Periodic Table |
 |
 |
 |
 |
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
The INTERNET Database of Periodic Tables
There are thousands of periodic tables in web space, but this is the only comprehensive database of periodic tables & periodic system formulations. If you know of an interesting periodic table that is missing, please contact the database curator: Mark R. Leach Ph.D. The database holds information on periodic tables, the discovery of the elements, the elucidation of atomic weights and the discovery of atomic structure (and much, much more).
| Year: 2013 | PT id = 1164, Type = formulation spiral 3D |
Periodic Pyramid
The Periodic Pyramid by Jennifer N. Hennigan and W. Tandy Grubbs, J. Chem. Educ. 2013, 90, 8, 1003-1008, https://doi.org/10.1021/ed3007567.
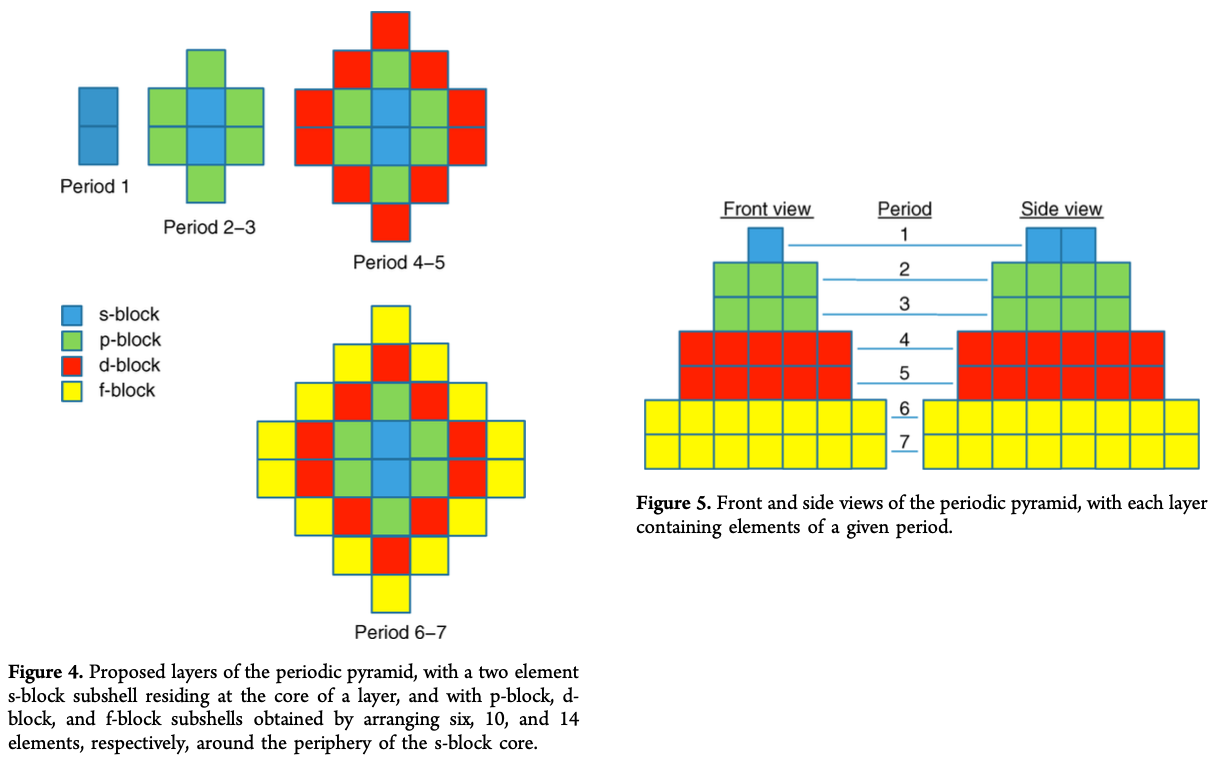
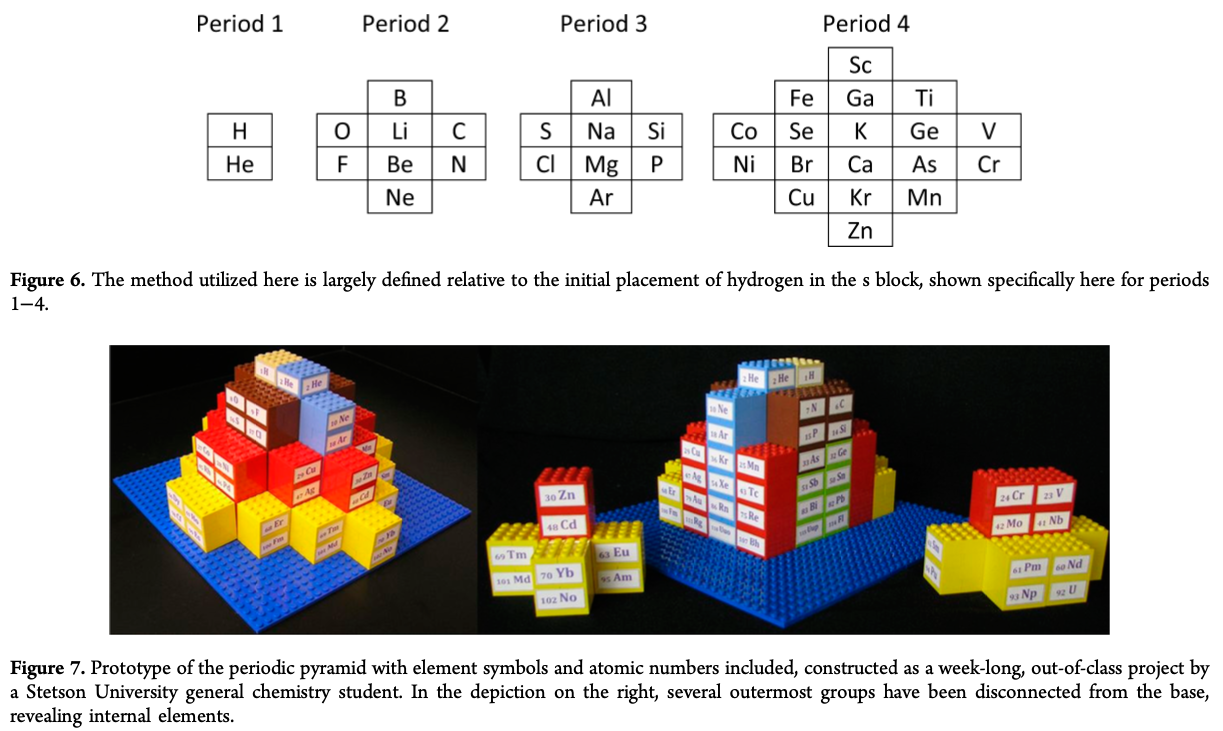
"The chemical elements present in the modern periodic table are arranged in terms of atomic numbers and chemical periodicity. Periodicity arises from quantum mechanical limitations on how many electrons can occupy various shells and subshells of an atom. The shell model of the atom predicts that a maximum of 2, 8, 18, and 32 electrons can occupy the shells identified by the principle quantum numbers n = 1, 2, 3, and 4, respectively.
The numbers 2, 8, 18, and 32 are shown in this work to be related to the triangular numbers from mathematical number theory. The relationship to the triangular numbers, in turn, suggests an alternate method for arranging elements in terms of periodicity. The resulting three-dimensional 'periodic pyramid' is highly symmetric in shape. Just as is true in the modern periodic table, each layer of the periodic pyramid can be separated into shell and subshell contributions. Examining the pyramid's structure is arguably a pedagogically useful activity for college-level introductory or physical chemistry students, as it provides an opportunity to further ponder the shell model of the atom and the origins of periodicity. The connections to number theory are used to show that the outermost subshell of a given shell contains (2n – 1) orbitals."
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
© Mark R. Leach Ph.D. 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com
This free, open access web book is an ongoing project and your input is appreciated.