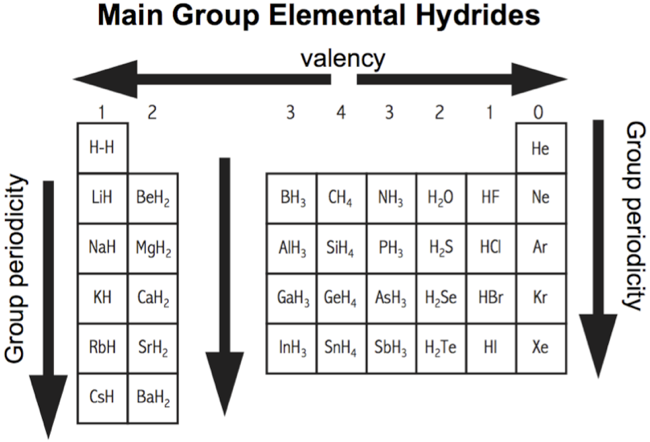
Periodicity works well – but not perfectly – with the s & p-block main group elements, but it is actually rather poor (even very poor) with the d & f-block elements. That said, as the Chemogenesis Web Book is mainly concerned with the chemistry of s & p-block main group elements, we deem the periodicity analysis to be most useful.
Which Is The Best Periodic Table Formulation To Use?
There are many, many formulations of the periodic table – just see the previous page of this web book – but the formulation that most clearly exhibits and displays periodicity is the conventional medium form periodic table, as displayed by WebElements:
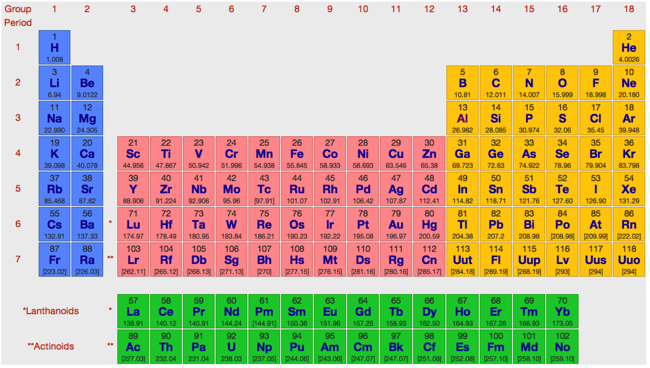
The medium form PT is derived from the long form periodic table (the transformation is explained in more detail in two place, an entry in the PT database and more fully in a paper, published in Foundation of Chemistry:
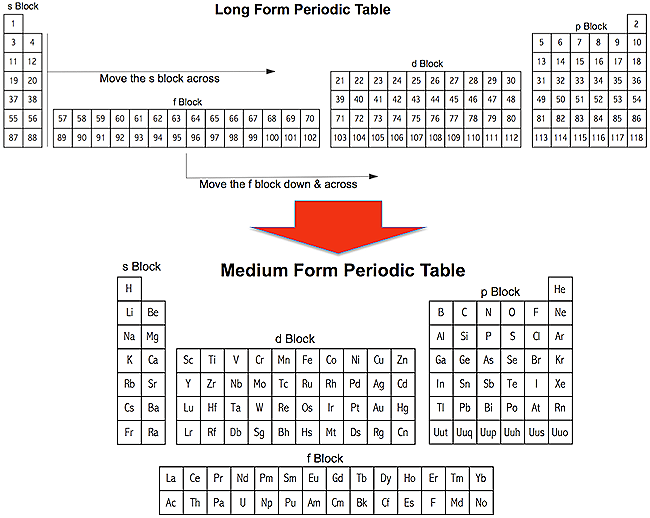
Likewise, the medium form periodic table can be reduced to a short form PT, showing just the main group elements:
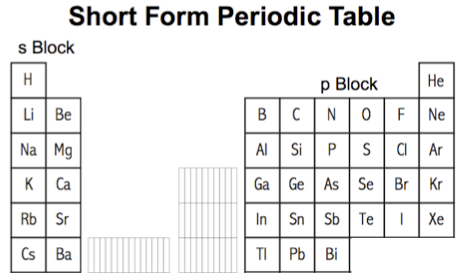
In different situations, periodicity is can be mapped to the long form periodic table, at other times medium or short form periodic tables are employed.
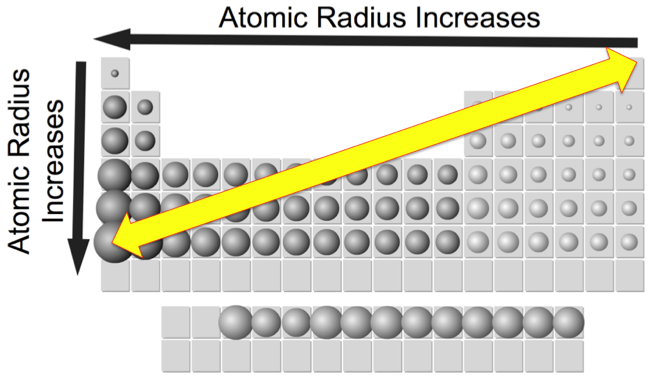
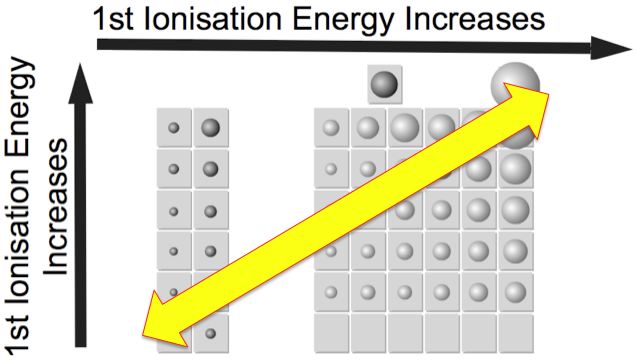
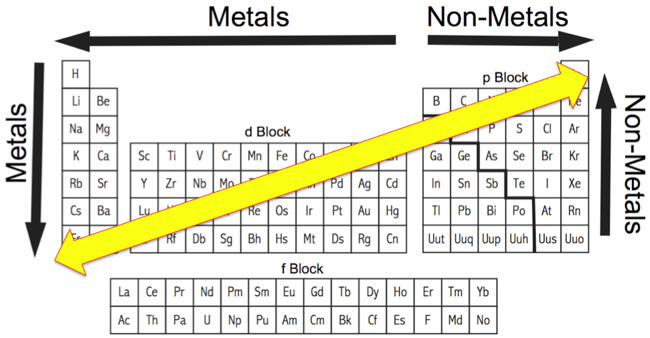
Periodicity
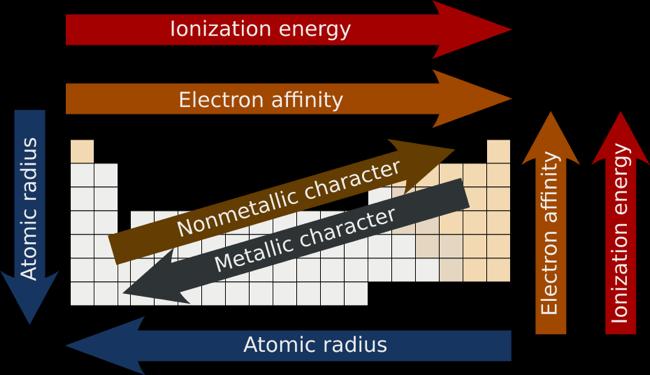
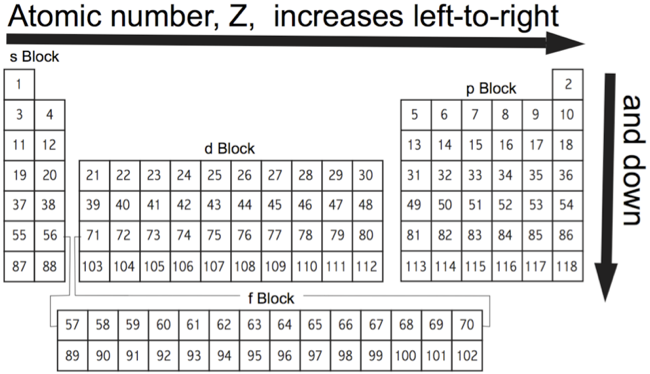
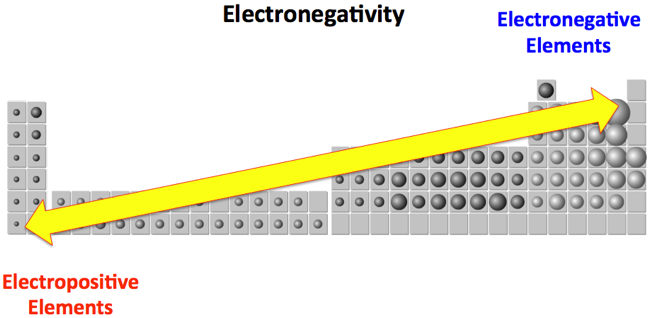
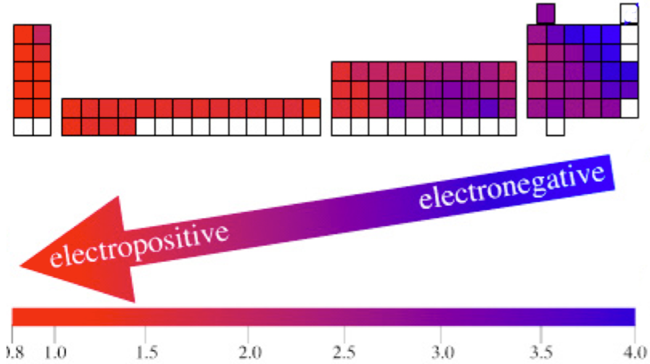
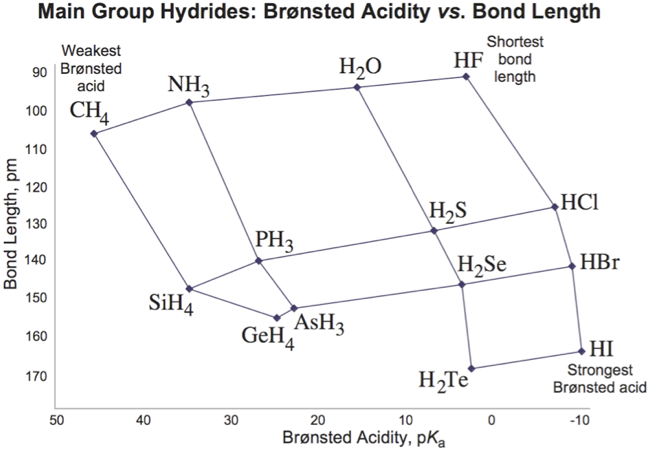
Atomic Number:
 <
<
 |
 |
 |
| INTERNET Database of Periodic Tables | Binary Compounds |
© Mark R. Leach 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com
This free, open access web book is an ongoing project and your input is appreciated.