Periodic Table |
 |
 |
 |
 |
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
The INTERNET Database of Periodic Tables
There are thousands of periodic tables in web space, but this is the only comprehensive database of periodic tables & periodic system formulations. If you know of an interesting periodic table that is missing, please contact the database curator: Mark R. Leach Ph.D. The database holds information on periodic tables, the discovery of the elements, the elucidation of atomic weights and the discovery of atomic structure (and much, much more).
| Year: 1965 | PT id = 525, Type = formulation spiral 3D |
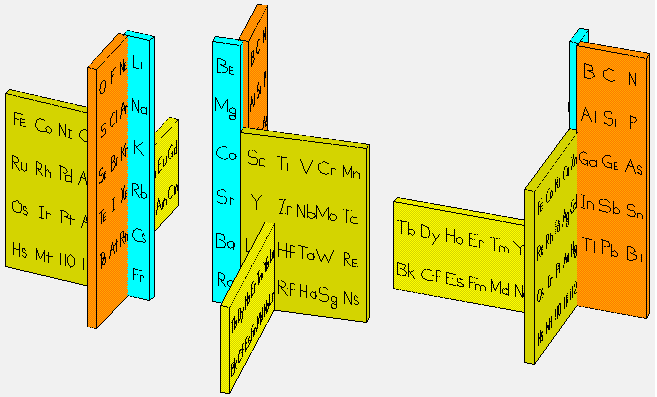
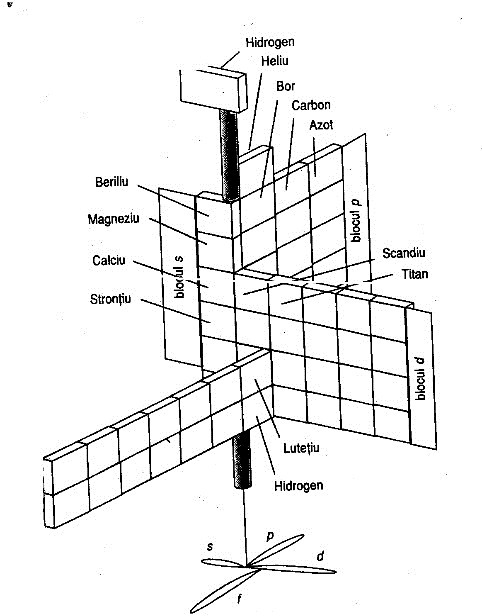
Giguère's Periodic Table
Paul Giguère's Periodic Table formulation, "The 'new look' for the periodic system". Chemistry in Canada vol. 18 (12): 36–39 (see p. 37). More info here: https://github.com/groverlab/giguere-3D-periodic-table.
René Vernon writes:
"I have not considered Giguère’s table at any length, so the following pros and cons are off the top of my head:
Pros:
- Elegant and visually appealing overall design.
- Offers an impression of continuity through its (almost) spiral layout.
Cons:
- In practice, this formulation does not resolve the discontinuity of periods any better than the conventional table; one still needs to complete one turn of the spiral and then mentally leap to the next, much as one moves from one row to the next in the flat form.
- Includes the He-over-Be placement, which remains controversial.
- Quantitative comparisons (group or period properties) become less readable; there’s no immediate visual sense of columns.
- Despite its elegance, the f-block placement appears somewhat awkward.
- Communicates feel more than data.
- It seems to imply relationships between groups on opposite sides of the p-, d- and f-blocks (for instance, between Sc-Y-Lu-Lr and Zn-Cd-Hg-Cn), whereas the actual correspondences run the other way; that is, between the early and later transition groups, such as 3–7 and 8–12. As Imyanitov (2018) observed: "In a generalised form, the properties of the early d? (f?) elements and their compounds are similar to those of the late d? + 5 (f? + 7)."
It’s striking that there are only two pros but several cons, perhaps a reflection of the inherent difficulties faced by three-dimensional periodic tables in improving on the conventional form?"


 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
© Mark R. Leach Ph.D. 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com
This free, open access web book is an ongoing project and your input is appreciated.