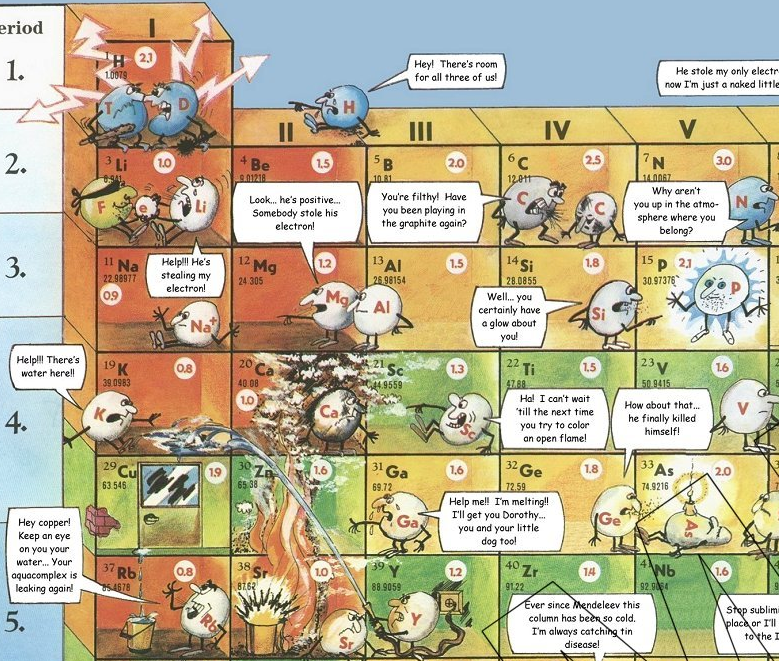
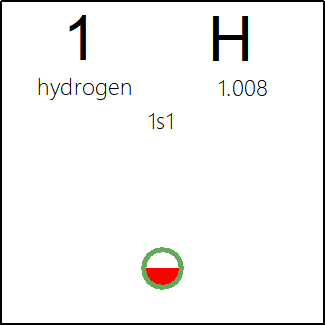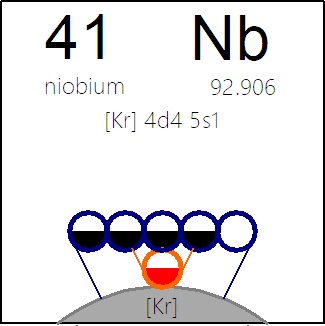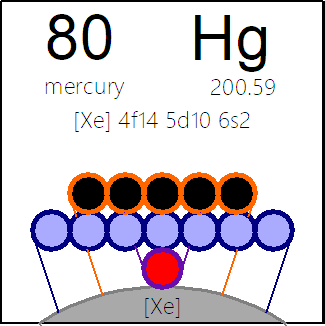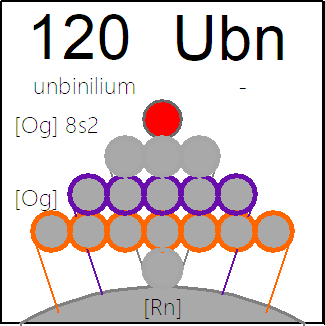
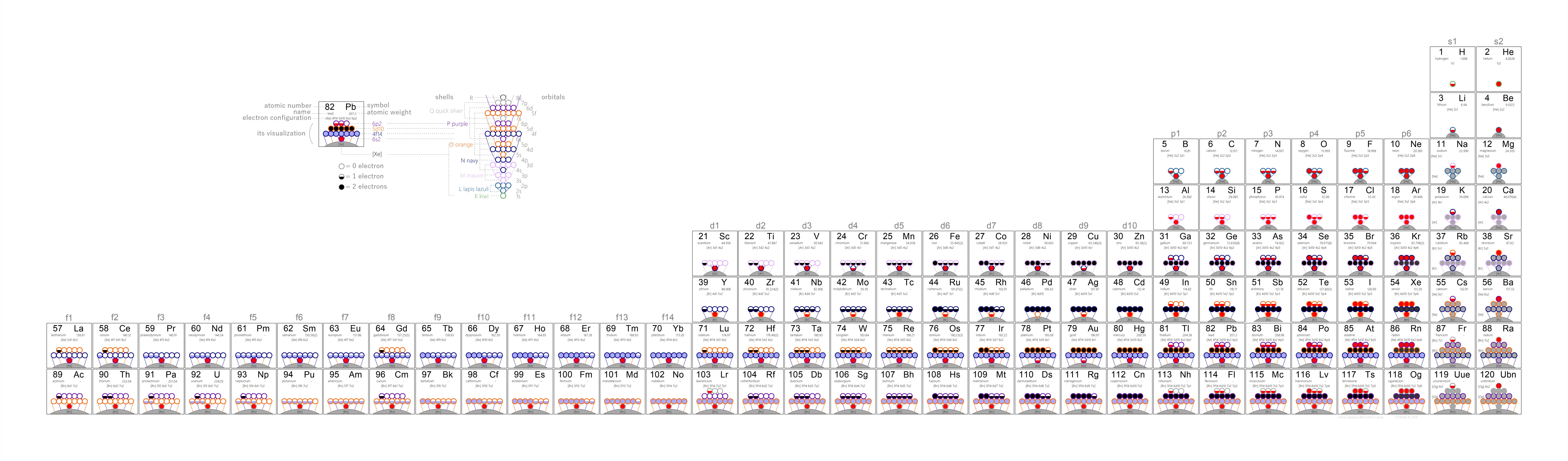
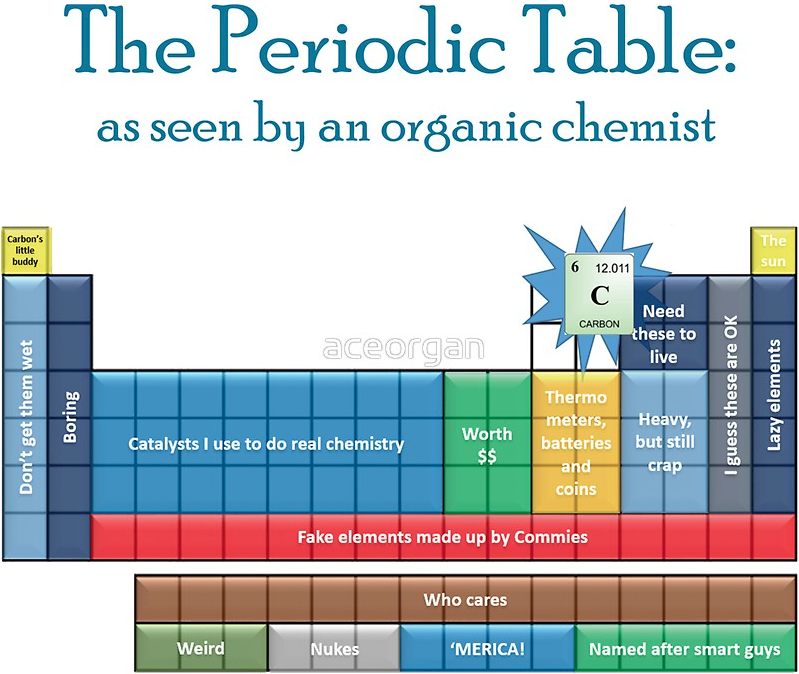
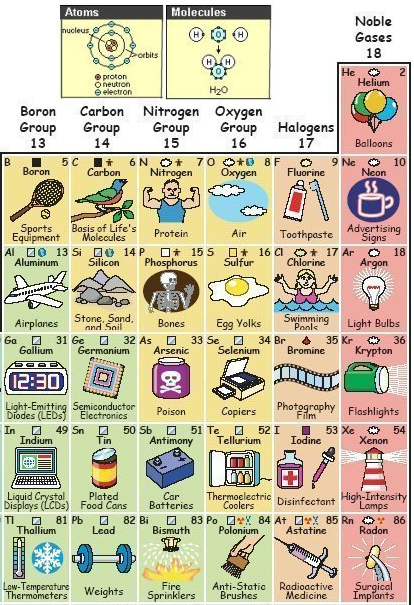
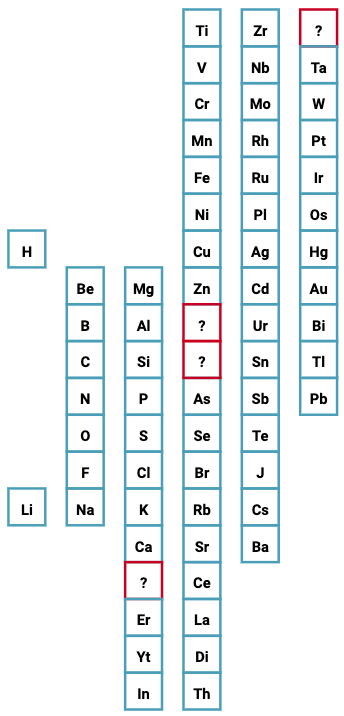
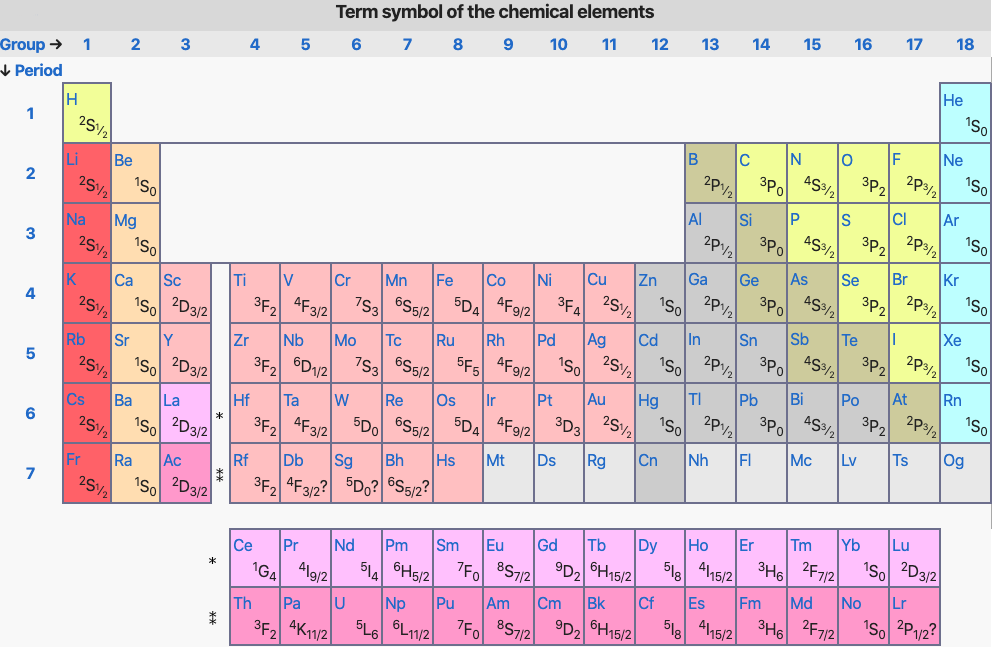
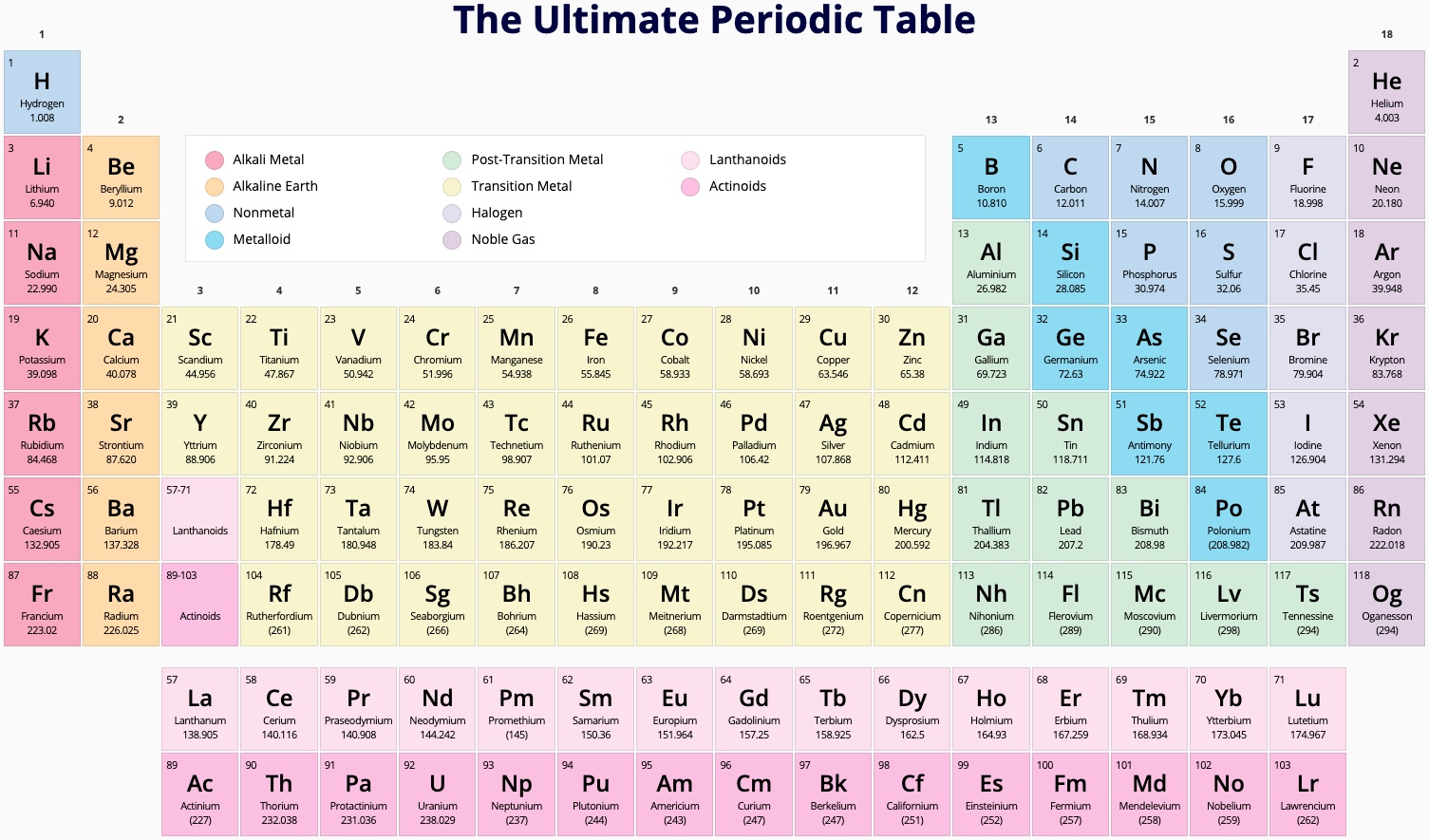
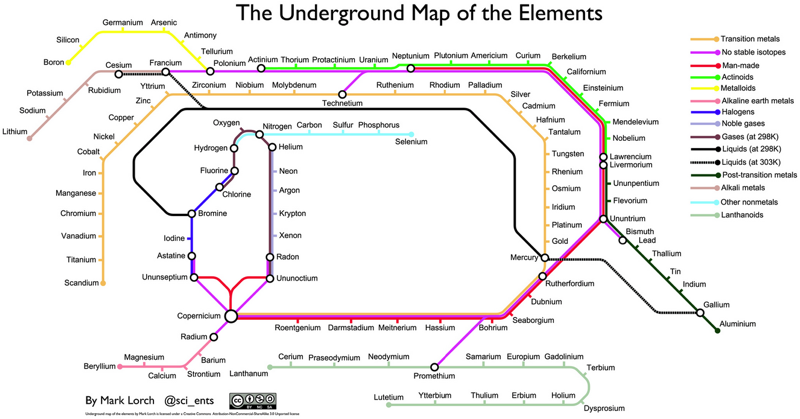
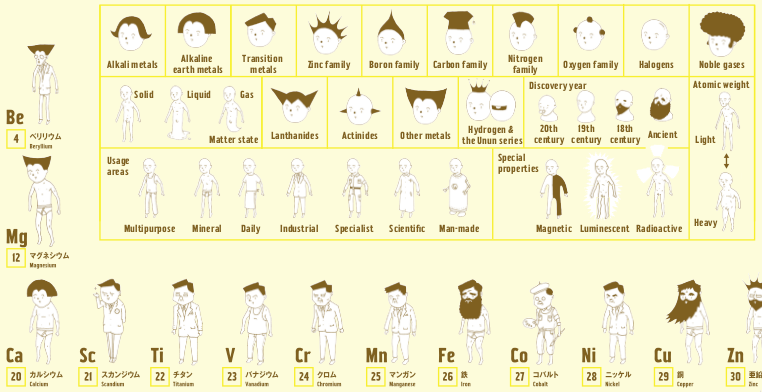
Periodic Table |
 |
 |
 |
 |
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
The INTERNET Database of Periodic Tables
There are thousands of periodic tables in web space, but this is the only comprehensive database of periodic tables & periodic system formulations. If you know of an interesting periodic table that is missing, please contact the database curator: Mark R. Leach Ph.D. The database holds information on periodic tables, the discovery of the elements, the elucidation of atomic weights and the discovery of atomic structure (and much, much more).
Miscellaneous Periodic Tables:
| Year: 2010 | PT id = 1338, Type = misc |
元素 (The Elements song in Japanese)
| Year: 2012 | PT id = 482, Type = misc |
3 Year-Old Sings Tom Lehrer's Elements Song
Rose turned 3 in November. It's been a little over a year since her initial elements video.
She's still interested in elements, but not so much by playing the cards anymore, mostly via this song:
| Year: 2010 | PT id = 279, Type = formulation misc 3D |
3-D Strange Periodic Table
As Lewis Page of The Register puts it: "Top flight international reverse-alchemy boffins say they have managed to transmute gold into an entirely new form of 'negatively strange' antihypernucleic antimatter...", here.
The effect is to add a third dimension of quark strangeness to the periodic table. Read the abstract by the STAR Collaboration.
| Year: 2012 | PT id = 480, Type = data misc |
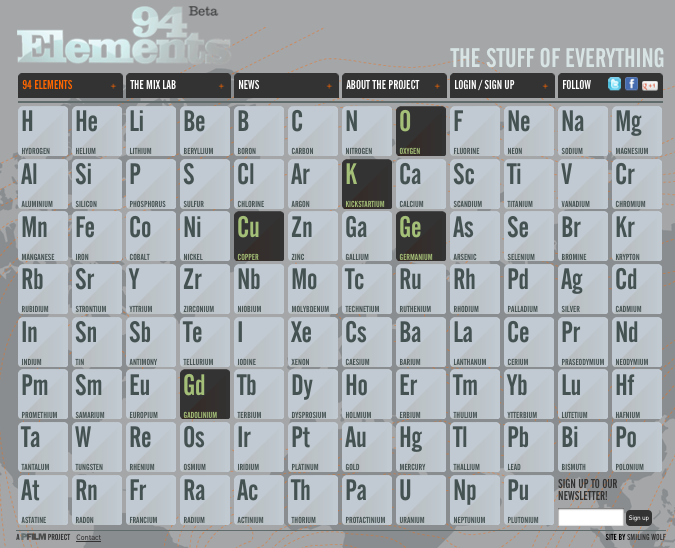
94 Elements: The Stuff of Everything
There are 94 naturally occuring elements, from hydrogen to plutonium. Together they make up everything in the world.
94 Elements is a global filmmaking project, exploring our lives through the lens of the elements. Everything that surrounds us is made from these 94 building blocks, each with its own properties and personality. Our own bodies are mostly made from just 6 of them.
The stories of the elements are the stories of our own lives. They reveal the patterns of our economies and the state of our relationships with our natural resources. The project is in part a celebration of the art of documentary film and some of the best filmmakers working today are making new films for the project. There'll also be opportunities for talented new and emerging filmmakers and animators to pitch their own films, with the winners chosen by you - the project community.
| Year: 1970 | PT id = 321, Type = formulation misc data |
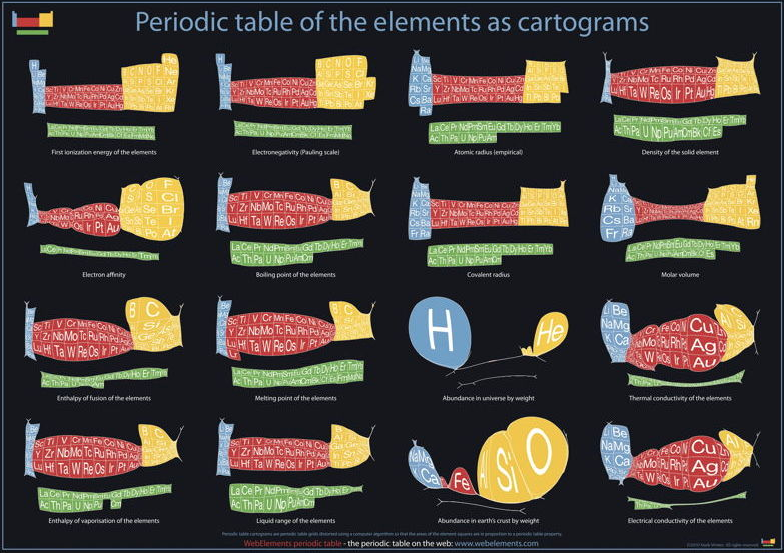
Abundance of the Elements
A 1970 periodic table by Prof. Wm. F. Sheehan of the University of Santa Clara that claims to show the elements according to relative abundance at the Earth's surface. [However, we dispute the relative areas given to the various elements; there is almost no helium at the Earth's surface, for example.] Click the image to enlarge:
Below are some cartiogram representations, including the relative abundance of the elements in the Earth's crust, from Mark Winter's WebElements website:
| Year: 2016 | PT id = 934, Type = misc |
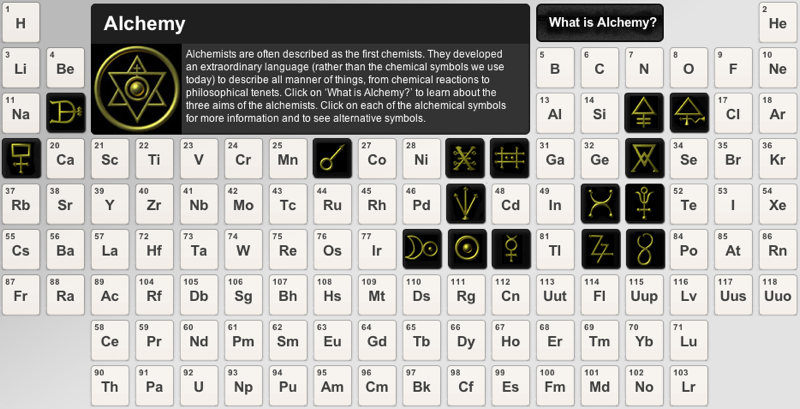
Alchemical Table of Symbols
The Alchemical Table of Symbols was designed by Aristotle Pramagioulis of egregoredesign. The periodic table is available as a poster and many other forms:
Thanks to Fathi Habashi for the tip!
| Year: 2019 | PT id = 982, Type = formulation misc |
Archetypes of Periodic Law
Archetypes of Periodic Law by Dmitry Weise, read more on the website.
One of the creators of quantum mechanics Wolfgang Ernst Pauli wrote in his work The Influence of Archetypal Ideas on the Scientific Theories of Kepler (1948):
"The process of understanding nature as well as the happiness that man feels in understanding – that is, in the conscious realization or new knowledge – seems thus to be based on a correspondence, a 'matching' of inner images pre-existent in the human psyche with external objects and their behavior. This interpretation of scientific knowledge, of course, goes back to Plato and is, as we shall see, advocated very clearly by Kepler. These primary images, which the soul can perceive with the aid of an innate 'instinct', are called by Kepler archetypal. Their agreement with the 'primordial images' or archetypes introduced into modern psychology by C. G. Jung and functioning as 'instincts of imagination' is very extensive. A true spiritual descendant of the Pythagoreans, he attached the utmost importance to geometric claiming that its theorems 'have been in the spirit of God since eternity'. His basic principle was: 'Geometria est archetypus pulchritudinis mundi' (Geometry is the archetype of the beauty of the world)."
Dmitry writes:
"The key archetype, in our opinion, is the concept of the square and its gnomon. This is due to the well-known fact that the electron filled shell contains 2n2 electrons, and the number of electrons on the subshell is twice the odd number; the gnomon of the square. Triangle, tetrahedron, square pyramid, octahedron, pyramid-like figures composed of square layers are also considered. The methodical concept for these constructions is the figurate numbers, actively studied by the Pythagoreans. The tables of the periodic law built on the motifs of ancient folk and modern ornaments take a special place. They include not only geometric archetypes, but also magic-symbolic, cultural and religious archetypes of the collective unconscious. Note that the periodic law table, built on the basis of the Native American ornament, surpasses the modern Mendeleev table in the parameter reflecting quantum numbers in its structure."
Note the final photograph below shows Prof. Martyn Poliakoff of The University on Nottingham and Periodic Videos:



| Year: 2025 | PT id = 1335, Type = formulation misc |
Armenian Periodic Table
An Armemian periodic table given to a Belgian student in Dubai. The title reads: Periodic Table of Chemical Elements by D. Mendeleev. This PT is based on Mendelejeff's (Meneleev's) 1880 formulation.
Koen Binnemans writes:
"What attracted my attention was the inclusion of the transactinides in the short-form periodic table. This was the first time I saw this."

Thanks to Koen Binnemans and his son Alexander Binnemans for the tip!
| Year: 2015 | PT id = 711, Type = misc non-chem |
Art of The Elements
An Exhibition "Periodic Tales: The Art of the Elements", the Compton Verney Gallery, 3 October 2015 to 13 December 2015
"The iconic periodic table represents the ultimate expression of order, containing the volatile elements in rows and columns. This exhibition explores a selection of the elements drawn from the periodic table (neon, uranium, gold, silver, carbon, iron, copper, mercury, colbolt, aluminium, sulphur, bronze, tin, lead, calcium) and looks at how artists have used them and their cultural meanings in their art.
"Inside the exhibition you will experience the elements in unique and unexpected ways through historic and contemporary works by artists including Eduardo Paolozzi, Joseph Beuys, Joseph Wright of Derby, John Constable, Antony Gormley, Cornelia Parker, Marc Quinn, Lucy Skaer, Danny Lane, Bill Woodrow, Maria Lalic, Fiona Banner, Thomas Heatherwick, David Nash, Ken + Julia Yonetani and Roger Hiorns.
There are also two new commissions. A stunning neon work by Tim Etchells and a thoughtful carbon sculpture by Annie Cattrell."
The show is reviewed in New Scientist.
Thanks to Marcus Lynch for the tip!
| Year: 2017 | PT id = 916, Type = misc |
Atomic Nuclei Periodic Table
From the Pyramids on Nuclei of Elements blog, a periodic table of atomic nuclei using 'pyramidal cube theory':
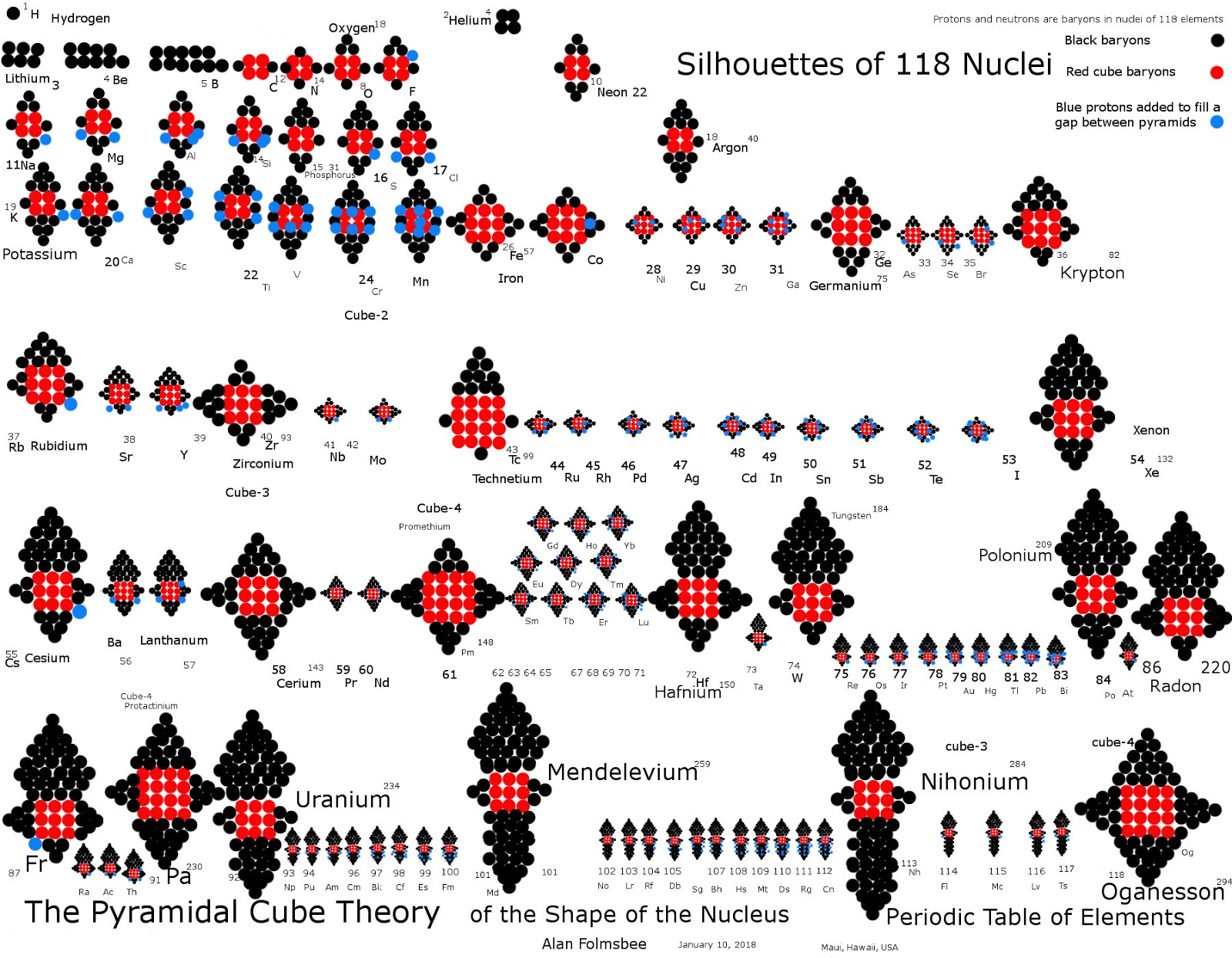
Click image below (updated in 2020) to enlarge:
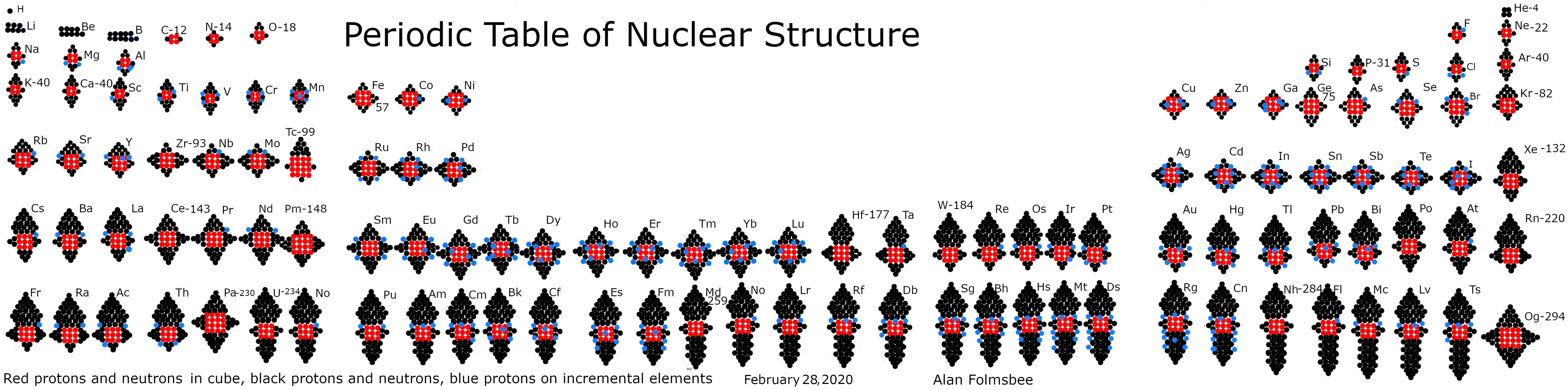
| Year: 2012 | PT id = 925, Type = misc data |
Atoms, Orbitals & The Periodic Table
One of several animations and explanations/realisations of quantum physics from Data-Burger, scientific advisor: J. Bobroff, with the support of: Univ. Paris Sud, SFP, Triangle de la Physique, PALM, Sciences à l'Ecole, ICAM-I2CAM.
Mark Leach writes:
"What I particularly like about this video is that it shows the quantum fuzziness of the atoms. This explains/shows how and why induced-dipole/induced-dipole (London force) interactions occur, an important class of van der Waals interaction. At any moment, the electron distribution is not perfectly spherical, which means that there is an instantaneous dipole on the atom. This instantaneous dipole is able to induce a dipole on an adjacent atom, with the effect that the two atoms are attracted when they touch. It is as if atoms are 'sticky' like Velcro.
"This effect explains why the Group 18 noble gas elements are able to form liquids and solids [not He] at low temperatures, and why non-polar molecules, such as P4, S8 and hydrocarbons are able to condense."
| Year: 2011 | PT id = 448, Type = misc |
BASF Periodic Table
A BASF advert showing a periodic table of school children:
| Year: 2018 | PT id = 985, Type = misc |
Better Call Saul - Gale sings The Elements
The Better Call Saul Season 4 Episode 3 Clip starring David Costabile singing The Elements Song:
Thanks to Conal for the tip!
| Year: 2004 | PT id = 143, Type = data misc |
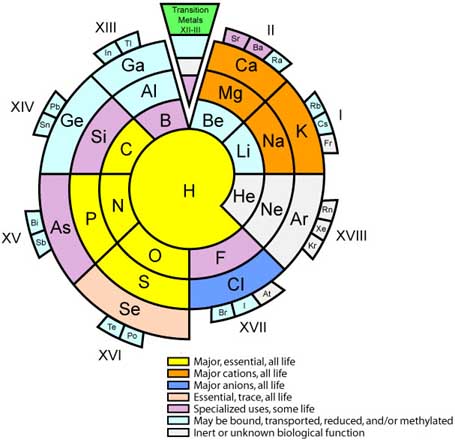
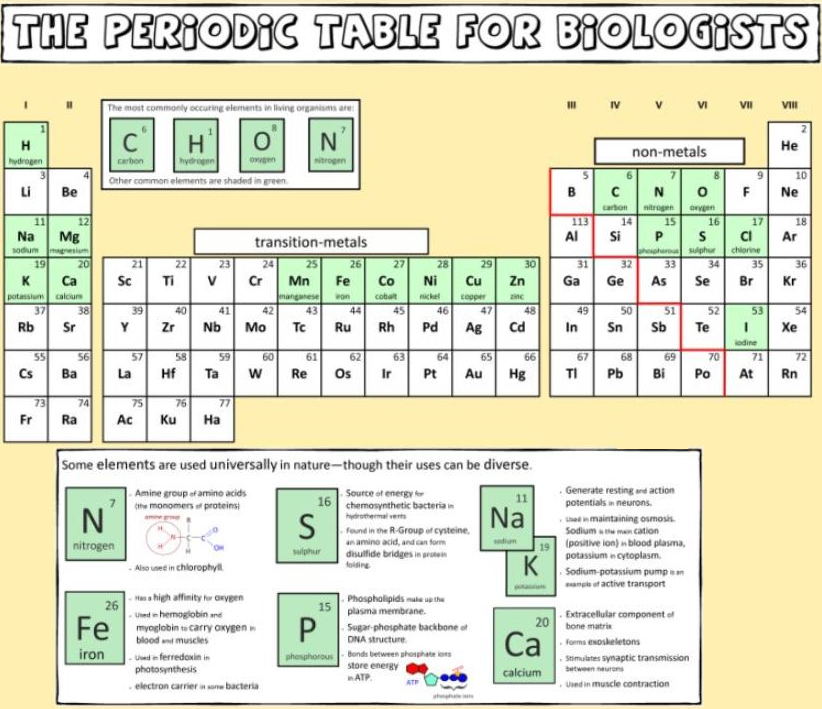
Biologist's Periodic Tables
A periodic table showing where biologically essential (green), essential trace (purple), toxic (red), radioactive (yellow) and of low – but not zero– biological impact (gray) elements are found. Only highly toxic elements are shown in red. Li (as Li+) is biologically active and is used as an antidepressant.
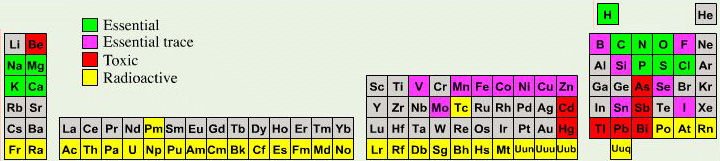
By Mark Leach
or here:
And a periodic table for biologists from Science Videos:
| Year: 2019 | PT id = 1060, Type = data misc review |
Bloomberg Businessweek Special Issue: The Elements
A Bloomberg Businessweek Special Issue on The Elements.
Using state of the art [2019] web graphics, and packed with interesting business stories:
Thanks to Eric Scerri for the tip!
See the website EricScerri.com and Eric's Twitter Feed.
| Year: 2008 | PT id = 333, Type = misc review |
Braille Guidebook Interactive Periodic Table Study Set
Azer's Interactive Periodic Table Study Set is designed to make learning about the Periodic Table of the Elements accessible to students with visual impairments or blindness.
The tangible materials included with this study set complement APH's Periodic Table of the Elements Reference Chart and allow students to enhance their understanding of concepts consistent with the National Science Standards.
Inspired by Samir Azer, a science teacher at the Kentucky School for the Blind, this set can assist in the instruction and demonstration of concepts related to the arrangement of the periodic table, atomic structure, ionic and covalent bonding, and balancing of chemical equations to students who benefit from a hands-on, interactive model.
Special attention was given to make the materials tactually discriminable and visually appealing to the target population, yet appropriate for all students regardless of visual acuity:
| Year: 2013 | PT id = 617, Type = misc |
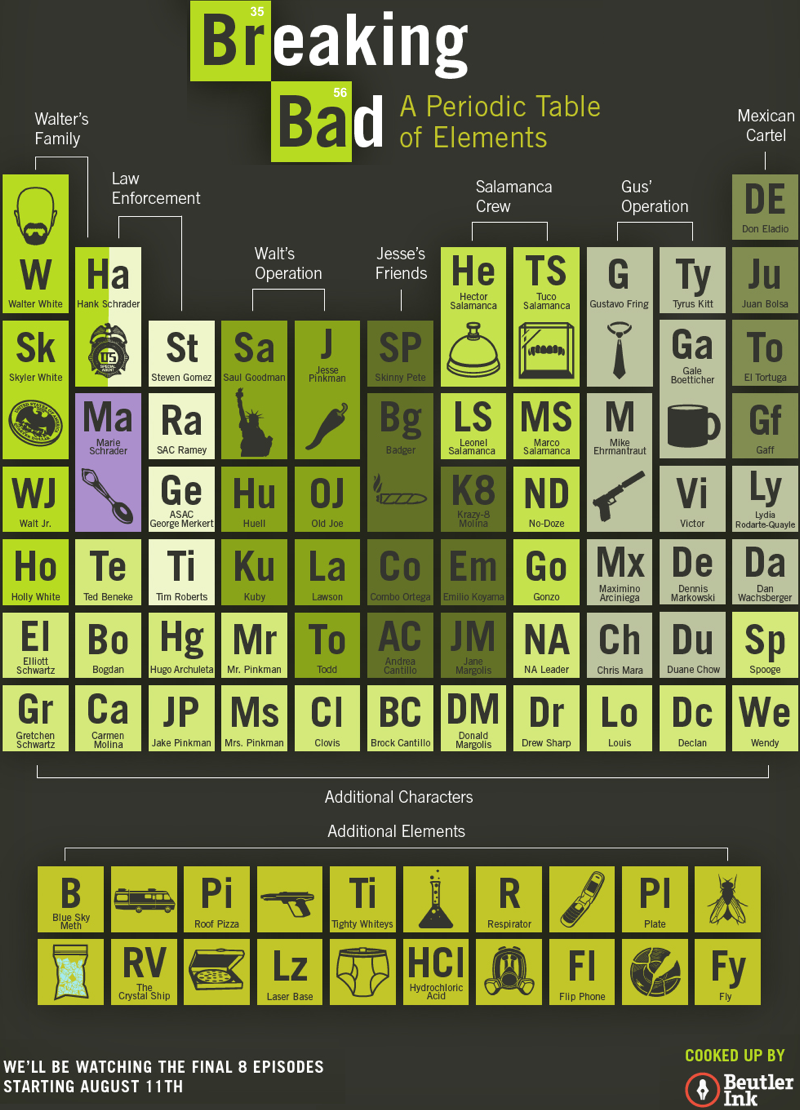
Breaking Bad Periodic Table
The TV series Breaking Bad uses Br (35) and Ba (56) in the logo, and Beutler Ink have constructed a full periodic table or characters and 'additional elements':
| Year: 2014 | PT id = 644, Type = misc |
Breaking Bad Periodic Table
More Breaking Bad PT images:
Thanks to Eric Scerri for the tip!
See the website EricScerri.com and Eric's Twitter Feed.
| Year: 2015 | PT id = 712, Type = misc |
Brielle, 3 Years Old, Recites The Periodic Table of Elements!
From Ellentube:
| Year: 2012 | PT id = 553, Type = misc |
Building Block Elements
From Think Geek, element building blocks... so you can build your own PT Formulation:
| Year: 2007 | PT id = 161, Type = misc |
Bus Periodic Table
A bus dressed as a Periodic Table used to advertise The Oxford Science Park:
And a Taxi:

| Year: 2024 | PT id = 1299, Type = misc data |
Can I Lick It? Periodic Table
From Reddit, a "A cool guide to element lickability".
Mark Leach writes:
"I think I would colour calcium yellow as it bubbles hydrogen gas when added to water."
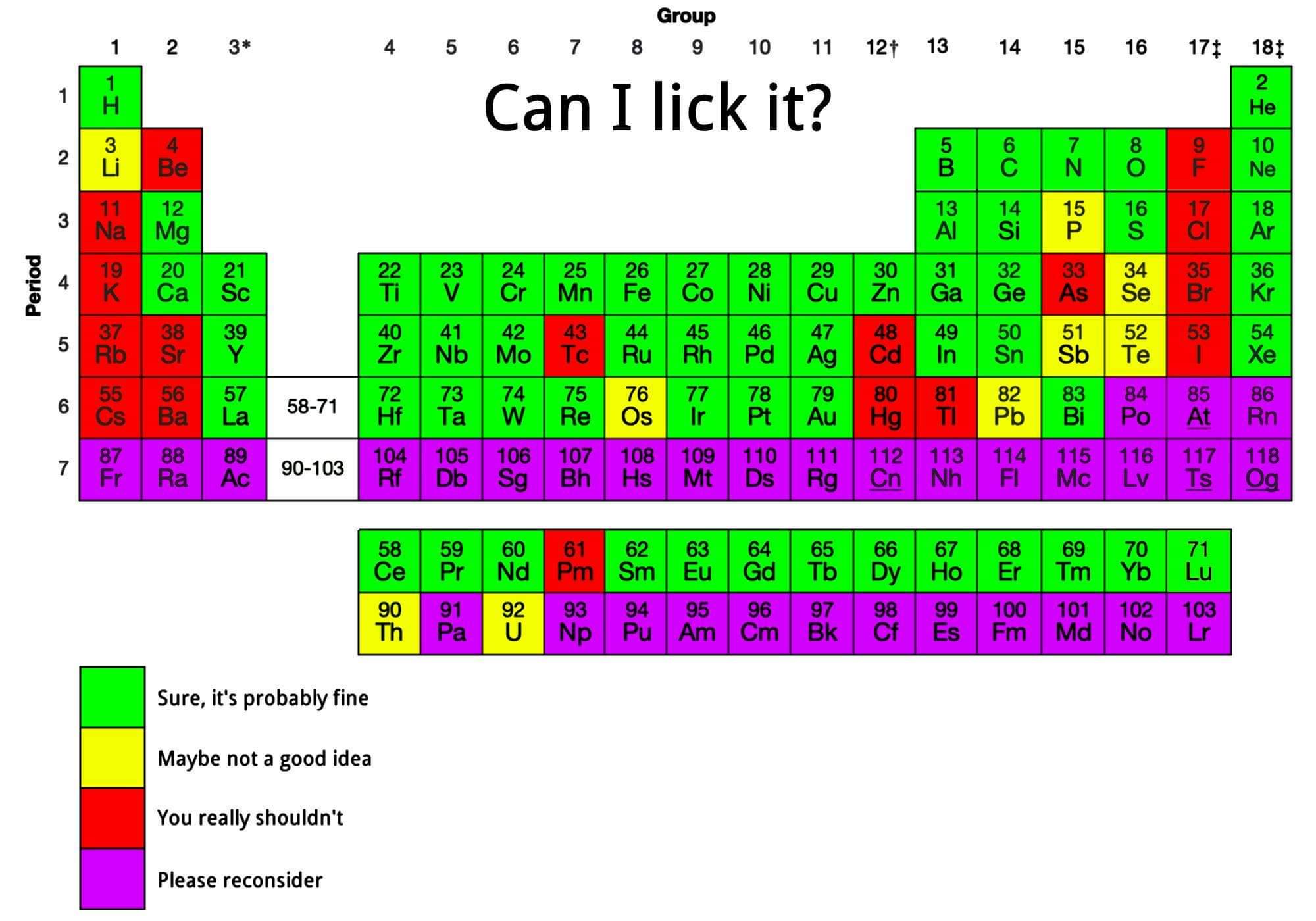
Thanks to René Vernon for the tip!
| Year: 2011 | PT id = 446, Type = misc |
Cartoon about The Elements
A cartoon about the elements from xkcd.com:
| Year: 2019 | PT id = 1036, Type = misc |
Celebrate 150 Years Of The Periodic Table By Tying 200,000 Tiny Knots
Jane Stewart decided to Celebrate 150 Years Of The Periodic Table By Tying 200,000 Tiny Knots:



| Year: 1992 | PT id = 1045, Type = formulation misc review |
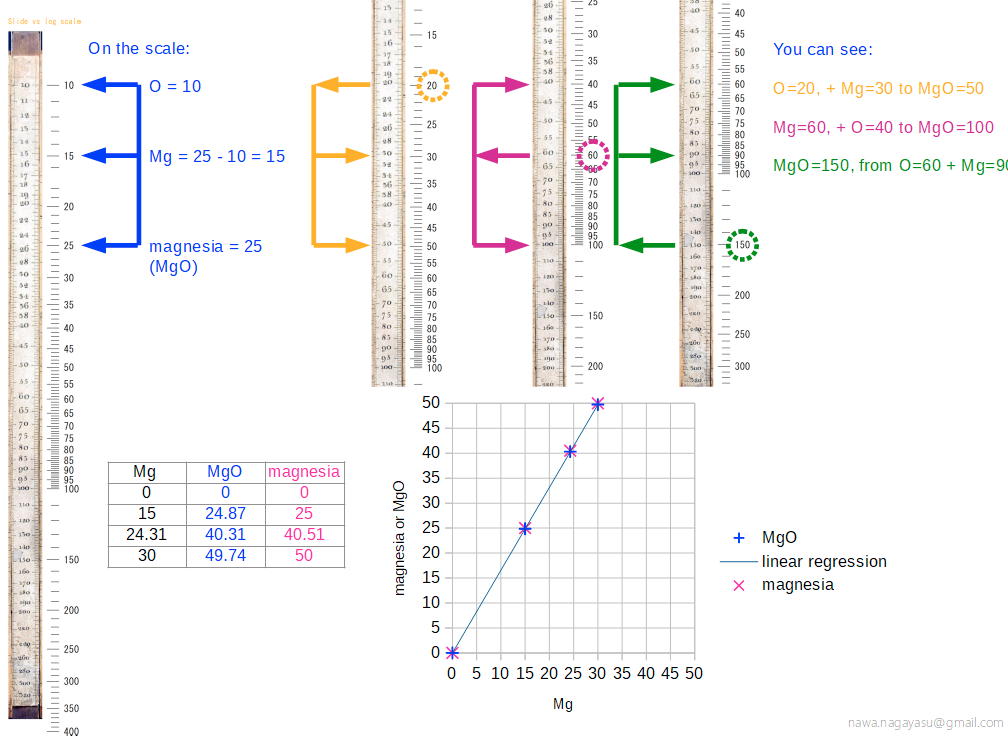
Chemical Slide Rules
The first chemical slide rules are of interest here because they are, in effect, early periodic tables. But the are more than this, as they can be used for performing chemical calculations. Writing in Bull. Hist. Chem. 12 (1992) (and here), William D. Williams of Harding University writes:
"An article by George Bodner in the Winter 1990 issue of the Bulletin described a rare chemical slide rule designed by Lewis C. Beck and Joseph Henry - their little-known Improved Scale of Chemical Equivalents. [My] paper attempts to place this slide rule in context by describing its origins, as well as some of its predecessors and successors."
Some chemical slide rules mentioned in the text:

- Chemist's Adjustable Duplex Slide Rule made by Keuffel & Esser Co., n.d., ca. 1936-1940. Here are the full instructions for use.
Nagayasu Nawa writes and provides an explanation as how Wollaston's chemical equivalents slide rules should be used:
"It is very interesting slide rule for me. Because we actually used slide rule in 1960s. There were not the electronic calculator in the world. I think it would be used as a simple slide rule of The Law of Definite Proportions by J.L. Proust 1799."
- '10 water', for example, may be hydrating water in chemical compound
- 'Chlorine' may be HClO: HCl(35) + O(10) = HClO(45), etc.
Click image to enlarge:
Thanks to Nawa for the tip!
| Year: 2016 | PT id = 755, Type = misc |
Chemistry Eye Chart
From Cascadia Press, an Eye Chart for Chemists:
Thanks to Roy Alexander for the tip!
| Year: 1993 | PT id = 905, Type = formulation misc |
Chemistry Imagined: The Periodic Table
From Roald Hoffmann & Vivian Torrence's book, Chemistry Imagined: Reflections of Science, a picture entitled The Periodic Table:
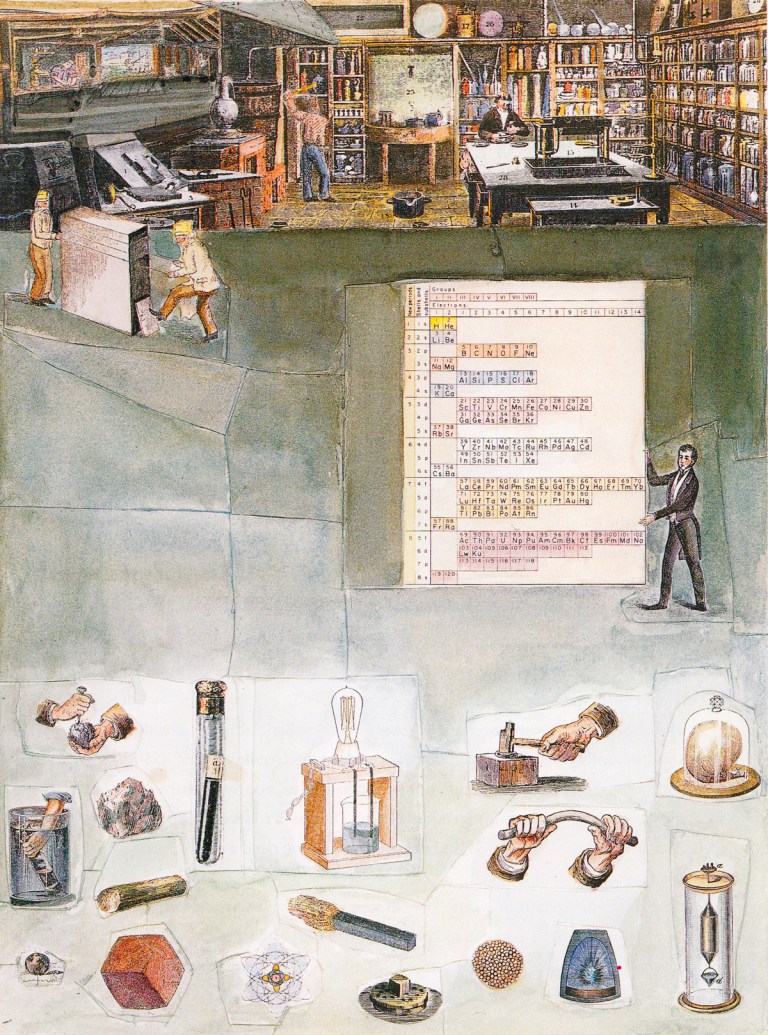

Thanks to Marcus Lynch for the tip!
| Year: 2012 | PT id = 518, Type = misc |
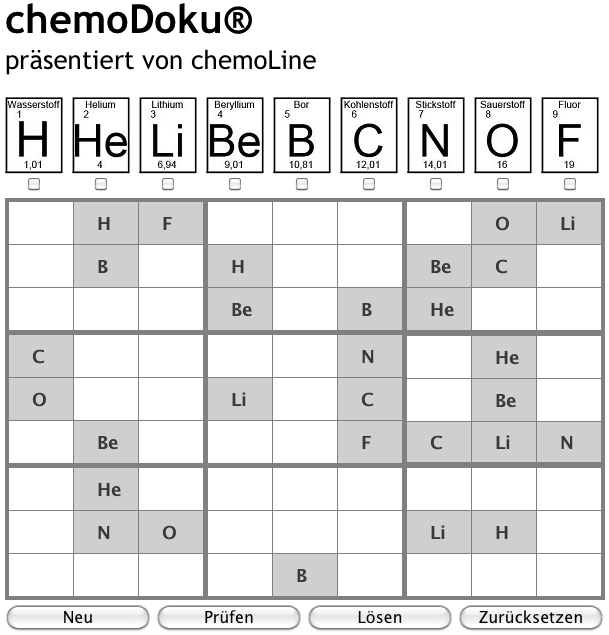
chemoDoku
Chemoline an on-line shop, based in Germany with a multilingual site (click the flag), sells various science artifacts that feature the periodic table.
The site also has an on-line chemical element version of Sudoku called chemoDoku, click to play:
| Year: 2021 | PT id = 1179, Type = data review misc |
Chemogenesis In 700 Seconds
The Chemogenesis analysis – by Mark Leach – tells the story of how chemical structure and reactivity emerge from the periodic table of the elements. This video is a rapid dash through the story... which unfolds over many pages of the Chemogenesis Web Book.
| Year: 2012 | PT id = 517, Type = misc |
Chemoline Shop
Chemoline is an on-line shop, based in Germany with a multilingual site (click the flag).
The shop sells various science artifacts. Several products feature the periodic table, including:
| Year: 2002 | PT id = 55, Type = misc |
Chinese Character Periodic Tables
Chinese character periodic tables, here, here & here:
| Year: 2015 | PT id = 703, Type = misc |
Names of the Chemical Elements in Chinese
An interesting Language Log web page that discusses the chemical elements in chinese.
Thanks to Marcus Lynch for the tip!
| Year: 2018 | PT id = 1144, Type = misc non-chem |
Christmas Periodic Table of Substances
By Tom Gauld for New Scientist 22/29 December 2018 pp90:

| Year: 2021 | PT id = 1180, Type = misc |
Christmas Tree Periodic Table
We have developed a new model in the form of a Christmas tree to represent periodic table. We have tried to combine both the triangle model of periodic table and modern periodic table in form of a christmas tree.
Mr G. Gnanamani Simiyon
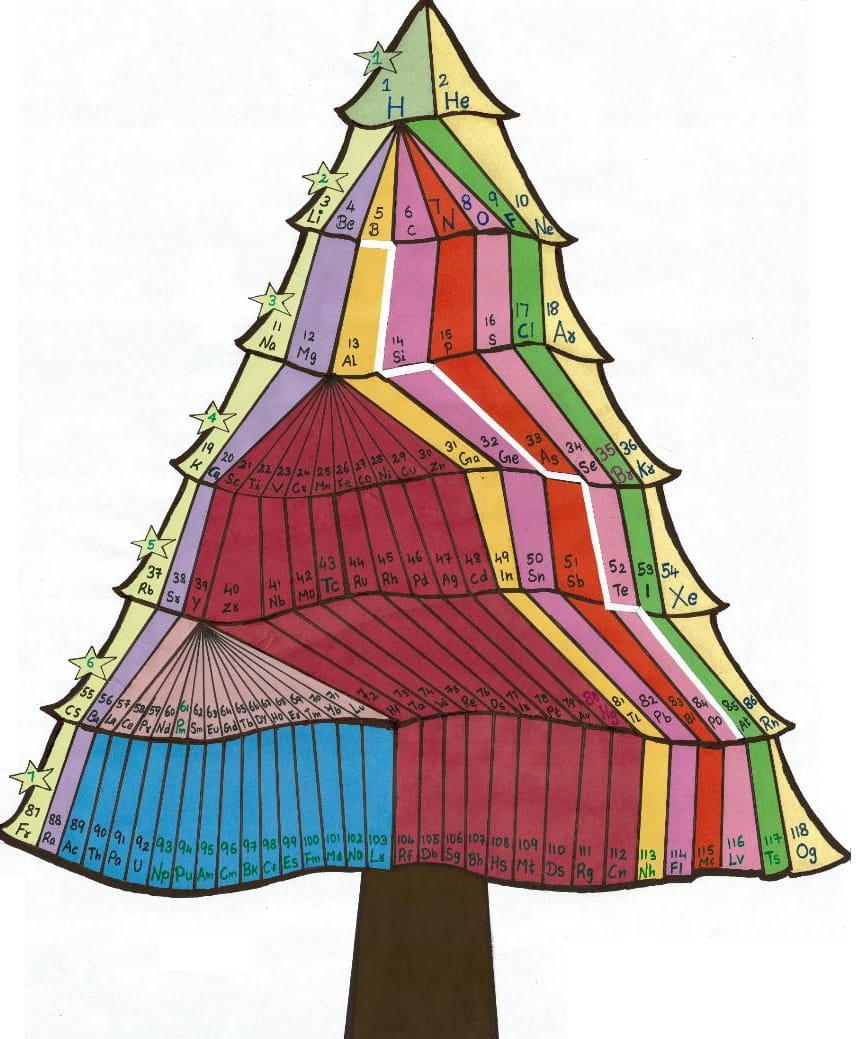
| Year: 2010 | PT id = 523, Type = misc |
Circlon Model of Nuclear Structure & Periodic Table
The complete nature and description of The Circlon Model of Nuclear Structure is contained in the book The Other Theory of Nuclear Physics available from www.living-universe.com. However, for the purpose of understanding nuclear structure it is only necessary to assume that the components of nuclear structure (protons, mesons, and neutrons) are all composed of hollow, ring-shaped, mechanical particles called Circlons that are held together within the nucleus by their physical shapes.
Within the nucleus, the proton and the meson are always connected in a two piece unit called a Promestone. The proton encircles the ring-shaped body of the meson, and the neutrons fit inside of the meson's hollow body and can only be located at four places within the meson's body called nucleon receptors. A proton is always located at one of a meson's nucleon receptors. One Promestone makes up the nucleus of a hydrogen-1 atom and two Promestones plus two neutrons make up the helium-4 nucleus, also know as an alpha particle. An element's atomic number indicates the number of Promestones in its nucleus and an isotope's atomic weight indicates the total number of Promestones and neutrons in that particular nucleus.
Within the alpha particle that forms the center of each nucleus, a proton and a neutron are located at each junction where the two mesons intersect. However, when two mesons cross in other parts of the nucleus, each intersection can contain only one proton or one neutron (see nitrogen model above).
In the nucleon models displayed in each of the element boxes of the periodic table, the protons are represented by white circles and the neutrons are represented by white stars. The mesons are represented by ovals which take the color of the element that is formed by their addition to the nucleus:
| Year: 2024 | PT id = 1318, Type = misc |
Classical Periodic Table
A Classical Periodic Table from xkcd:

Thanks to Marcus for the tip!
| Year: 2010 | PT id = 217, Type = misc |
Classical Periodic Table
A periodic table of the classical elements: air, fire, earth, water & aether available as a t-shirt:

Or, just air, fire, earth, water, the 'old school' elements from here:
| Year: 2004 | PT id = 1096, Type = misc |
Classroom Kids Periodic Table
From a paper by René Vernon, a drawing of the elements as classroom personality kids, drawing by Richard Thompson 1957-2016.
From a National Geographic coffee table book: Curt Suplee, The New Everyday Science Explained, National Geographic Society, Washington DC, p. 130 (2004). The undated credit is given to Richard Thompson.
- Sodium is actively practicing a "salt-formation" passing play with corrosive chlorine, who devours the delivery.
- Potassium, magnesium and barium are having an active metal huddle.
- The two hydrogens are hot desking.
- Iron is reading a weightlifting workout book.
- Platinum introduces gold to iridium, all three being noble metals.
- Lead, as a heavy frontier metal, is playing air guitar.
- Carbon is having a link up with frontier metal silver (masquerading as a transition metal); an unidentified frontier metal; and two "other" nonmetals.
- Silicon, as a metalloid and a semiconductor, is catching up on an assignment.
- Helium and neon, having each had a cup of chamomile tea during the break, are sleeping.

| Year: 2014 | PT id = 671, Type = formulation spiral misc |
Clock Periodic Table
Prof. Martyn Poliakoff of the University of Nottingham, and star of the Periodic Videos YouTube Channel, explains how he was given a periodic table clock by a Japanese School teacher... which he likes very much:
Thanks to Eric Scerri for the tip!
See the website EricScerri.com and Eric's Twitter Feed
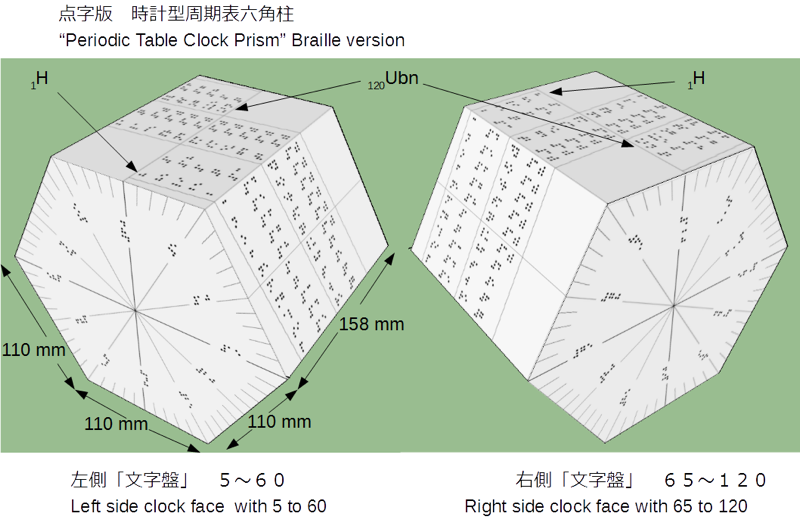
| Year: 2017 | PT id = 760, Type = formulation 3d misc |
Clock Prism Periodic Table, Braille Version
From the prolific Nagayasu Nawa, a Braille version of the Clock Prism periodic table:
| Year: 2012 | PT id = 458, Type = misc |
Coat of Arms Periodic Table
Amy Gramour has created a version of the Periodic Table that presents a coat of arms for each element. The attributes of the coats of arms symbolize the electron configuration and other selected features of each element.


| Year: 2014 | PT id = 648, Type = misc |
Coffee Table, Periodic Table Table
A Periodic Table Table - Coffee Table, from Bristol Design Forge via Folksy:
Thanks to Marcus Lynch for the tip!
| Year: 2004 | PT id = 968, Type = data misc |
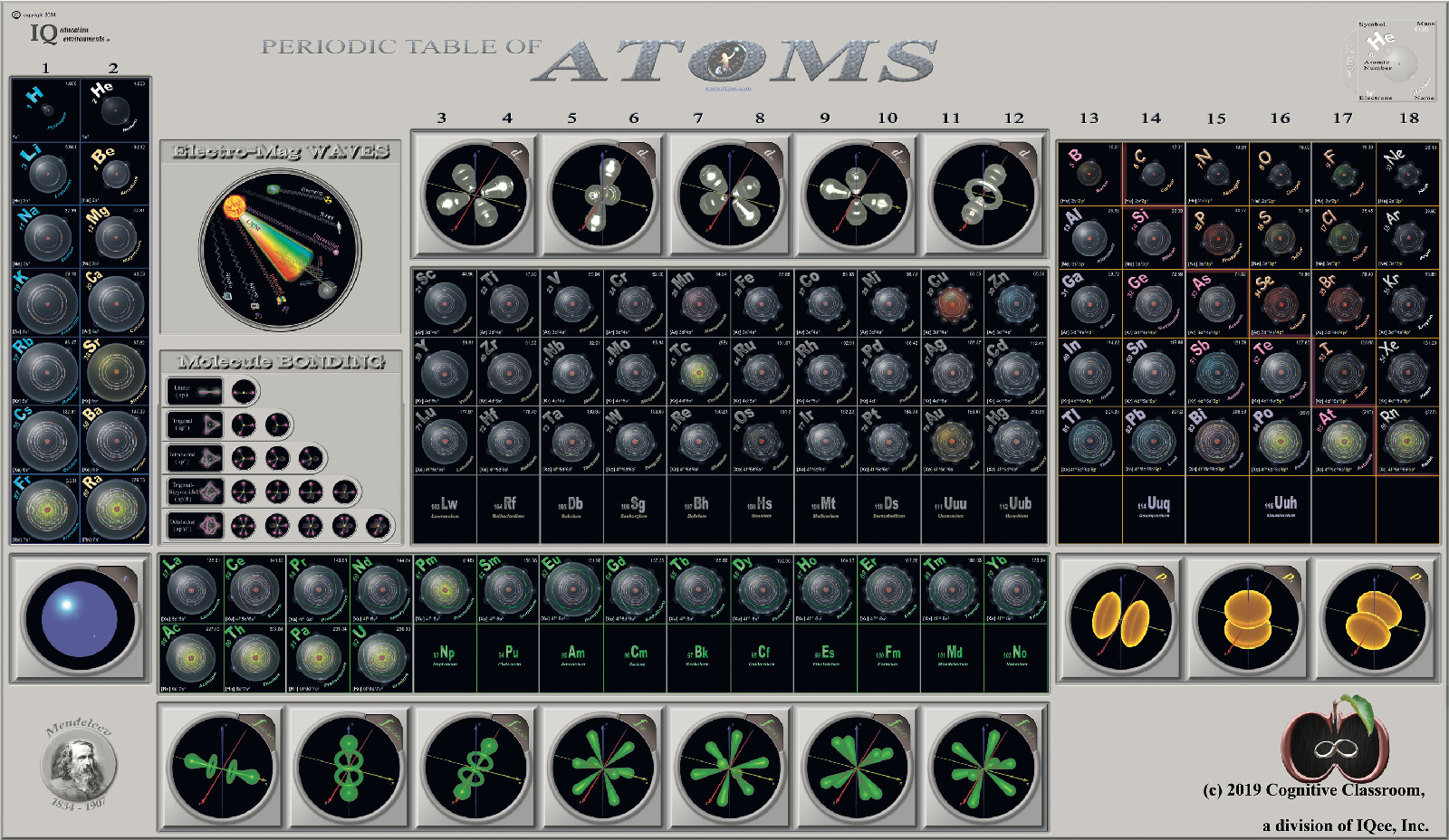
Cognitive Classroom's Periodic Table of Atoms
From Cognitive Classroom, a Periodic Table of Atoms. Richard Lambrecht writes:
"We have developed a visual periodic table that groups by orbitals, making He no longer contentious. But by including an orbital cloud, we give the student a great offset to the Bohr model used to place each and every single electron in the periodic table."
Click image or here to enlarge:
| Year: 2016 | PT id = 719, Type = data misc |
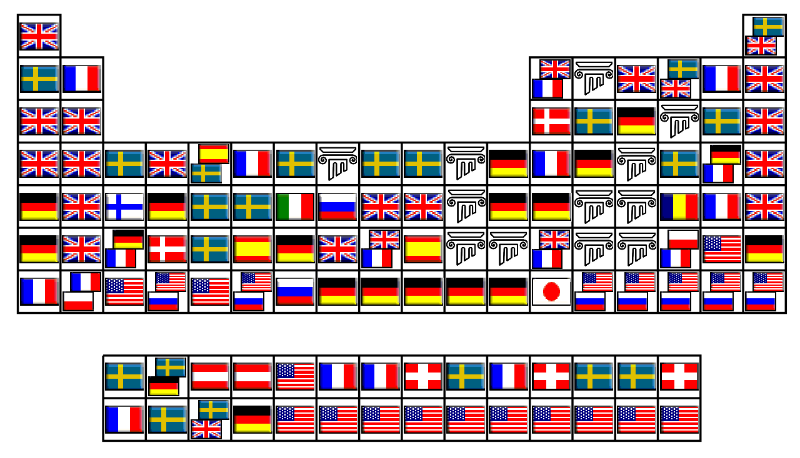
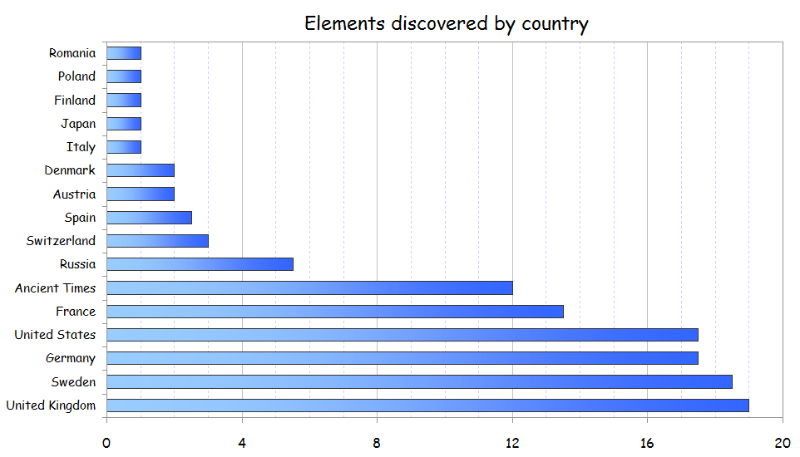
Collective Work of Chemists
From an article on LinkedIn:
Twelve elements were known from the Ancient Times, and were described by Romans and Greeks. The remaining 106 elements have been discovered by scientists of 15 different countries during the last 4 centuries. In addition, 19 elements of those 106 (18%) have been co-discovered by researchers of two countries.
Although some of them (like Bromine or Thallium) were isolated separately at the same time by chemists of different nationalities within the race to discover new elements in 18th-21st centuries, most of them have been obtained since then through collaborative research, like the recently discovered Ununpentium, Ununseptium and Ununoctium.
Another example is the isolation of Radium and Polonium by the Polish Maria Skłodowska-Curie and her French husband, Pierre Curie.
Thus, Periodic Table is the result of a collective and long-term work of hundreds of scientists.
It is noteworthy to see that Russia and United States have discovered mainly artificial elements.
| Year: 2010 | PT id = 374, Type = data misc |
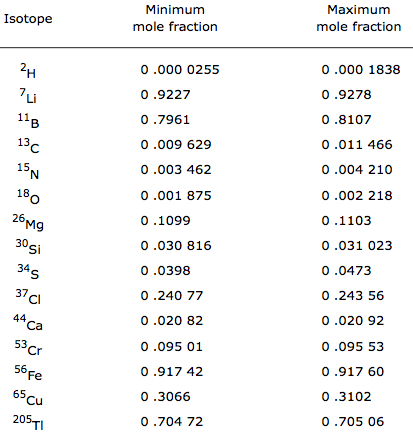
Compilation of Minimum and Maximum Isotope Ratios of Selected Elements
Documented variations in the isotopic compositions of some chemical elements are responsible for expanded uncertainties in the standard atomic weights published by the Commission on Atomic Weights and Isotopic Abundances of the International Union of Pure and Applied Chemistry.
This report summarizes reported variations in the isotopic compositions of 20 elements that are due to physical and chemical fractionation processes (not due to radioactive decay) and their effects on the standard atomic weight uncertainties. For 11 of those elements (hydrogen, lithium, boron, carbon, nitrogen, oxygen, silicon, sulfur, chlorine, copper, and selenium), standard atomic weight uncertainties have been assigned values that are substantially larger than analytical uncertainties because of common isotope abundance variations in materials of natural terrestrial origin. For 2 elements (chromium and thallium), recently reported isotope abundance variations potentially are large enough to result in future expansion of their atomic weight uncertainties. For 7 elements (magnesium, calcium, iron, zinc, molybdenum, palladium, and tellurium), documented isotope-abundance variations in materials of natural terrestrial origin are too small to have a significant effect on their standard atomic weight uncertainties.
Compilation of Minimum and Maximum Isotope Ratios of Selected Elements in Naturally Occurring Terrestrial Materials and Reagents
This report is available as a pdf.
U.S. GEOLOGICAL SURVEY
Water Resources Investigation Report 01-4222
| Year: 2002 | PT id = 220, Type = misc |
Corning Museum of Glass Periodic Table
A periodic table made from glassware at the Corning Museum of Glass:


| Year: 2014 | PT id = 635, Type = misc data |
Correspondences Between The Classical Thomson Problem and The Periodic Table of The Elements
By Tim (TJ) LaFave, a very detailed pdf discussing the correspondences between the classical Thomson Problem and the Periodic Table of the Elements. You will need to click thru and zoom in:
| Year: 2009 | PT id = 219, Type = misc |
Crab Periodic Table
A crab PT. I know nothing about this, other than this photograph found on a blog:

| Year: 2012 | PT id = 481, Type = misc |
Cupcakes, Periodic Table
From Kayla N. Green, Assistant Professor of Chemistry (Texas Christian University) comes a periodic table constructed from cupcakes baked for Chemistry Week 2012:
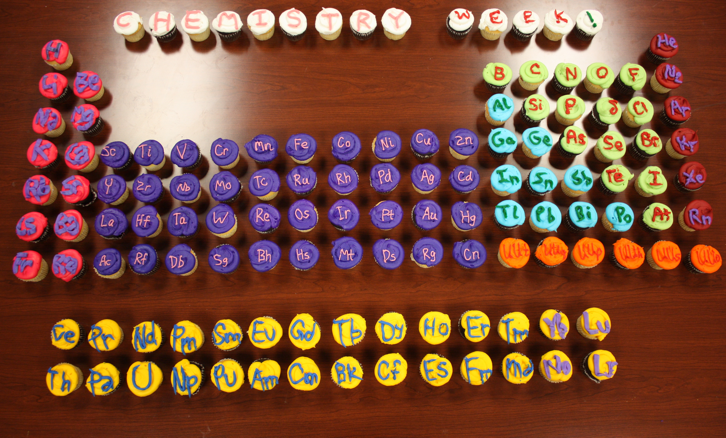
| Year: 2014 | PT id = 641, Type = misc |
Cutting Board Periodic Table
From Etsy, a Cutting Board periodic table:
Thanks to Marcus Lynch for the tip!
| Year: 2023 | PT id = 1274, Type = formulation misc non-chem |
DALL·E Pop Art Periodic Table
I asked DALL·E to generate a: "periodic table as pop art", and the AI produced this:

| Year: 2007 | PT id = 224, Type = misc non-chem |
Death Metal Periodic Table
| Year: 2013 | PT id = 612, Type = misc |
Don't Trust Atoms...
From Facebook... or buy the T-Shirt:
Thanks to Marcus Lynch for the tip!
| Year: 2011 | PT id = 581, Type = review misc |
Dufour's Periodic Tree: Two Short Films
Elsewhere in this database we can see the 1990 Dufour's Periodic Tree, now two short films have been made about this 3D formulation, here & here:
Eric Scerri Letter from Ben Ged Low on Vimeo.
Five Foot 3D Model from Ben Ged Low on Vimeo.
Thanks to Eric Scerri for the tip!
See the website EricScerri.com and Eric's Twitter Feed.
| Year: 2004 | PT id = 46, Type = misc |
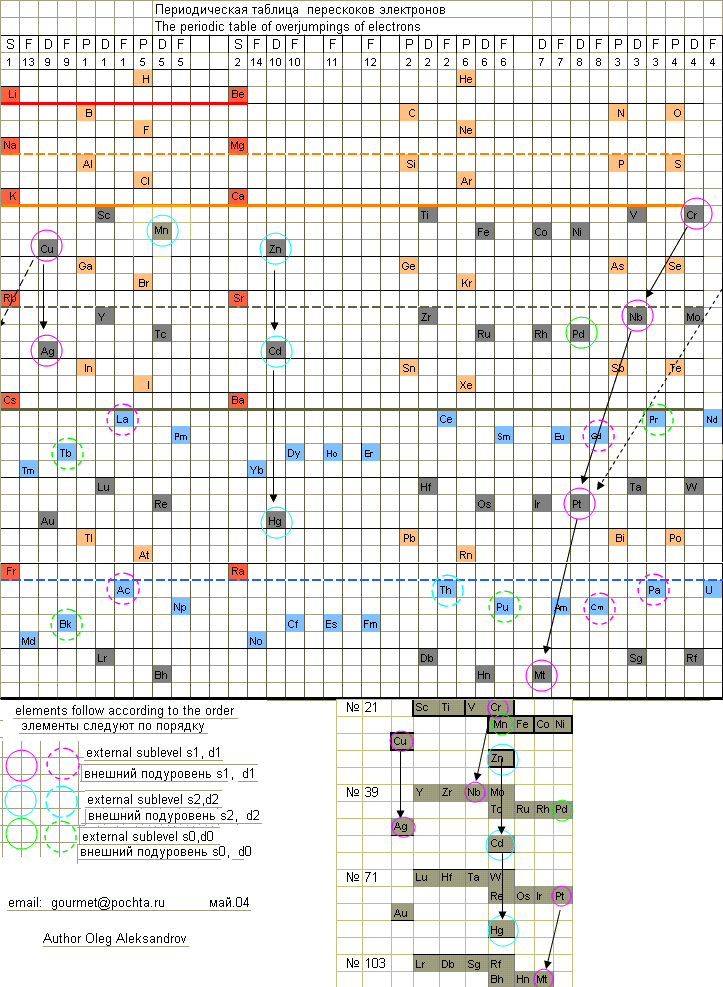
Electron Overjump Periodic Table
Here are some origional periodic table ideas, including history and electron overjumpings by Oleg Aleksandrov, from here.
| Year: 2011 | PT id = 381, Type = misc |
Element Game
Rose, a cute & smart 2 year old girl showing her excellent knowledge of the Periodic Table:
tnx to Boing Boing for the tip
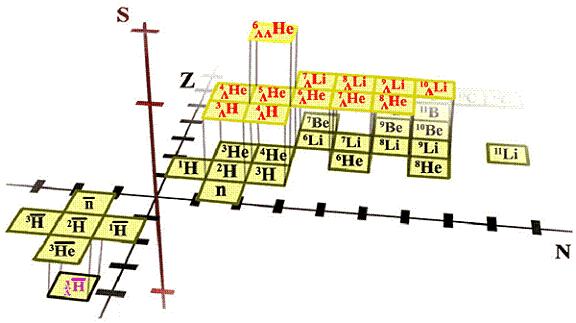
| Year: 1955 | PT id = 1086, Type = element misc data |
Element Hunters
A YouTube video, The Element Hunters.
The text accompanying the video says:
"Scientist in Berkeley discover new elements [Californium & Einsteinium] from hydrogen bomb debris in 1951 and then use the 60 inch Cyclotron to create Mendelevium, element 101. The team included Nobel Prize winner Glenn Seaborg and famed element hunter, Albert Ghiorso."
Thanks to Roy Alexander for the tip!
| Year: 2023 | PT id = 1283, Type = data misc non-chem element |
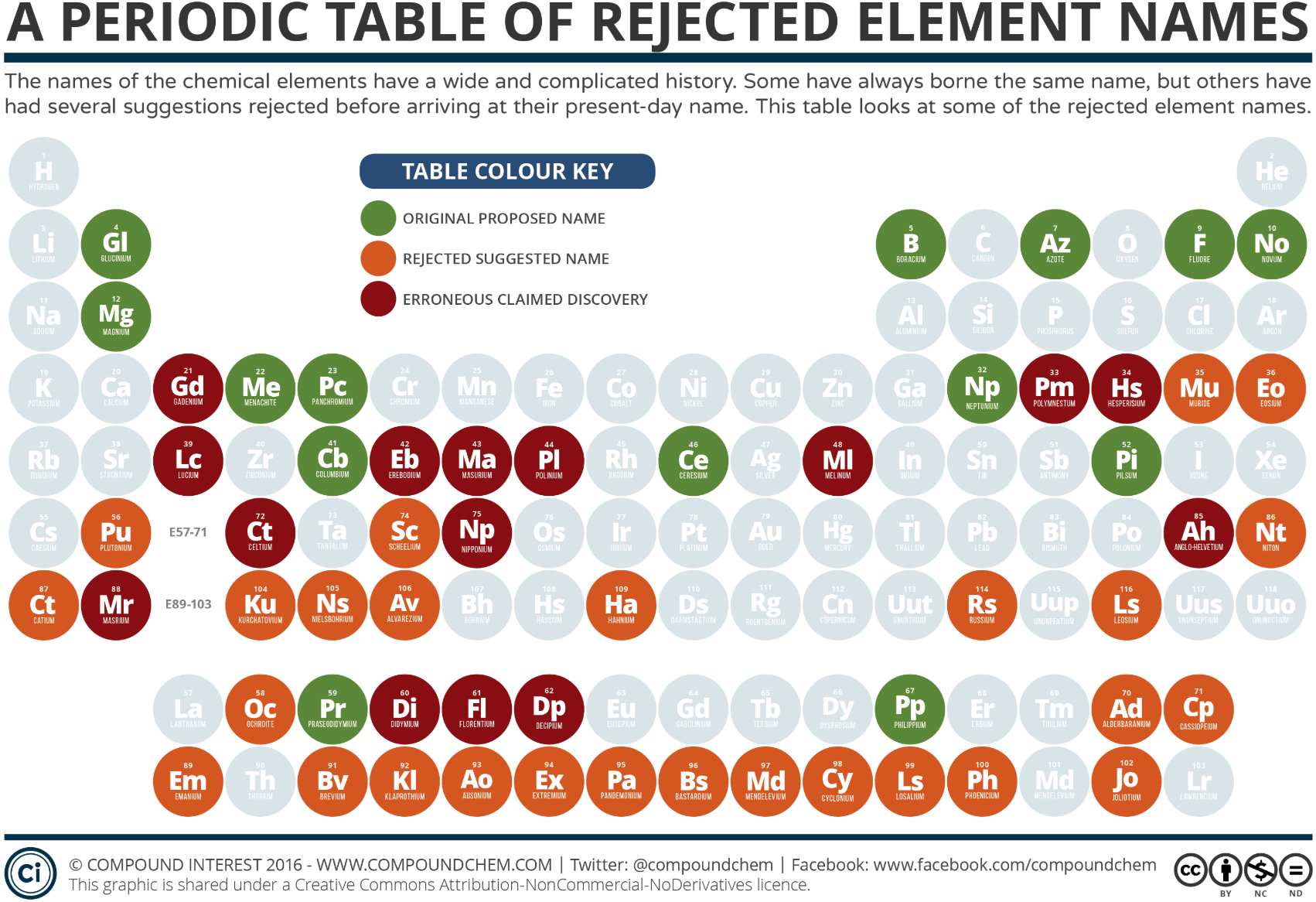
Element Names: The Etymology of The Periodic Table
An excellent video by RobWords about the names of the chemical elements and how they came about:
| Year: 2019 | PT id = 961, Type = data misc |
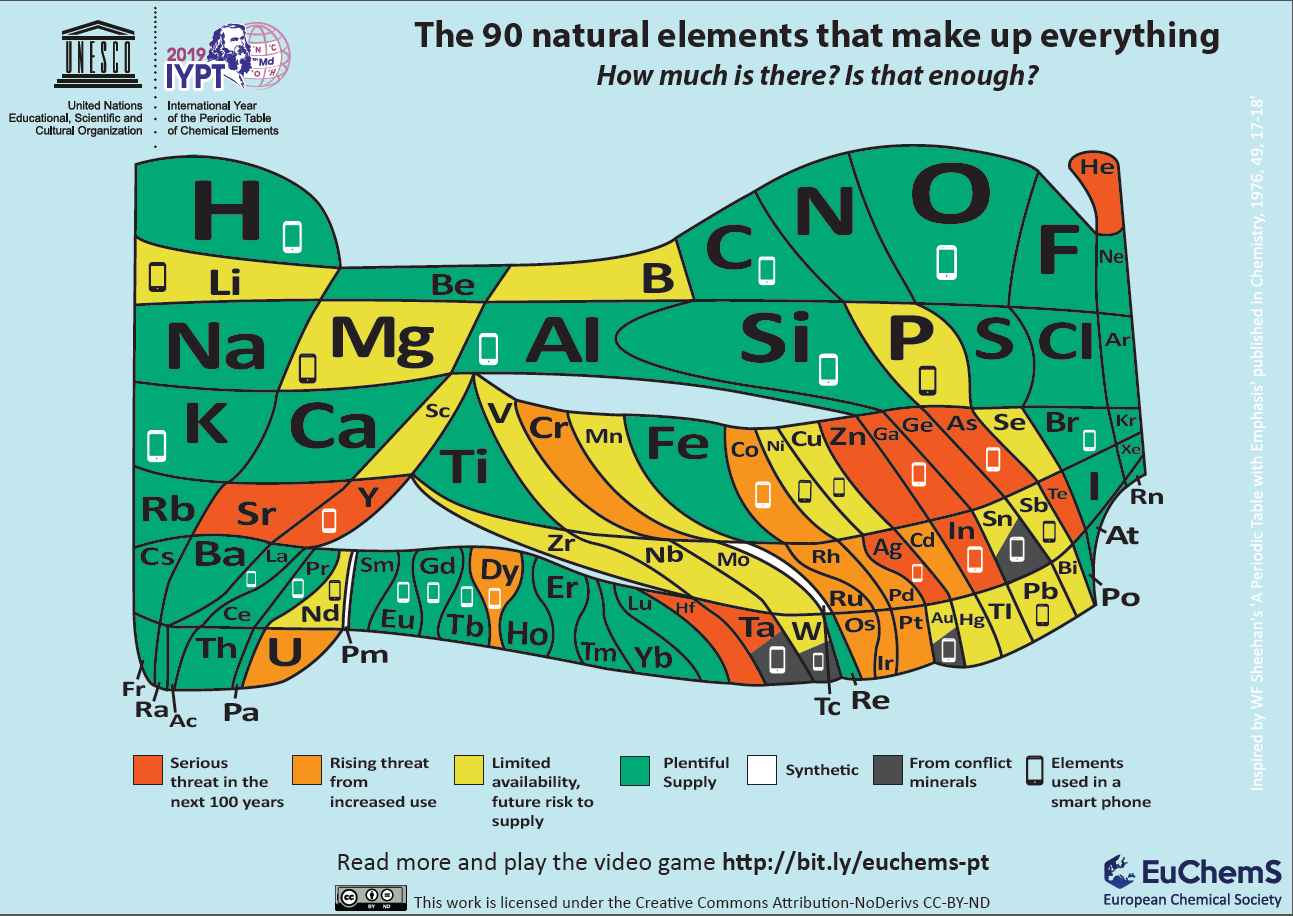
Element Scarcity, Periodic Table of
The European Chemical Society Periodic Table depicting element scarcity was unveiled and discussed at a EuChemS event in the European Parliament on Tuesday 22nd January 2019.
The event, chaired by MEPs Catherine Stihler and Clare Moody, presented an encompassing overview of what element scarcity means for us: both on a scientific level, but also economically and politically. A presentation from speaker Natalia Tarasova, IUPAC Past President, contextualised EuChemS' work within the celebrations of the International Year of the Periodic Table, whilst M Pilar Gil, from the University of St Andrews, delivered a remarkable and exhilarating talk on how the recently discovered oldest known wallchart of the Periodic Table was uncovered and dated.
An article in The Conversation, by David Cole-Hamilton of the University of St Andrews, uses this periodic table to look at elements that are overexploited in the modern world.
"Red indicates that dissipation will make the elements much less readily available in 100 years or less: helium (He), silver (Ag), tellurium (Te), gallium (Ga), germanium (Ge), strontium (Sr), yttrium (Y), zinc (Zn), indium (In), arsenic (As), hafnium (Hf) and tantalum (Ta).
"Helium is used to cool the magnets in MRI scanners and to dilute oxygen for deep sea diving. Vital rods in nuclear reactors use hafnium. Strontium salts are added to fireworks and flares to produce vivid red colours. Yttrium is a component of camera lenses to make them shock and heat resistant. It is also used in lasers and alloys. Gallium, meanwhile, is used to make very high-quality mirrors, light-emitting diodes and solar cells."
| Year: 2019 | PT id = 1124, Type = formulation misc |
Elements & Anti-Elements (Atom-to-Adam)
"The underlying order of the Atoms and asking the question of Intelligent Design has inspired my work. I have developed a new and improved Periodic Table of Elements that restores the Lanthanides and Actinides in their proper positions while also applying a complete and accurate numbering system. (1-8) numbering in s and p blocks correspond to Valence Electrons with (9-18) and (19-32) corresponding to core Electrons in the d and f block Elements.
"I have also inverted the Periodic Table of Elements which reflects the "missing" Anti Matter of our Universe. Unique to my Periodic Table of Elements is the ability to easily identify any Elements Electron Spin for Orbital Diagrams by shifting Elements up or down. Lastly I have highlighted some of the AUFBAU Exceptions or Electron Configuration anomalies in the transition metals. Interestingly, standing the Periodic Tables upright resembles the image of human beings, hence the title of my project, from Atom to Adam."
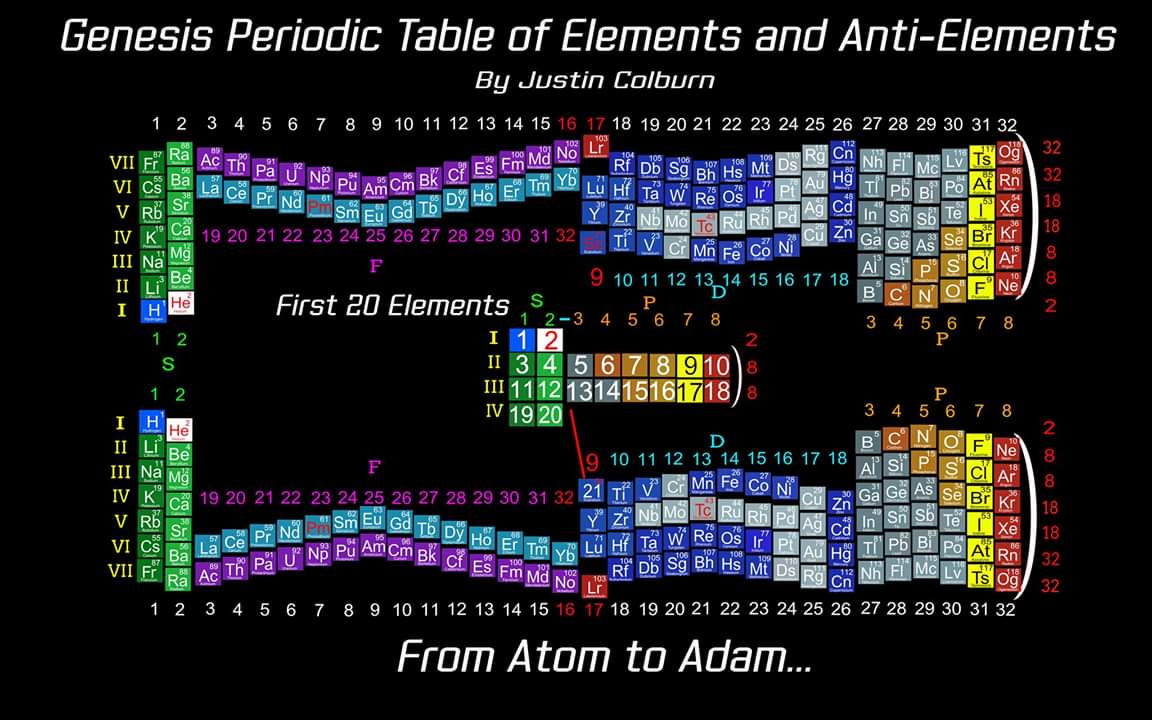
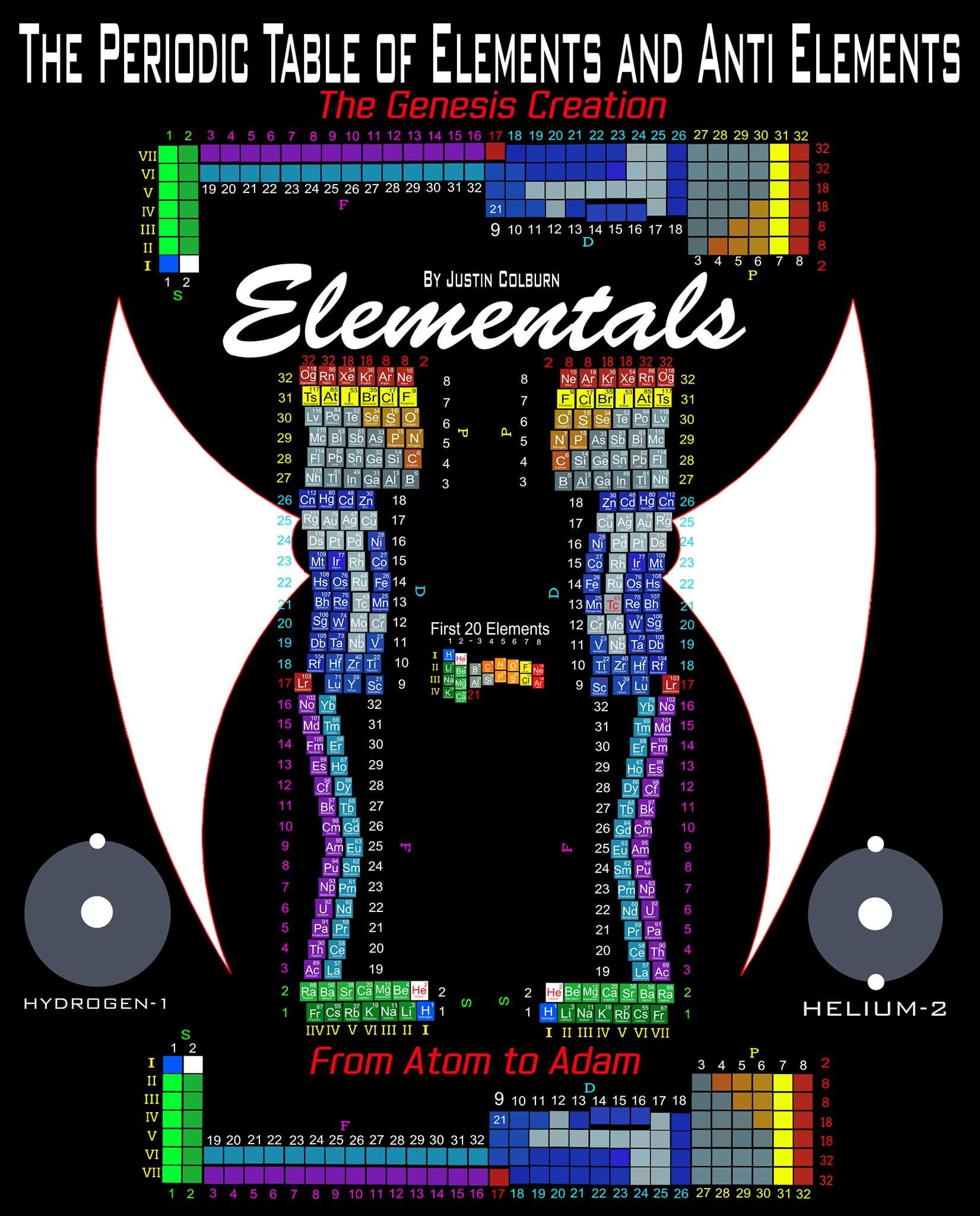
| Year: 2003 | PT id = 228, Type = formulation misc |
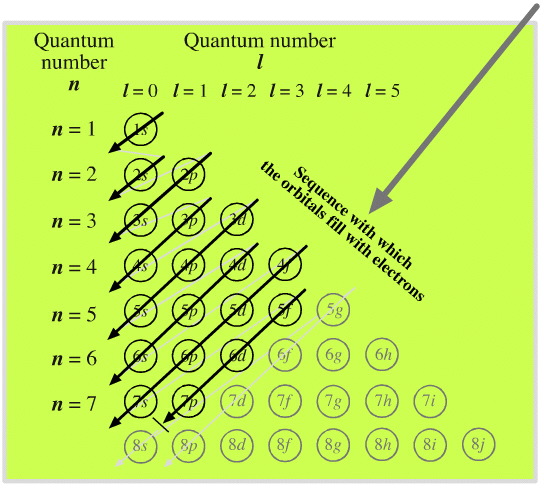
Elements by Orbital
From elsewhere in Mark Leach's Chemogenesis webbook:
Madelung's Rule tells us that the orbitals fill in the order n + l (lowest first). This gives the sequence:
Electronic structure can be illustrated adding electrons to boxes (to represent orbitals). This representation shows the Pauli exclusion principle, the aufbau principle and Hund's rule in action.
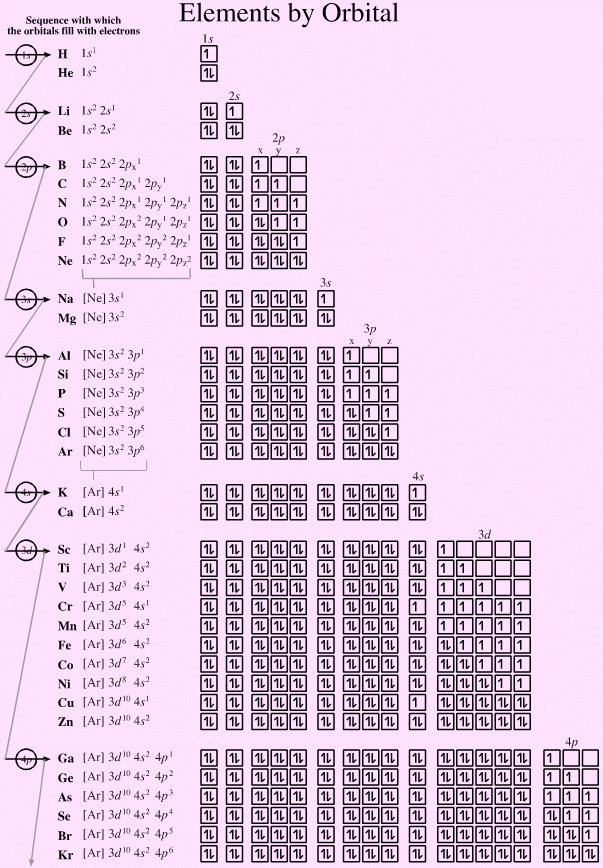
There are some subtle effects with the d block elements chromium, Cr, and copper, Cu. Hund's rule of maximum multiplicity lowers the energy of the 3d orbital below that of the the 4s orbital, due to the stabilisation achieved with a complete and spherically symmetric set of five 3d orbitals containing five or ten electrons. Thus,
- Chromium has the formulation: [Ar] 3d5 4s1 and not: [Ar] 3d4 4s2
- Copper has the formulation: [Ar] 3d10 4s1 and not: [Ar] 3d9 4s2
| Year: 2011 | PT id = 392, Type = data misc |
Elements in Bottles Periodic Table
A nice web site with a physical periodic table of elements:
| Year: 2024 | PT id = 1319, Type = misc |
Elements of Fire & Light
René Vernon presents Elements of Fire and Light: The Majesty of The Periodic Table


| Year: 1967 | PT id = 230, Type = formulation misc non-chem |
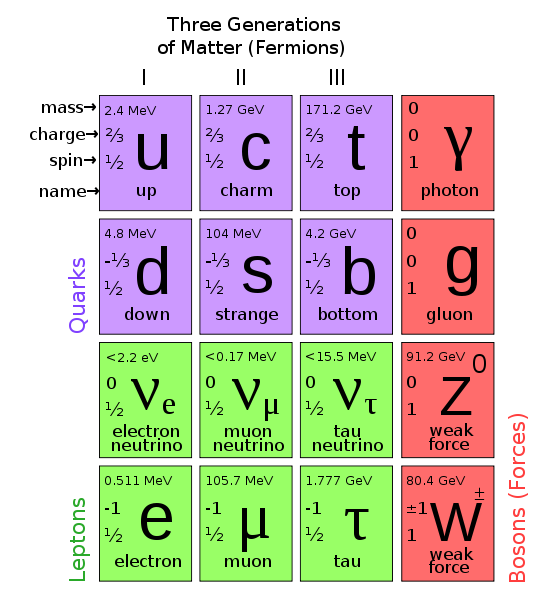
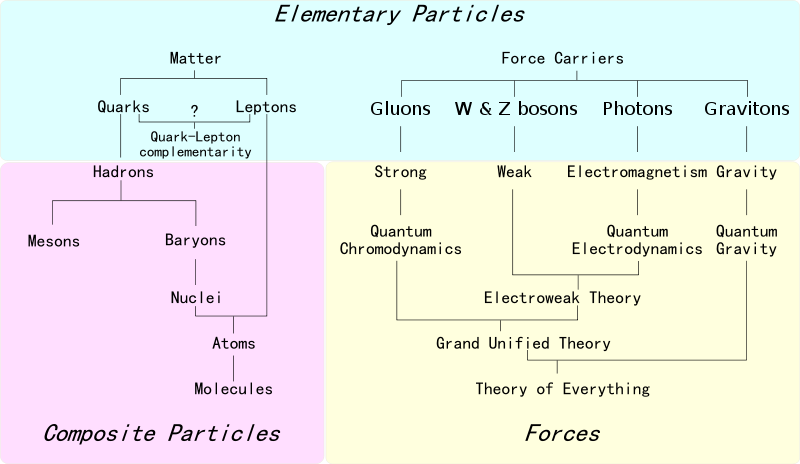
Elements of The Standard Model
The first step towards the Standard Model of particle physics was Glashow's 1960 discovery of a way to combine the electromagnetic and weak interactions. In 1967, Weinberg & Salam incorporated the Higgs mechanism, giving the standard model its modern form of: quarks leptons and bosons.
These diagrams are the periodic tables of elementry particle physics:
| Year: 2019 | PT id = 1090, Type = misc |
Elements Song 2019
From the Chemistry World YouTube channel: The Elements Song 2019
Join Helen Arney, the Waterbeach Brass Band and chemists from around the world in an updated version of Tom Lehrer's Elements Song:
| Year: 1959 | PT id = 215, Type = misc |
Elements Song by Tom Lehrer
The Elements Song by Tom Lehrer recorded live in Copenhagen in 1967:
And, an animated version from 2008 of the 1959 original:
| Year: 2011 | PT id = 428, Type = misc |
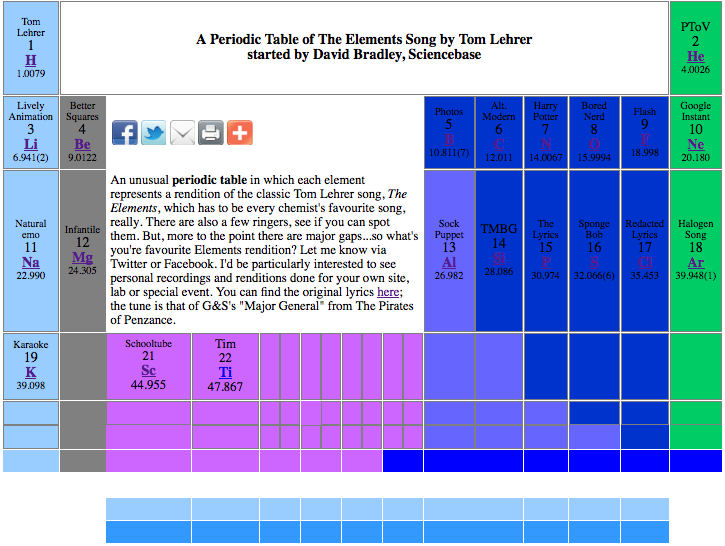
The Elements Song by Tom Lehrer Periodic Table
Started by David Bradley of Sciencebase, a selection of songs about the Periodic Table including the classic Tom Lehrer track.
"An unusual periodic table in which each element represents a rendition of the classic Tom Lehrer song, The Elements, which has to be every chemist's favourite song, really. There are also a few ringers, see if you can spot them. But, more to the point there are major gaps...so what's you're favourite Elements rendition? Let me know via Twitter or Facebook. I'd be particularly interested to see personal recordings and renditions done for your own site, lab or special event. You can find the original lyrics here; the tune is that of G&S's "Major General" from The Pirates of Penzance.":
| Year: 2016 | PT id = 736, Type = misc |
Elements Song Updated by Helen Arney
Tom Lehrer's Elements Song Updated by Helen Arney with all 118 elements [as known to Harvard, as they have been discovered], including the newest ones: nihonium, moscovium, tennessine and oganesson.
| Year: 2003 | PT id = 54, Type = formulation 3D misc |
Elephant Periodic Table
The periodic table does not map to an elephant very well:

Click on the poster below to go to a large version:
| Year: 2009 | PT id = 658, Type = misc |
enkana's Periodic Table
A nice periodic table with a simple graphic for each element by enkana:
| Year: 2012 | PT id = 484, Type = review misc |
Eric Scerri.com
ericscerri.com is the personal internet domain and website of Eric Scerri: chemist and leading philosopher of science specializing in the history and philosophy of the periodic table. He is founder and editor-in-chief of the international journal Foundations of Chemistry, which publishes academic papers concerned with the PT, and is the author of the respected book: The Periodic Table and Its Significance (Oxford University Press, 2007).
The website has links to all of Eric's extensive publications, including online video lectures and interviews and external links.
| Year: 2007 | PT id = 937, Type = data misc |
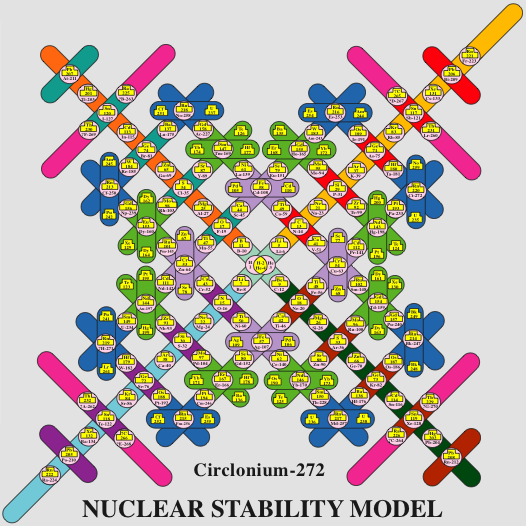
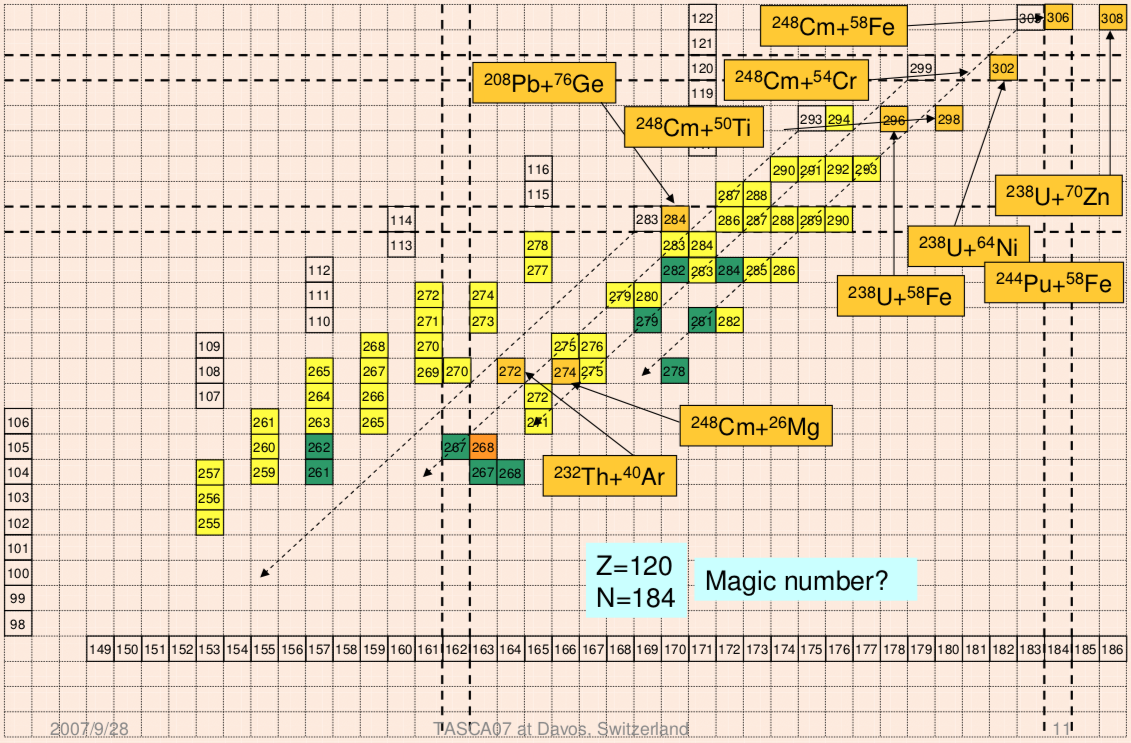
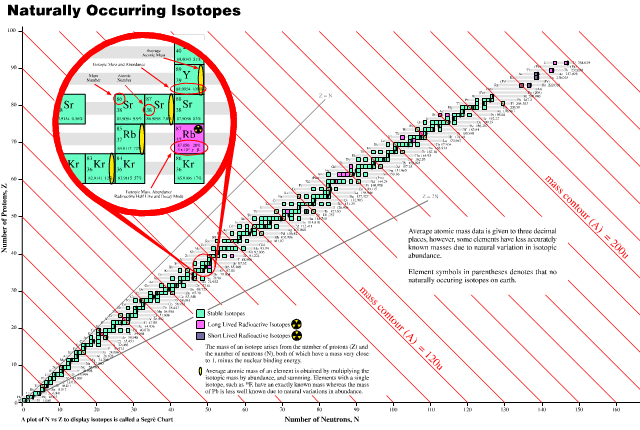
Extending the Periodic Table
The periodic table now extends to element 118, Oganesson, and scientists are attempting to go further. Below is part of a Segre chart, proton number on the y-axis and neutron number of the x-axis, from a report from the Japanese Superheavy Element Laboratory, RIKEN Nishina Center, RIKEN.
The diagram shows various nuclear reactions, for example: 232Th + 40Ar to make 272Hs.
Thanks to Larry Tsimmerman for the tip!
| Year: 2008 | PT id = 659, Type = misc |
f--l--A--r--k's Fractal Periodic Table
A fractal periodic table by f--l--A--r--k:
After nearly a year of work and research, the Periodic Table is complete.
I have endeavored to the best of my ability to accurately represent each element as a fractal. The table itself is up to date with current findings and research as of 2008.
Each element has been individually rendered at a resolution of 3200 x 2400, and is available for a full-view in my gallery. Every fractal was designed, composed, and rendered using Apophysis and then the final assembly done with Photoshop.
Many thanks go to Tony (~atd85) for his assistance in rendering quite a few of these elements, and to my wife for her inspiration and encouragement:
| Year: 1996 | PT id = 238, Type = formulation misc |
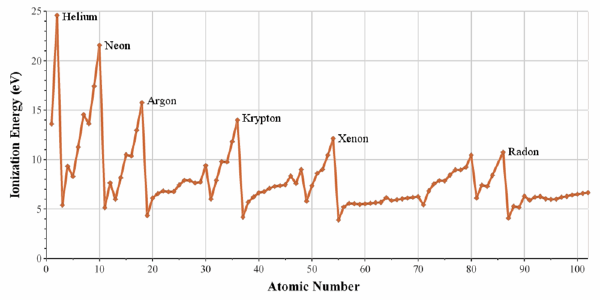
First Ionisation Energy of The Elements
Periodic trend for ionization energy, for example Mg → Mg+ + e–
Each period begins at a minimum for the alkali metals, and ends at a maximum for the noble gases. From Wikipedia:
Based on data from: Martin, W. C.; Wiese, W. L. (1996). Atomic, Molecular, & Optical Physics Handbook. American Institute of Physics. ISBN 156396242X.
| Year: 2020 | PT id = 1104, Type = misc non-chem |
FReNeTic
FReNeTiC is the multi-Award winning 'Frenzied word game of the Elements' where players race against the clock to form as many words as possible using the Element Symbols of The Periodic Table.
In this fast and furious word game players score points equivalent to the atomic numbers of each tile used to create the word, for example Ba Na Na = Banana = 78 points.
The first player to score 1000 points wins!
Everyone plays all the time, quick set up and easy-to-follow rules with FRaNTiC FUN AcTiON! (And no, you don't need to know the Periodic Table or be a GeNiUS to play).

Thanks to Marcus for the tip!
| Year: 2019 | PT id = 1040, Type = formulation 3D misc |
Frog Periodic Table
One of the frogs from Stockport's (UK) Giant Leap Frog Art Trail. This frog is Chemit.

Thanks to Helen P for the tip!
| Year: 2001 | PT id = 555, Type = misc |
Funny Periodic Table
By Eric J Stone a Funny Periodic Table of chemical reactivity.
"This periodic table is unique -- it is informational, educational, and humorous at the same time. Arranged in the standard Mendeleev layout, this table depicts the elements interacting with each other in many interesting ways. The jokes are designed to impart useful information within the context of humor. Ideal for science buffs of all ages -- this is truly the periodic table for the masses. It can be appreciated by children and professionals alike. Children especially like the table, which draws them in with its funny vignettes. This poster is based on the original art of Slavomir Koys. The poster makes a great promotional item. Use it to promote your schools chemistry club or as science fair prizes":
| Year: 2019 | PT id = 1127, Type = data misc |
Geological Periodic Table
Alvarez & Cordoba's Periodic Table of the Elements Associated with Geology [from Spanish using Google Translate]
"It is a simple and innovative table where each element has the shape of its respective crystalline system. It also has several novelties linked to earth sciences such as: illustrative images that show where the element can be found naturally on our planet, geochemical classification and different types of relevant characterizations (radioactivity, synthetics, alloys, majority elements in bark and mantle). Likewise, various useful tools were included in the area such as the well-known Bowen series, categorizations of compatible and incompatible elements, typical cases of the Piper diagram and Stiff diagrams.
"To increase the interaction and understanding between the user and the table, it has elements external to it (letters) that incorporate augmented reality, which allows learning in a simpler, didactic and entertaining way about the atomic structure of chemical elements in 3D. Just scan the back of the letter with your cell phone to see its structure."
Click the image to see the PDF file

| Year: 1971 | PT id = 1269, Type = formulation data misc |
Goldanskii's Chess Board Version of The Madelung Rule (For Orbital Filling)
Ref: Goldanskii, V I: The Periodic System of D I Mendeleev and Problems of Nuclear Chemistry pp 137-162 ex: Verde M (ed.): 1st International Conference on the Periodic Table, Vincenzo Bona, Torino 1971.
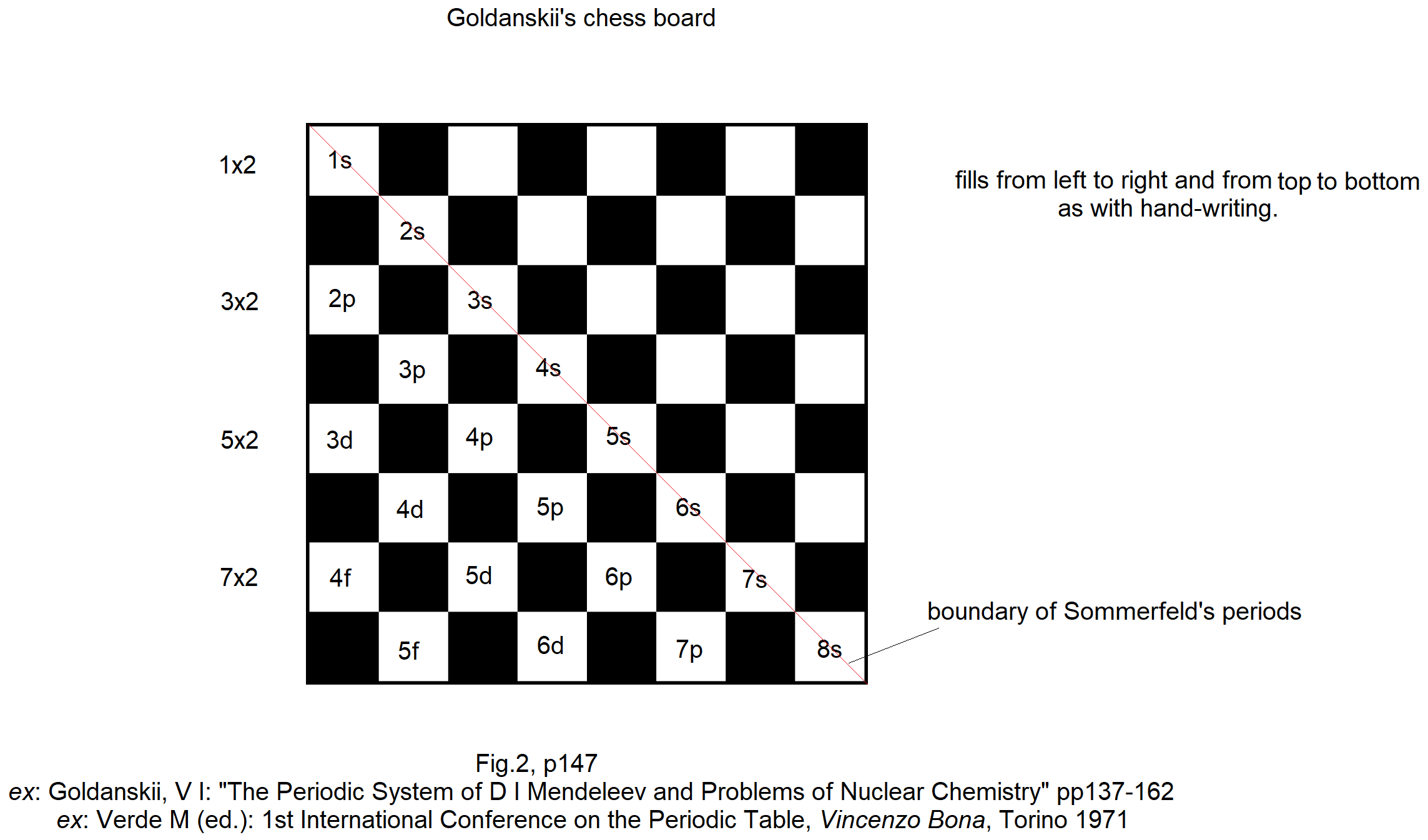
Thanks to John Marks for the tip!
| Year: 2008 | PT id = 122, Type = misc |
Google Image Search Periodic Table
Davebug has made a periodic table using the top Google Image search result for each element. Cool and very www:
| Year: 2022 | PT id = 1253, Type = formulation misc review |
Gorodkov's Periodic System of Periodic Systems
Mikhail Gorodkov writes:
"I hope you'll find attached version of Periodic System not only funny but also informative!"
| Year: 2009 | PT id = 253, Type = formulation misc spiral 3D review |
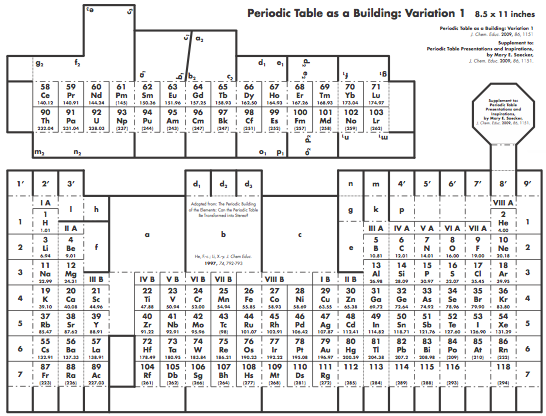
Graphic Representations of the Periodic System
Mary E. Saecker writes an article in Chemical Education Digital Library, Periodic Table Presentations and Inspirations: Graphic Representations of the Periodic System, that reviews some periodic table formunations.
The paper contains a link to this pdf file which gives templates and instructions for several print, cut-out & build periodic table formulations:
Supplement to: Periodic Table Presentations and Inspirations by Mary E. Saecker, J. Chem. Educ., 2009, 86, 1151.
Construction Directions A Cut-Out Chart of the Periodic System (Periodic Table Cylinder)
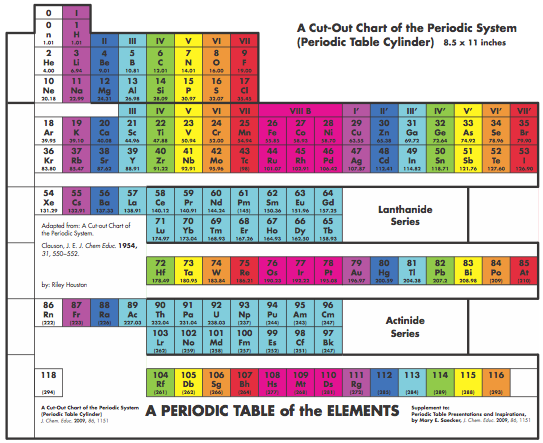


| Year: 1998 | PT id = 116, Type = misc data |
Gray's Wooden Periodic Table Table
Theodore Gray's Wooden Periodic Table Table – a wooden table that incorporates a periodic table – is a treasure trove, both on the web and in reality (his office).
The web site contains over 12 gig of data and beautiful images. Explore!
Theo's new site is periodictable.com.
| Year: 2019 | PT id = 1172, Type = data misc |
Green & Sustainable Chemistry, Periodic Table of
The periodic table of the elements of green and sustainable chemistry by Paul T. Anastas & Julie B. Zimmerman, Green Chem., 2019,21, 6545-6566, (DOI: 10.1039/c9gc01293a). Also, there is a review article in Chemistry World.
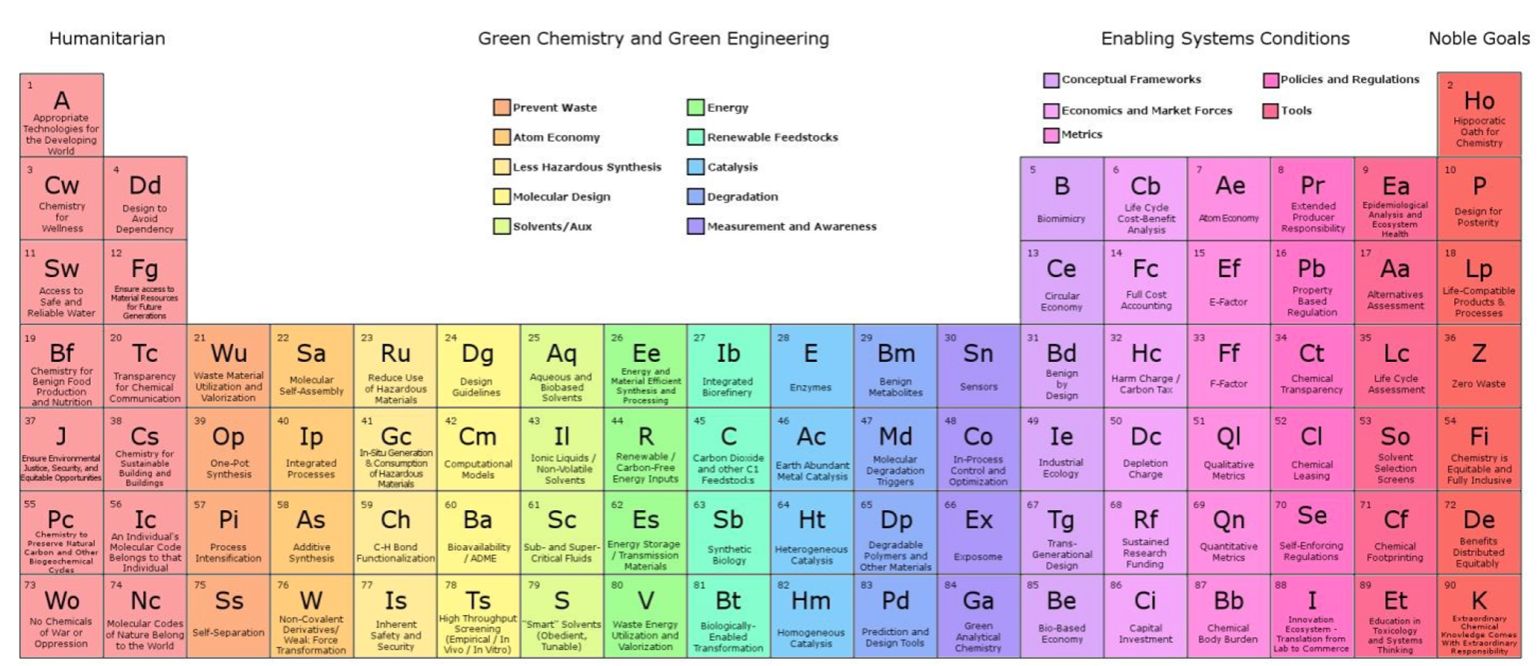
Thanks to Eric Scerri for the tip!
See the website EricScerri.com and Eric's Twitter Feed.
| Year: 2006 | PT id = 134, Type = data misc |
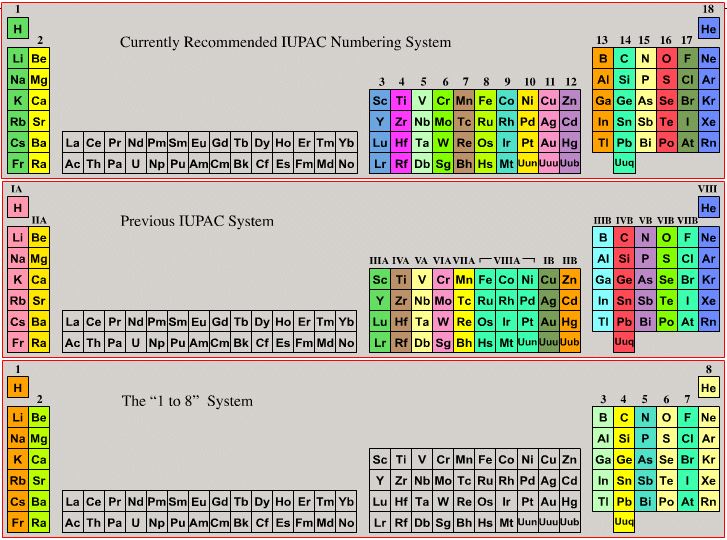
Group Numbering Systems
Phase State: Solid, Liquid, Gas at 20°C & 700°C
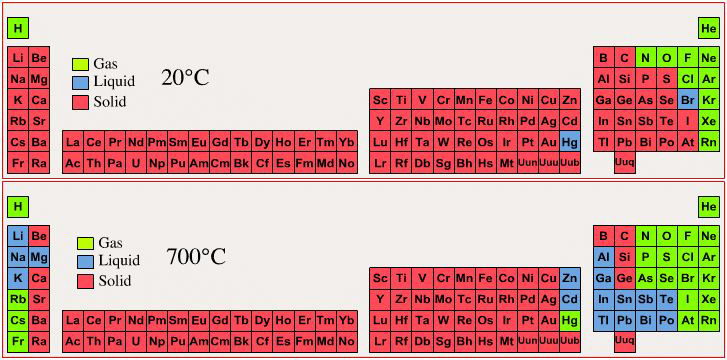
By Mark Leach
| Year: 2001 | PT id = 336, Type = misc non-chem |
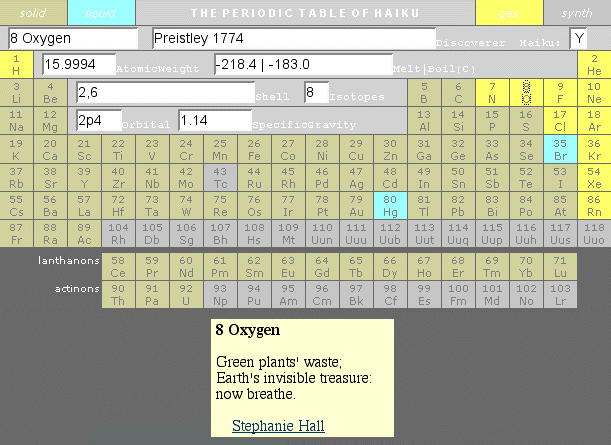
Haiku Periodic Table
The excellent Periodic Table of Haiku has re-emerged from the 'Way Back" web-archive website.
A second 2009/10 Periodic Table of Haiku, from the University of Minnesota is available here.
| Year: 2017 | PT id = 904, Type = misc |
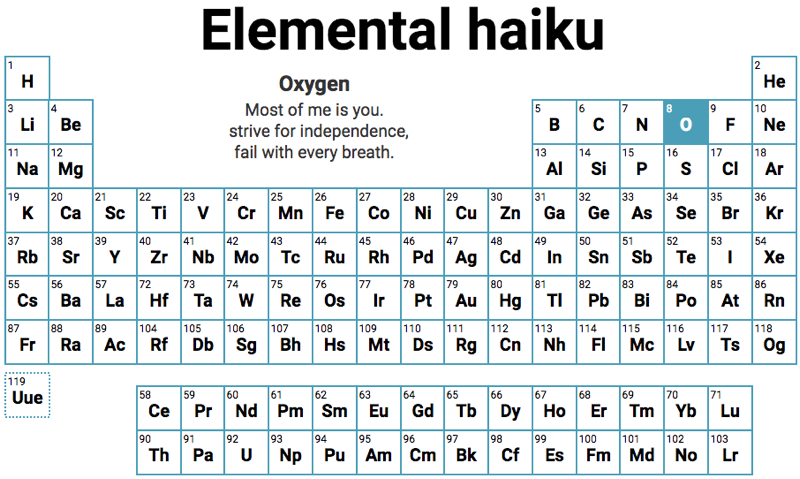
Haiku, Elemental
By Mary Soon Lee, a review of the Periodic Table composed of 119 science haiku, one for each element, plus a closing haiku for element 119 (not yet synthesized). The haiku encompass astronomy, biology, chemistry, history, physics, and a bit of whimsical flair. Click here, then hover over an element on the Periodic Table to read the haiku.
| Year: 2019 | PT id = 1029, Type = data misc |
Heritage Periodic Table Display
By Engineered Labs, the Heritage Periodic Table Display.
"Introducing the world's first and only miniature Periodic Table with the actual elements in it.
"Over the last year, we have successfully collected each and every stable element. After considerable R&D, we have finally developed a method of embedding each element in acrylic and we have to say, the result is awesome!
"The Heritage Periodic Table pretty much speaks for itself. The collection looks great on a desk, in your hands, and anywhere else it can be displayed."

| Year: 2013 | PT id = 580, Type = formulation non-chem misc |
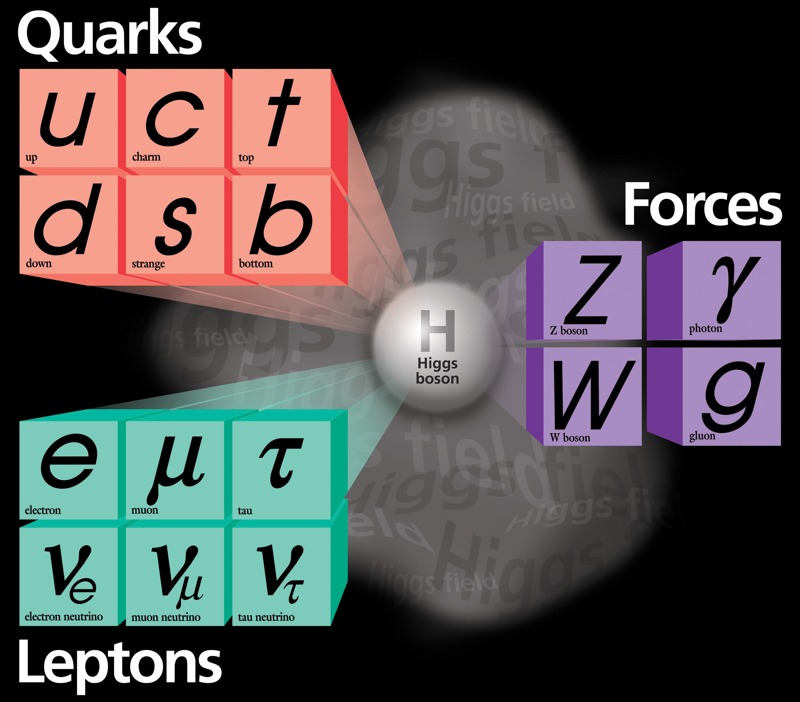
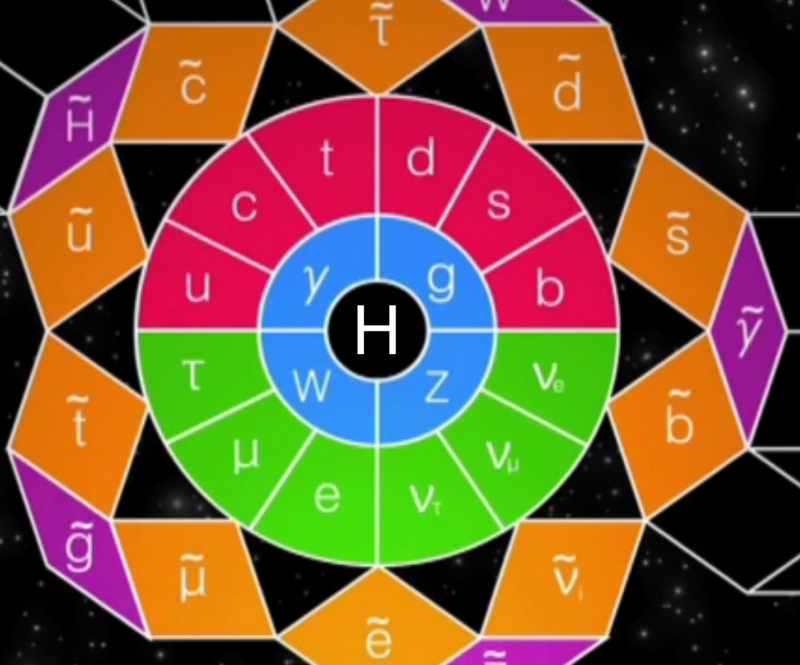
Higgs Boson and Fundamental Particle/Force Periodic Tables
The Higgs boson sits at the heart of the Standard Model of particle physics, and so is at the centre of periodic table type representations of quarks, leptons and forces.
Three representations by the UCR Today, a video interview with Particle Fever editor Walter Murch: "The Higgs boson is kind of a MacGuffin" and from im9.eu:
| Year: 2021 | PT id = 1217, Type = data element misc |
History [of the] Elements and Periodic Table
From the Royal Society of Chemistry (RSC) an interactive Elements and Perioid Table History web page:

Thanks to Eric Scerri for the tip!
See the website EricScerri.com and Eric's Twitter Feed.
| Year: 2019 | PT id = 987, Type = misc |
Homage to The Elements
Eulalia Bosch writes:
"As a curator of the Eugènia Balcells Foundation, I would like to share with you the project to celebrate the 2019 UN decreed International Year of the Periodic Table (IYPT).
"Eugènia Balcells included the mural Homage to The Elements in her exhibition FREQUENCIES at the Santa Monica Art Center in Barcelona in 2009. The exhibit incorporates the spectrum of light that identifies each element. The result is not just another presentation of the periodic table, but a tribute to the set of elements that, in their intertwining, make up the material world and to those spectra that, as Eugènia Balcells like to say are: 'the voice of matter'.
"Over the last few years, the mural Homage to The Elements has also been incorporated at the Pascual Vila Research and Development Center of the CSIC in Barcelona, at the Science Museum, CosmoCaixa, in Barcelona and we are finishing the formalities for its installation in the Universities of Tarragona and Girona. It has also been acquired by the Technische Universität Berlin, and by the Friedrich-Alexander-Universität Erlangen-Nürnberg, both in Germany. In the city of NY, where Eugènia lived for more than thirty years, the mural has found its place at the Maxine Greene High School for Imaginative Inquiry, located at the Martin Luther King Educational Campus in New York, in front of the Lincoln Center, the great ally artistic ally of the School.
"The Eugènia Balcells Foundation wants to actively collaborate in the celebration proposed by the United Nations offering to the educational world the mural Homage to The Elements, this sign that represents universal unity, and records the human knowledge acquired to this day.

| Year: 2011 | PT id = 505, Type = misc |
Homenatge Als Elements
From Eugènia Balcells' blog (and translated using Google Translate):
TRIBUTE TO THE ELEMENTS was born as a counterpoint to the video-installation Eugènia Balcells often, a film without end where the trace elements that each emit light merges with the other and forming a true metaphor for origin of the universe.
Coinciding with the International Year of Chemistry, TRIBUTE TO THE ELEMENTS has been published in two formats: a poster in which each element is represented by its chemical symbol and its own emission spectrum and a version where each element, printed separately, part of a collection that can be stored as such or are available as a mural on a temporary or permanent exhibition space, as presented in the exposure:
Thanks to Eric Scerri for the tip!
See the website EricScerri.com and Eric's Twitter Feed.
| Year: 1993 | PT id = 1268, Type = formulation misc data |
Huheey's Version of The Madelung Rule (For Orbital Filling)
Huheey, J.E., Keiter, E.A., Keiter, R.L.: Inorganic Chemistry: Principles of Structure and Reactivity. 4th edn. HarperCollins College Publishers (1993), p. 22
René Vernon comments: "A peculiar depiction of the Madelung Rule order of filling diagram."
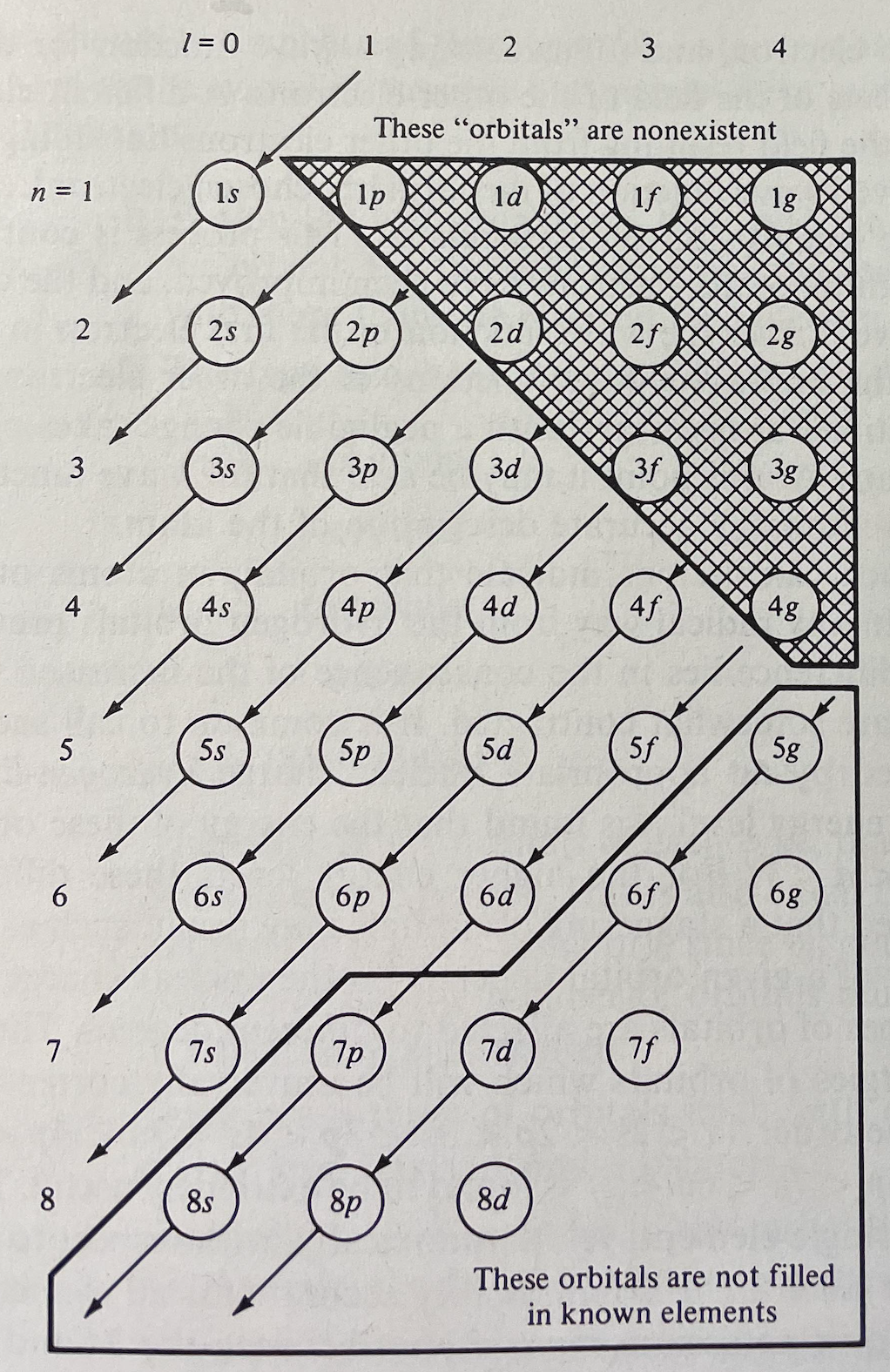
| Year: 2018 | PT id = 942, Type = misc |
I Wear This Shirt Periodically T-Shirt
From Shared.com, a "I Wear This Shirt Periodically" T-Shirt:
Thanks to Clare Cheetham for the tip!
| Year: 2010 | PT id = 382, Type = misc non-chem |
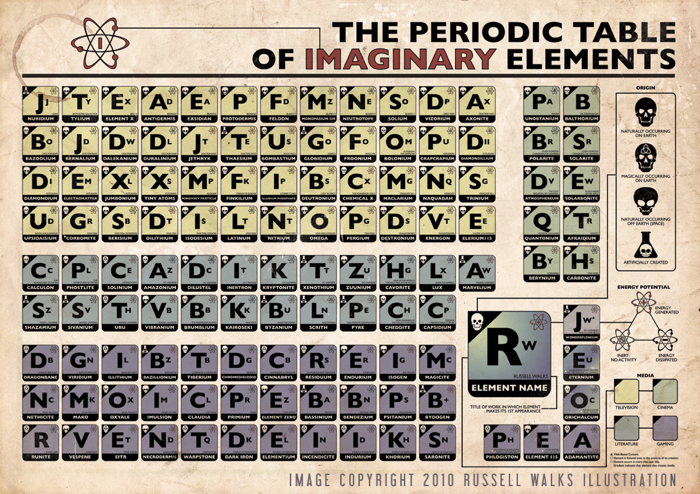
Imaginary Elements
An image of a Periodic Table Imaginary Elements by Russell Walks:
| Year: 2008 | PT id = 338, Type = data misc |
Instruments, Periodic Table of
A periodic table of various scientific instruments and techniques from Thermo Scientific and C&EN.
Download, zoom in & explore the interesting pdf file:
| Year: 2019 | PT id = 1320, Type = formulation misc |
International Year of the Periodic Table – Artwork Competition
From Chemistry A European Journal the results of a Periodic Table artwork competition, where the full stories can be read.
First Place: Víctor Duarte Alaniz from Mexico City with "Cycles in Space, In Time... and in Chemistry"

Second Place: Yuliia Oleksii from Vinnytsia, Ukraine "Noble Gases"

Third Place: Joanna Cwynar-Wojtonis from Poland

| Year: 2019 | PT id = 983, Type = misc review |
International Year of the Periodic Table (in Paris and Moscow)
Prof. Martyn Poliakoff of The University on Nottingham and Periodic Videos at the opening of the International Year of the Periodic Table:
| Year: 2019 | PT id = 1066, Type = formulation misc |
International Year of the Periodic Table with Eric Scerri
A YouTube video about IUPAC's International Year of the Periodic Table:
Thanks to Eric Scerri – who appears – for the tip!
See the website EricScerri.com and Eric's Twitter Feed.
| Year: 1966 | PT id = 248, Type = formulation misc |
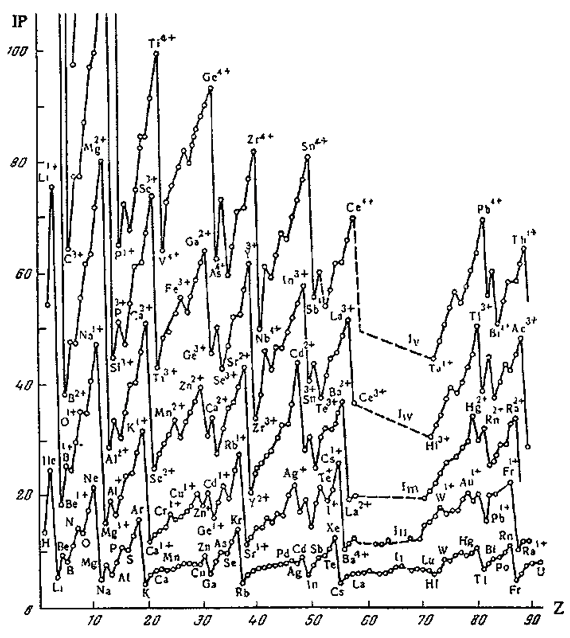
Periodic Table of Ions
From Concept of Chemical Periodicity: from Mendeleev Table to Molecular Hyper-Periodicity Patterns E. V. Babaev and Ray Hefferlin, here.
"One intriguing problem that arises from with the periodic table of atoms is the possibility of constructing periodic systems of ions, V. K. Grigorovich, Periodic Law of Mendeleev and Electronic Structure of Metals, Nauka Publ.: Moscow, 1966 (in Russian). An atom can be completely or partially ionized to a cation by removing electrons or transformed into an anion by the addition of new electrons. The energy required for a few consecutive ionisations of atoms is plotted against the atomic number. One can see that the curves are periodic, and hence it is possible to construct periodic tables for mono-, di-, and multi- charged cations. If we look at the dispositions of the maxima and minima of the curves and compare them with those for atoms, it becomes evident that the magic numbers of electrons for ions are the same as for neutral atoms. Therefore, the number of electrons (but not the charge of the nucleus) is responsible for the periodicity of ions."
| Year: 2012 | PT id = 559, Type = data misc |
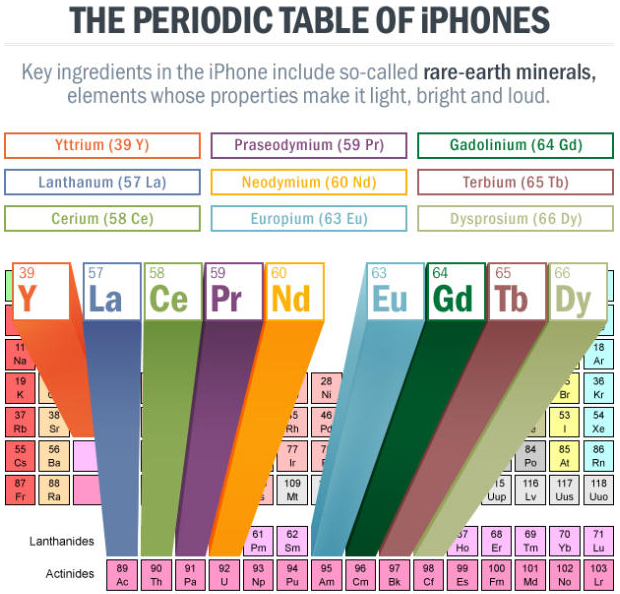
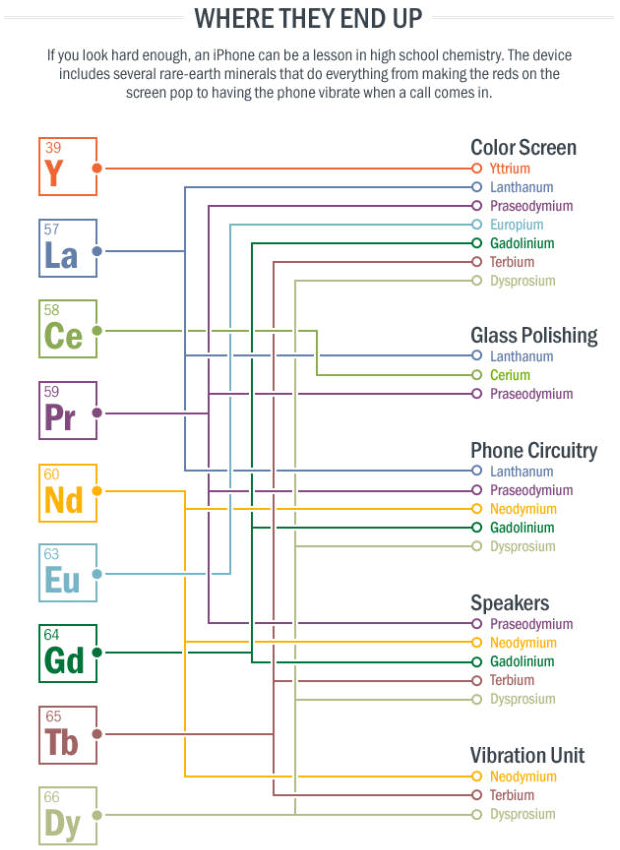
iPhone, Periodic Table of
An article in Scientific American Digging for Rare Earths: The Mines Where iPhones Are Born.
"About 60 miles southwest of Las Vegas, in a mine some 500 feet deep, the beginnings of an iPhone come to life. But the sleek, shiny iPhone is far, far removed from the rocks pulled out of this giant hole, which looks like a deep crater on the moon. Inside the rocks from this mine are rare-earth minerals, crucial ingredients for iPhones, as well as wind turbines, hybrid cars, and night-vision goggles. Minerals such as neodymium are used in magnets that make speakers vibrate to create sound. Europium is a phosphor that creates a bright red on an iPhone screen. Cerium gets put into a solvent that workers use to polish devices as they move along the assembly line, etc.":
Thanks to Eric Scerri for the tip!
See the website EricScerri.com and Eric's Twitter Feed.
| Year: 2012 | PT id = 1163, Type = misc |
Itch: A Book & TV Drama About a Boy Who Collects The Chemical Elements
Meet Itch – an accidental, accident-prone hero. Science is his weapon. Elements are his gadgets.
All-action adventure perfect for fans of Alex Rider and Young Bond.
Itchingham Lofte – known as Itch – is fourteen, and loves science, especially chemistry. He's also an element-hunter: he's collecting all the elements in the periodic table. Which has some interesting and rather destructive results in his bedroom.
Then, Itch makes a discovery. A new element, never seen before. At first no one believes him – but soon someone hears about the strange new rock and wants it for himself. And Itch and his family are catapulted into a breathless adventure with terrifyingly high stakes...
The debut novel from BBC radio presenter Simon Mayo.
Children's book Itch by Simon Mayo is available from Amazon and all good bookshops, ISBN: 9780552565509. The book has been made into a TV Series, filmed in Western Australia, and is available on the BBC iPlayer.

| Year: 2019 | PT id = 1194, Type = misc formulation |
IUPAC Periodic Table Challenge: Nobelium Contest
IUPAC Periodic Table Challenge: Nobelium Contest. DOI: https://doi.org/10.1515/ci-2020-0204
After a first round based on the knowledge of the participant of the chemical elements, in this new phase of the Challenge, participants were invited to share their passion and creativity about chemistry. The entries were supposed to highlight the role of the Periodic Table in a creative manner. We received a variety of submissions including videos, poems, songs, and paintings. Entries were categorized into science, art, and outreach or community activities related to the science education and the Periodic Table. We were genuinely surprised and inspired by the dozens of pieces of art that we were receiving from all around the world. A jury as well as popular votes casted for each entry decided who were to receive the special limited edition IUPAC Periodic Tables signed by the Nobel laureates. That was one of the most difficult tasks of this activity, as the quality, originality, and diversity of the entries were truly amazing.
The artist is Swaprabha Chattopadhyay, a high school student from India.
- https://iupac.org/100/pt-challenge-entry/page/3/
- https://iupac.org/100/pt-challenge-entry/adorned-with-elements/
- https://iupac.org/100/summer-winners-of-the-nobelium-contest-announced/

| Year: 2009 | PT id = 249, Type = formulation misc non-chem spiral 3D |
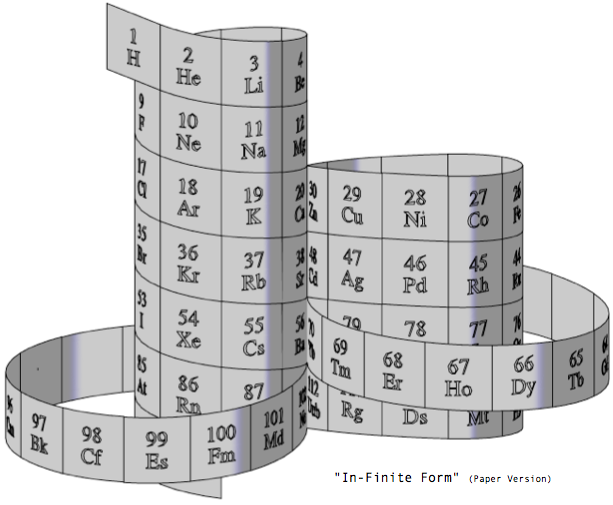
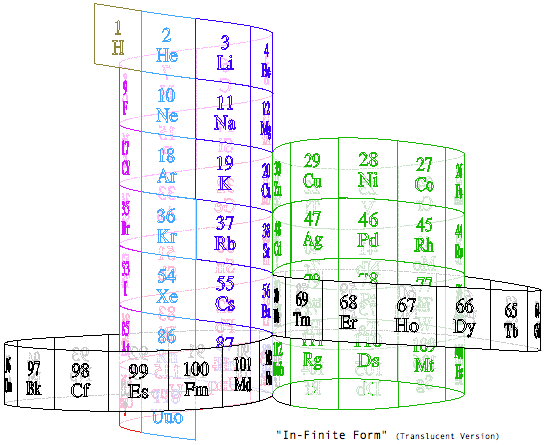
Steve Jensen's "In-Finite Form"
"I'm a figurative sculptor, living in Minneapolis MN. A few years ago, while looking at a two dimensional version of the periodic table, I too wondered if it would be possible to create a Periodic Table without any visual breaks in its numerical sequence. Although I had never seen anything other than the rectangular flat table, I thought I might be able to solve this spatial continuity problem three dimensionally. I also wanted to limit myself to using a 3-D "line" that had no sudden changes in direction. After coming up with what I thought was a new and unique sculptural resolution, I put the project aside. Only recently (after re-building my paper model out of a translucent material) did I do some research on the web, and immediately recognized the strong likeness between my version and the Alexander Arrangement. Even more surprising was my models' visual similarity to Crookes' figure eight design from some 111 years ago.
"Although there are obviously many inventive and well thought out responses to this design challenge, I believe that my solution is a unique one, and an improvement over some of the previous three dimensional forms. The "line" of my model allows for contiguous numerical placement of all the symbols (while maintaining group continuity along its vertical axis), even as the shape of its plan view makes visual reference to the well-known symbol for infinity. What's more, in my version, the Lanthanide & Actinide series do not occupy a separate field but are fully integrated into the continuous linear flow. This piece, which I've entitled "In-Finite Form" speaks to the mystery of the endless flow of space, even as it folds back onto itself within the confines of a finite system."
| Year: 2013 | PT id = 616, Type = misc |
Joke

Thanks to Marcus Lynch for the tip!
| Year: 2001 | PT id = 57, Type = misc |
| Year: 2012 | PT id = 493, Type = formulation data misc |
JR's Chemistry Set
For the iPhone and iPad, JR's Chemistry Set makes chemistry interesting and fun to learn. Based upon the innovative Rota Period, it is a handy and powerful reference tool for chemistry enthusiasts and practitioners at all ages and all levels.
| Year: 2010 | PT id = 626, Type = misc |
Kabbalistic Periodic Table
A Kabbalistic periodic table from www.inner.org that attempts to link the PT with the Torah version of Genesis:
| Year: 2007 | PT id = 192, Type = misc |
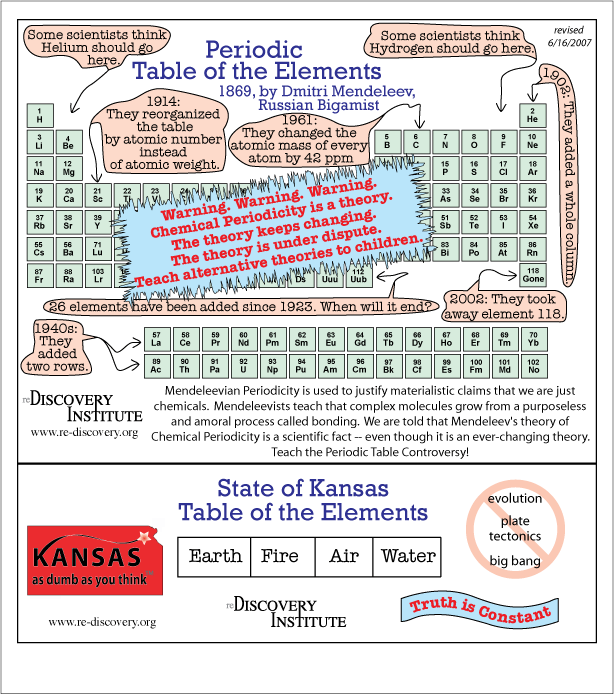
Kansas Periodic Table
The Kansas periodic table... with warnings... by the reDiscovery Institute.
Click the link, then on the top left hand side of the page go to Chemistry, then Just a Theory:
| Year: 2016 | PT id = 730, Type = formulation misc |
KAS Periodic Table
The KAS periodic table reproduces and depicts the nuclear properties of chemical elements. This periodic table depicts not only the trends of nuclear properties, but also reproduces their numerical values that remain very close to the experimental values (difference less than 4%).
The Segre Chart is based on the number of protons, Z, and the number of neutrons, N. It is like a library of nuclei and shows the recorded data only. The Segre Chart can not work when the number of neutrons is not given. But KAS Periodic Table works when the number of neutrons is not given.It does not require the number of neutrons to produce the results.This is a simple chart based on the number of protons of chemical element. We identify the following properties of elements:-
- Location that remains near the Neutron Dripline of element.
- Location that remains very close to stable or long-lived isotopes of the element. Location that remains near the Proton Dripline of element.
- In the case of superheavy elements, we identify which Compound Nuclei are involved in the Hot Fusion reaction and which Compound Nuclei are involved in the Cold Fusion reaction.
- We see the r-process path and assess the r-process abundance.
- The pattern of abundance of chemical elements.
- We identify which elements are the product of exothermal fusion.
- We identify the location of isotope on the basis of two-neutron separation energy.
- Nuclear binding energy trend. Beta decay trend.
- We see the Straight Line of Nuclear Stability.
- Empirical Law discovered.
- Periodicity in the nuclear properties.
- We can compare the nuclear properties of an element with the nuclear properties of almost all the chemical elements.
Read more here, here and here.
| Year: 2010 | PT id = 304, Type = formulation misc |
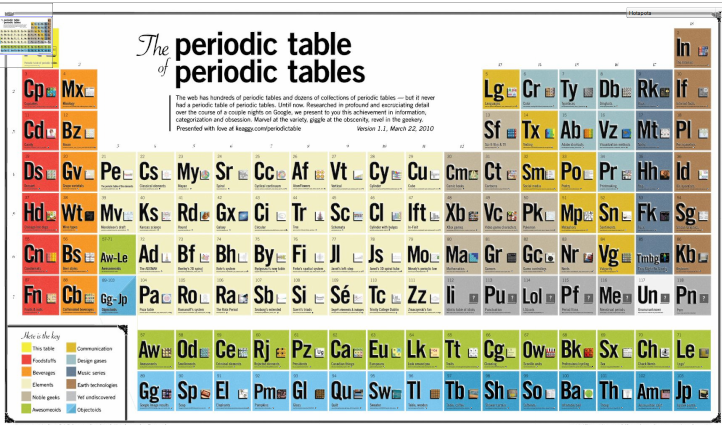
Periodic Table of Periodic Tables
Keaggy, of www.keaggy.com, has put together a rather cool 'Periodic Table of Periodic Tables', clearly using this web site as one of the major resources:
| Year: 2010 | PT id = 388, Type = formulation misc |
Khipu or Quipu Periodic Table
The Khipu or Quipu or Talking Knot Periodic Table, developed by Julio Antonio Gutierrez Samanez.
Google translated from the Spanish pdf file:
"As a result of bringing together each pair of periods in a single function or binod, the author has found a new regular on the subject, which has been defined as a new quantum number, since the number of orders or regulations binod growth elements in the table, under the appearance of pairs of new types of quantum structures or periods whose organization responds to a simple mathematical function: a parable of the type Y = 4 X ^ 2 - In this case report: a) That the strings correspond to pairs of periods or binod and knots are double for items with orbital s (in red), six nodes for p in orange, 10 yellow d knots and 14 knots for green f . b) That in each binod or rope, appear regularly in pairing mode or dual, new quantum or orbital structures, such as moving from within the orbital previous binod.":
| Year: 2019 | PT id = 969, Type = formulation misc |
Kid's Periodic Table
From Cognitive Classroom, a Kid's 'cut-down' Periodic Table:
| Year: 2019 | PT id = 1037, Type = misc |
Knitted Blanket Periodic Table, In Time to Celebrate 150th Anniversary
Trish Bosco thought we might be interested in her periodic table blanket.
"It took me almost 4 years to make it, but I finished in time to celebrate the 150th anniversary! You can see my progression here":
| Year: 2021 | PT id = 1215, Type = misc |
Largest Periodic Table in Eurasia Created in Dubna
From The Times of India:
"The largest [PT] in Eurasia, the Periodic Table of Mendeleev opened in Dubna near Moscow. The event is timed to coincide with the 65th anniversary of the Joint Institute for Nuclear Research located here and the city itself. It is noteworthy that it is at JINR, in the Laboratory of Nuclear Reactions. G N Flyorov under the guidance of Academician of the Russian Academy of Sciences Yuri Oganesyan, all known to date superheavy elements were obtained – from 113th to 118th (the latter is even named after the scientist – 'Oganeson Og'). Oganesyan is the second scientist in the world, after whom a new element of the Periodic Table was named during his lifetime (the first was the American scientist Glenn Theodore Seaborg)."

Thanks to Eric Scerri for the tip!
See the website EricScerri.com and Eric's Twitter Feed.
| Year: 2025 | PT id = 1331, Type = formulation misc spiral 3D |
LEGO Periodic Tower of The Elements
A LEGO Periodic Tower of The Elements, a form of Schaltenbrand's 1920 Helical Periodic Table, by Enrique Barrajón, as shown on the LEGO Ideas website.
Enrique writes:
"The Periodic Tower of Elements is a complex LEGO project designed for students with some chemistry knowledge. It serves as an educational tool to help with learning basic chemistry and physics concepts. Building this model enhances skills such as patience, balance, attention to detail, and innovation—qualities essential for mastering abstract scientific ideas. Additionally, the model offers insights into architectural stability concepts like spiral staircases."

| Year: 2018 | PT id = 918, Type = misc |
Lego® Periodic Table
Welcome to the Lego® Periodic Table of Elements.
Students and faculty at Spring Arbor University (MI), students and faculty at Hardin Valley Academy (TN), and members of the community surrounding Spring Arbor have worked together to construct a periodic table entirely out of Lego® blocks.
On each elemental square has been placed a small Lego® creation that somehow represents the element on which it is positioned.
You can learn a little bit about each element by clicking on its particular square on the table below.
| Year: 1934 | PT id = 296, Type = formulation misc |
Leningrad Monument To The Periodic Table
Leningrad monument to the periodic table, located near to the main chamber of weights and measures, 1934 (from van Spronsen):

From Wikipedia:
| Year: 2014 | PT id = 651, Type = misc |
Letters & Words Periodic Table
By Claude Ziad Bayeh:

| Year: 2009 | PT id = 513, Type = misc |
KU Leuven Periodic Table
On the ground floor of the Universiteitshal (University Hall) of KU Leuven in Belgium is a physical periodic table.
Each element can be explored from this page:
| Year: 2010 | PT id = 335, Type = data misc |
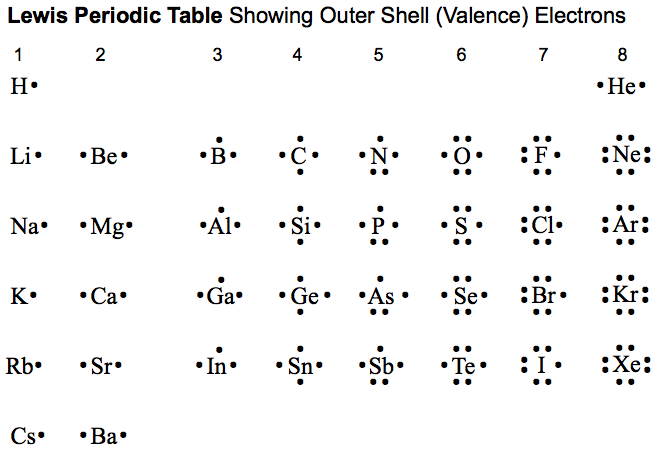
Lewis Octet Periodic Table
A periodic table showing the outer shell of valence electrons associated with Lewis atoms:
By Mark Leach
| Year: 2019 | PT id = 962, Type = formulation review misc |
Möbius-Escher Periodic Table
A comment article in Nature by Prof. Eric Scerri about quantum mechanics and the periodic table:
"Can quantum ideas explain chemistry's greatest icon? Simplistic assumptions about the periodic table lead us astray.
"Such has been the scientific and cultural impact of Dmitri Mendeleev's periodic table of the elements that many people assume it is essentially complete. [But] in its 150th year, can researchers simply raise a toast to the table's many dividends, and occasionally incorporate another heavy synthetic element?
"No – this invaluable compilation is still not settled. The placements of certain elements, even hydrogen and helium, are debated."
The article is accompanied by a fantastic illustration by Señor Salme with ideas from the Möbius strip and M.C. Escher:
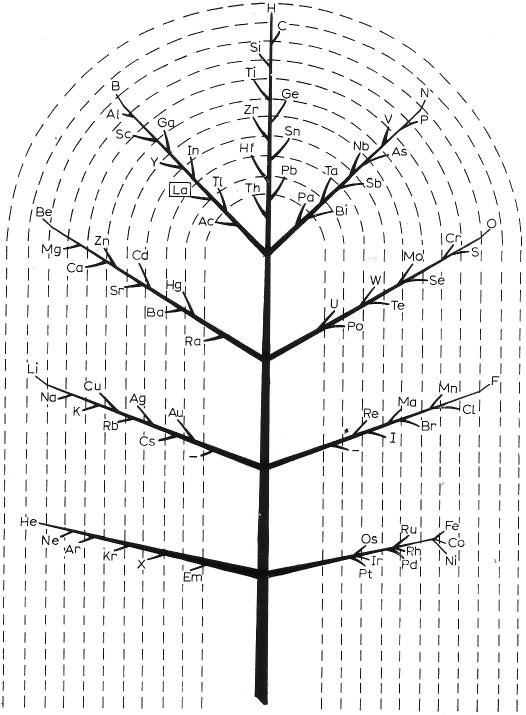
| Year: 1944 | PT id = 293, Type = formulation misc |
Müller's Tree System
In 1944 Müller produced a formulation based on Darwin's tree of life (from van Spronsen):
| Year: 2012 | PT id = 500, Type = formulation misc |
Magnetic Periodic Table
By Particle Zoo, sellers of Higgs Boson and Anticharm Quark soft toys, comes a magnetic periodic table which you can arrange into any formulation you like!
| Year: 2021 | PT id = 1200, Type = non-chem misc data |
Map of Fundemental Particles
By Domain of Science – Posters & YouTube Channel – a periodic table of the fundamental particles that make up the periodic table.
Domain of Science is produced by physicist Dominic Walliman who is on a quest to make science as easy to understand as possible.

| Year: 2012 | PT id = 479, Type = misc formulation |
Mathematical Expression of Mendeleev's Periodic Law
Valery Tsimmerman, of the ADOMAH Tetrahedron periodic table formulation and the Perfect Periodic Table website, presents a Mathematical Expression of Mendeleev's Periodic Law:

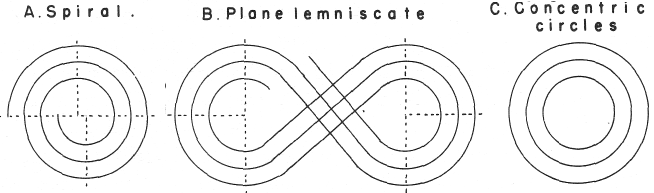
| Year: 1974 | PT id = 299, Type = formulation spiral 3D misc |
Mazurs' PT Formulation Analysis
In his 1974 book Edward G. Mazurs (2nd edition) Graphic Representations of the Periodic System During One Hundred Years, University of Alabama Press gives a comprehensive analysis of periodic table formulations.
Mazurs identifies most PT formulations as being:
- Spiral
- Plane lemniscate
- Concentric circles
- Helix on a cylinder
- Helix on a cone
- Space lemniscate
- Space concentric circles
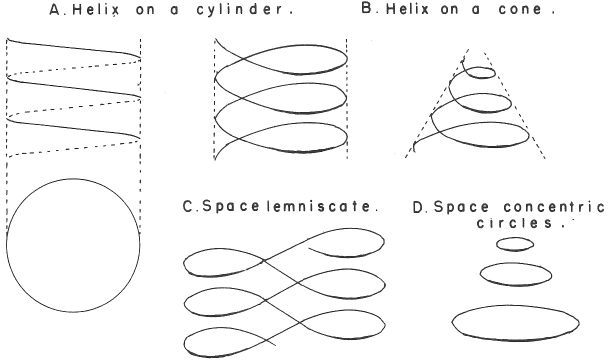
| Year: 2009 | PT id = 237, Type = misc non-chem |
Meet the Elements
"Meet the Elements", is a song & video from They Might be Giants, on bOING bOING:
| Year: 1997 | PT id = 437, Type = misc |
Memory Pegs Periodic Table
On John Pratt's website there is a periodic table of Memory Pegs.
"Each picture in this periodic table is designed to remind you of the element's name, atomic number, and abbreviation. Point to the element to see its name and number. Click on the element for more explanation and then Back to return to the table. There is also an explanation of how to use the pictures as memory pegs. You can also see the name and number of the element by pointing to it and reading the address in the status window at the bottom. At least memorize the first twenty! Each of those first 20 also has a unique color which can also be used for memorizing a list of twenty objects by associating a color with each.":
| Year: 2019 | PT id = 1065, Type = formulation review misc |
Mendeleev 150
Mendeleev 150 is the 4th International Conference on the Periodic Table. The event welcomed nearly 300 guests from over 30 countries and has become one of the key events of IUPAC's International Year of the Periodic Table.
Thanks to Eric Scerri – who appears – for the tip!
See the website EricScerri.com and Eric's Twitter Feed.
| Year: 1891 | PT id = 11, Type = misc |
Mendeleev's Properties of The Chemical Elements
Scanned from the first English edition of Dmitrii Mendeleev's Principles of Chemistry (translated from the Russian fifth edition) a table showing the periodicity of the properties of many chemical elements, taken from the Wikipedia from where a 2116 x 2556 version is available, or here.
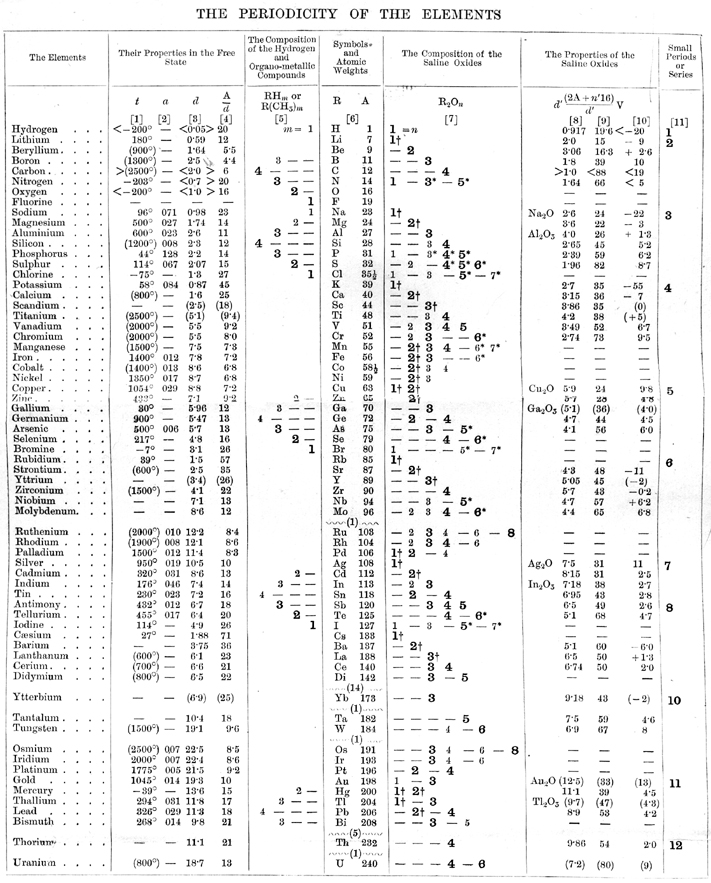
| Year: 1996 | PT id = 991, Type = data misc |
Metals in Medicine Periodic Table
From Metal Complexes in Aqueous Solutions by Martell & Hancock, a periodic table of metals in medicine.
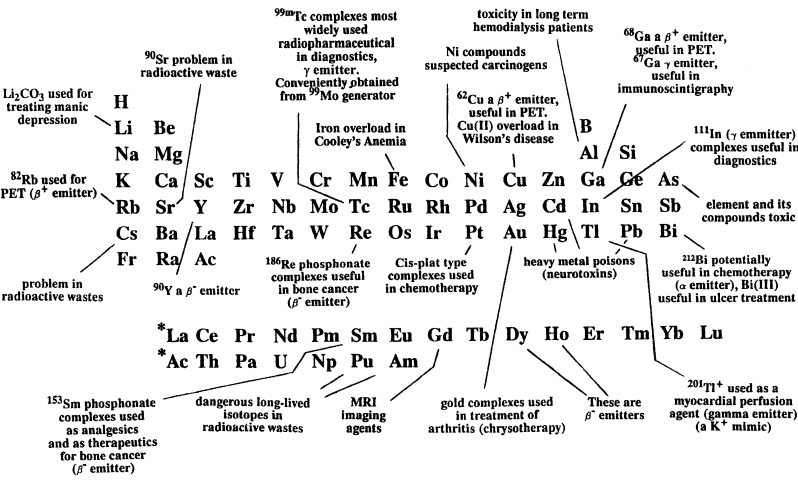
| Year: 2019 | PT id = 1057, Type = misc review formulation |
Meyer's NYT Graphic
A nice graphic by Alex Eben Meyer in the New York Times accompanying an article about the periodic table and some of Sir Martyn Poliakoff ideas.
Thanks to Eric Scerri for the tip!
See the website EricScerri.com and Eric's Twitter Feed.
| Year: 2000 | PT id = 757, Type = misc |
MIT Periodic Table Characters
Eric Scerri writes:
"This apparently hangs on a wall of Building 6 at MIT. I have identified the people around the old-school periodic table, they are (from left to right): Zosimos, Ko Hung, Jabir, Boyle, Lomonosov, Lavoisier, Berzelius, Wohler, Cannizzaro, Berthelot & Mendeleev":

Thanks to Eric Scerri for the tip!
See the website EricScerri.com and Eric's Twitter Feed
| Year: 2012 | PT id = 499, Type = misc |
Mnemonic Periodic Table Song
By Ballroom Jam, a mnemonic song to help memorise the chemical elements:
Thanks to Eric Scerri for the tip!
See the website EricScerri.com and Eric's Twitter Feed.
| Year: 2012 | PT id = 495, Type = misc |
Mug Periodic Table
From www.msmugs.com, a coffee mug with the periodic table of the elements with the elements Lu and Lr correctly positioned... and a gift from Chris H.:


| Year: 2018 | PT id = 932, Type = data misc |
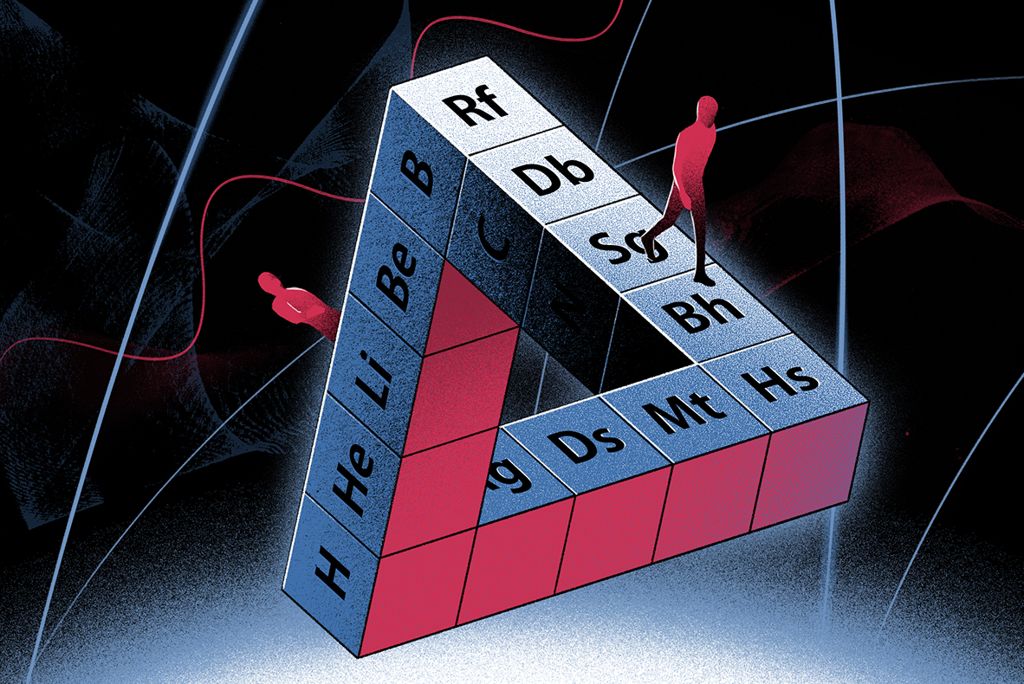
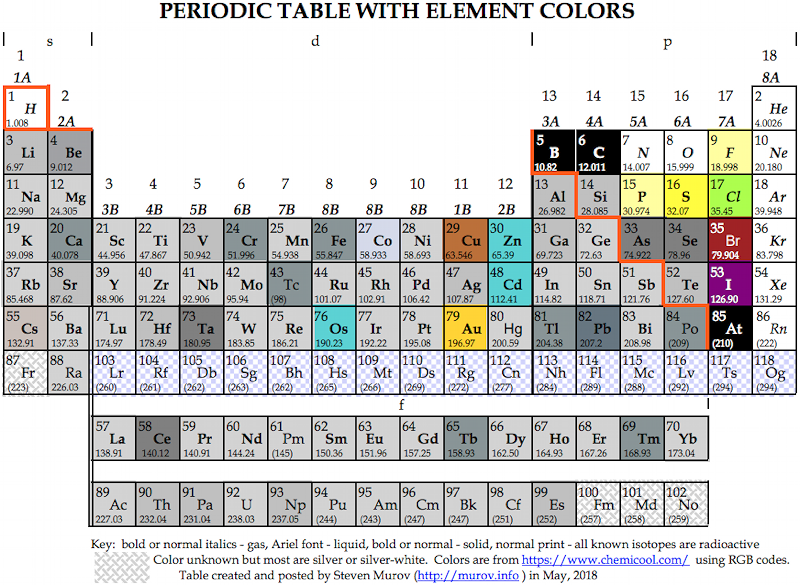
Murov's Colours of the Elements
Steven Murov writes :
"The element squares of this periodic table have colors resembling the actual colors of the elements. The table provides insight useful for helping to distinguish metals and non-metals as well as observations on elements of unusual color. The colors were taken from https://www.chemicool.com/ and applied with RGB codes."
The tables are available online at:
| Year: 2013 | PT id = 614, Type = formulation misc |
Music Notes of Periodic Table
By Claude Bayeh, a Musical Notes formulation:

| Year: 2006 | PT id = 1209, Type = misc |
Nandor's Exhaustive Lists of Chemical Words
From a really interesting PT website, Mark Nandor's Exhaustive Chemical Words:
So what, exactly, is a "chemical word"? It is an English word that can be spelt using element symbols as 'letters', [famously] for example: Beer (or BeEr):
Arches can be "spelt" in two ways:
ArCHeS [Argon, Carbon, Helium, Sulfur]
ArCHEs [Argon, Carbon, Hydrogen, Einsteinium]
On the other hand, there is no way at all to "spell" a work like pillar.
Mark Nandor provides several exhaustive lists:
- All 44,313 English words that can be spelled with element symbols.
- The various ways these can be spelt with element symbols (93,604 ways).
- 264 First Names
- etc.
| Year: 2018 | PT id = 949, Type = formulation data misc |
Nawa's V.E.T. Periodic Table & Hourglass
Nagayasu Nawa, the prolific designer of periodic tables, here and here, has come up with an orbital filling periodic table and a corresponding hourglass animation. Nawa writes:
"I have turned the v.e.c. PT into the GIF animation that I call the electron hourglass, 1 second for each element. It takes 120 seconds from 1H to 120 Ubn. I have coloured orbital with colour derived from each shell's name, such as:
- K kiwi
- L lapis lazuli
- M mauve
- N navy
- O orange
- P purple
- Q quick silver"
Click image to enlarge.
| Year: 2021 | PT id = 1196, Type = data misc |
Nawa's Rainbow Periodic Table
Nagayasu Nawa - "A Japanese school teacher and periodic table designer" - has developed a Rainbow Periodic Table that is stuffed full of data.
Click here to download the .pdf file.
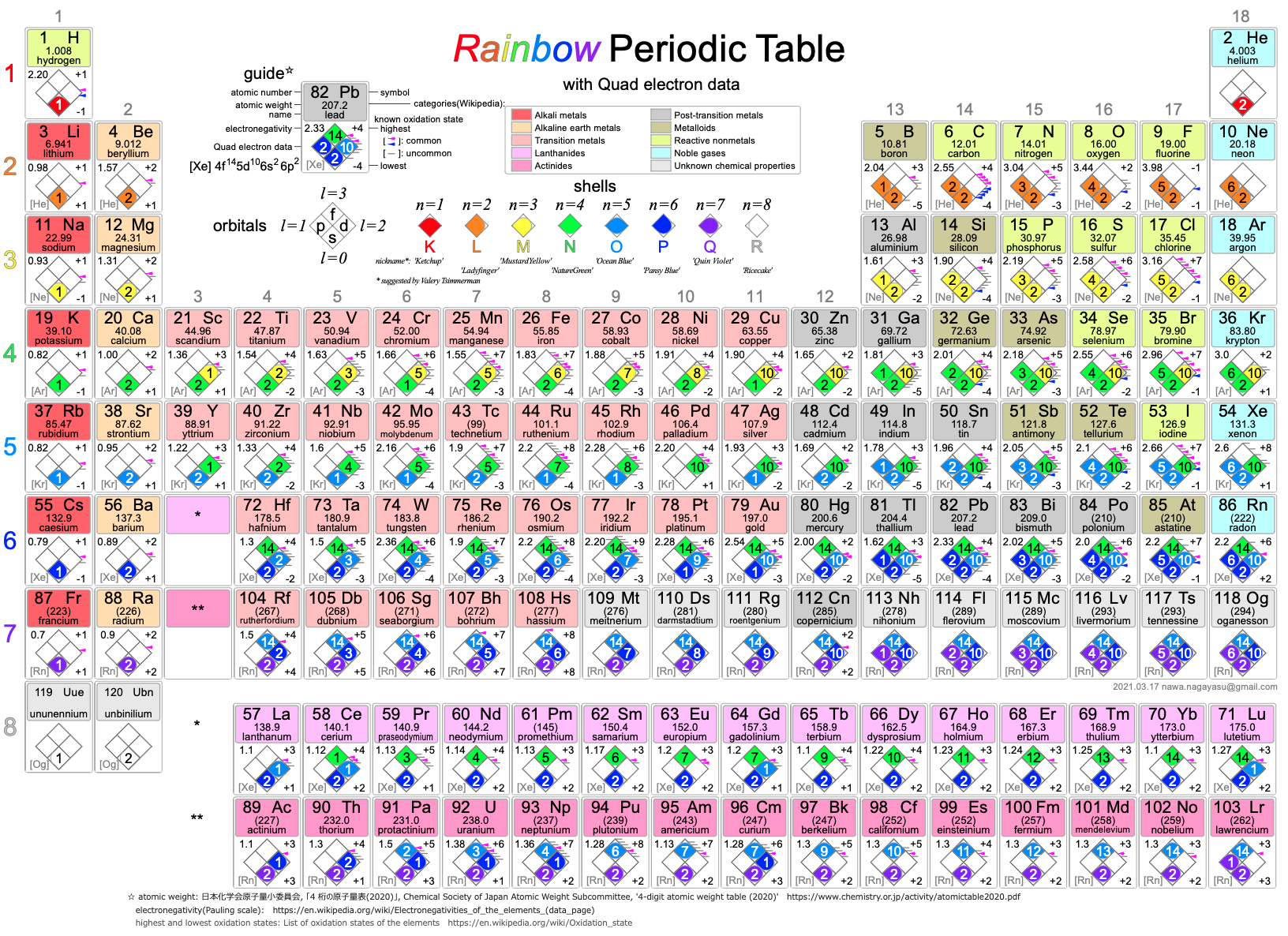
| Year: 2010 | PT id = 507, Type = misc formulation |
Neutronic Schema of the Elements
The Neutronic Schema of the Elements, with LATIN NOTATION by Families and Groups, by Earth/matriX, Science Today, 11" x 17" laminated, color, shows each element of the periodic table with its notation in Latin letters instead of their historically accidental names and symbols:
| Year: 1991 | PT id = 640, Type = non-chem misc |
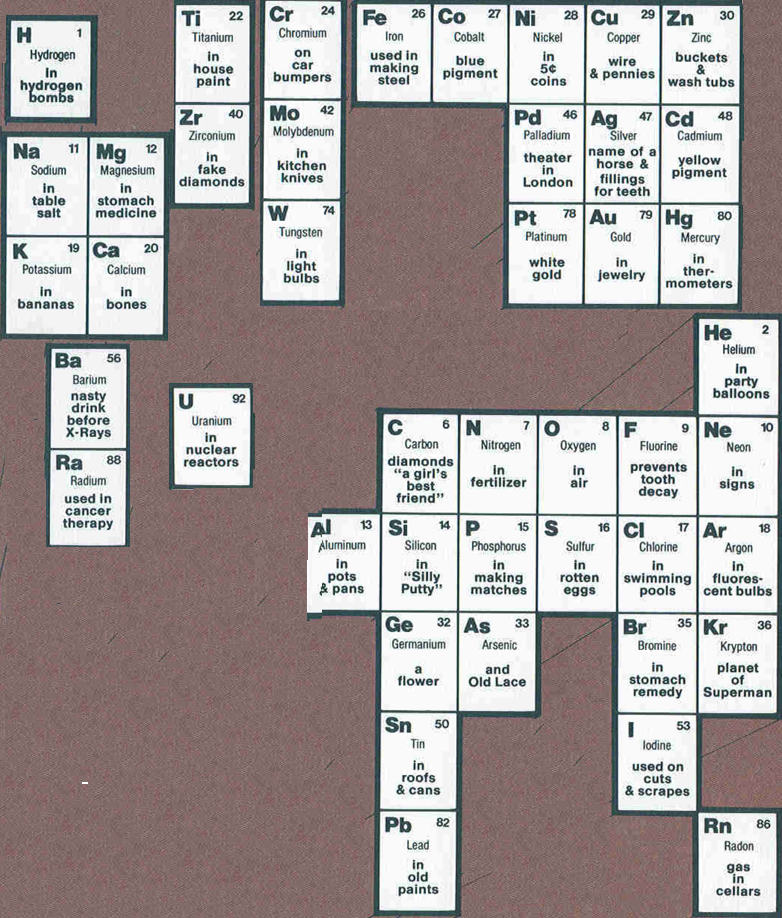
Non-Scientist's Periodic Table
By John T Hortenstine Jr. of the R.W. Johnson Pharmaceutical Research Institute, The Non-Scientists Concept of the Periodic Table of the Elements, for example "Zirconium, in Fake Diamonds", etc.
Click here for the big version.
Thanks to Eric Scerri for the tip!
See the website EricScerri.com and Eric's Twitter Feed.
| Year: 2001 | PT id = 1322, Type = review misc formulation |
Oliver Sacks, Uncle Tungsten: Memories of Tungsten of a Chemical Beyond
René Vernon writes:
On the paperback cover of Oliver Sack's Uncle Tungsten (below) the periodic table shows a 16–wide set of elements at its base. This is quite unusual since this set is normally shown as being 15— or 14— elements wide. See, for example, the table found on the site of the International Union of Pure & Applied Chemistry which shows a 15–wide set of elements at its base.
It looks like the second pair are La and Ac, but what then are two immediately preceding elements?
I suspect they are probably the alkaline earth metals, Ba and Ra. This may be an homage to Mr Rare Earth^ aka Karl A. Gschneidner Jr (1930–2016), who wrote that:
...since Ba has a 4f06s2 configuration, these three elements are the first (Ba), mid (Eu), and end (Yb) members of the divalent 4f transition series.
The notion of 4f0 is not unprecedented; the IUPAC periodic table, with its 15-wide f-block presumably implies La as 4f0 5d1 6s2.
There is some good chemistry going on here, given the pronounced similarities between Ba and the lanthanides, and the alkaline earth metals generally with about 20 properties involved:
- Most of the physical properties of Eu and Yb, "such as the atomic volumes, metallic radii, melting and boiling points, heats of sublimation, compressibilities, and coefficients of expansion are more like those of the alkaline-earth metals, Ca, Sr, and Ba, than those of the rare-earth metals" (Pauling 1960, p. 418; Gschneidner 1964, p. 286).
- Liquid ammonia dissolves certain alkali, alkaline earth, and Ln metals, and... combines with them to form solid compounds. Those metals whose compound-forming ability has been confirmed are Li, Ca, Sr, Ba, Eu and Yb. (Mammano (1970, p. 367)
- The lanthanides are sometimes regarded as trivalent versions of the alkaline earth metals (Evans 1982).
- The electron configurations of lanthanide cations are similar to those of alkaline earth metal cations, as the inner f- orbitals are largely or completely unavailable for bond formation; (Choppin & Rizkalla 1994)
- The lanthanide trivalent cations are essentially spherical and present an environment very similar to alkali and alkaline earth ions towards complex formation... the standard electrode potentials for the lanthanides have similar values and are comparable with the redox potentials of alkaline earth metals (Sastri et al. 2003)
- Ba-Eu-Yb have cubic crystalline structures whereas the rest of the Ln are hexagonal, or rhombohedral in the case of Sm (Russell & Lee 2005)
- There is a close alloying similarity between the lanthanides and Ca, Sr and Ba (Artini 2007)
- Lanthanides are effective mimics of calcium and can stimulate or inhibit the function of calcium-binding proteins (Brayshaw 2019)
- Lanthanide cations can substitute for Ca2+ and Sr2+ cations in host materials for solid state lasers (Ikesue 2013)
- There is a knight’s move relationship between Ca and La:
- The ionic radius of Ca2+ is 114 pm; that of La3+ is 117 pm
- The similarity in sizes means La3+ will compete with Ca2+ in the human body, and usually win on account of having a higher valence for roughly the same hydrated radius
- The basicity of La2O3 is almost on par with CaO2 Freshly prepared La2O3 added to water reacts with such vigour that it can be quenched like burnt lime (CaO)
- The electronegativity of Ca is 1.0; that of La is 1.1.
Kudos to Oliver.
^Pecharsky 2016
Sources
- Artini C (ed.) 2017, Alloys and Intermetallic Compounds: From Modeling to Engineering, CRC Press, Boca Raton, p. 92
- Brayshaw et al. 2019, Lanthanides compete with calcium for binding to cadherins and inhibit cadherin-mediated cell adhesion, Metallomics, vol. 11, no. 5, 2019, pp. 914–924
- Choppin GR & Rizkalla EN 1994, Solution chemistry of actinides and lanthanides, Handbook on the Physics and Chemistry of Rare Earths, pp. 559–590(560)
- Evans WJ 1982, Recent advances in the low valent approach to f-element organometallic chemistry, in McCarthy GJ, Silber HB and Rhyne JJ (eds), The Rare Earths in Modern Science and Technology, vol. 3, Plenum Press, New York, pp. 61–70(62)
- Gschneidner KA 1965, in Seitz F & Turnbull D (eds), Solid State Physics, vol. 16, Academic Press, New York, p. 286
- Ikesue A, Aung YL, Lupei V 2013, Ceramic Lasers, Cambridge University Press, Cambridge, pp. 26, 28
- Mammano N 1970, Solid metal ammonia compounds, in Metal–Ammonia Solutions, Proceedings of an International Conference on the Nature of Metal–Ammonia Solutions: Colloque Weyl II, pp. 367-393 (367), https://doi.org/10.1016/B978-0-408-70122-8.50030-4
- Pauling L 1960, The Nature of the Chemical Bond, 3rd ed., Cornell University Press, Ithaca, p. 418
- Pecharsky V 2016, Karl A. Gschneidner Jr (1930–2016), Nature Materials, vol. 15, no. 1059, https://doi.org/10.1038/nmat4751
- Russell AM & Lee KL 2005, Structure-property relations in nonferrous metals, John Wiley & Sons, Hoboken, inside cover
- Sastri et al. 2003, Modern Aspects of Rare Earths and their Complexes, Elsevier, Amsterdam, pp. 377, 878

| Year: 1936 | PT id = 777, Type = formulation data misc |
Orbital Filling With Electrons
Students of chemistry are often confused why the orbitals fill with electrons: 1s2, 2s2, 2p6, 3s2, 3p6, 4s2, 3d10, 4p6... etc., because the 3d10 seems to be 'out of sequence'.
This 'out of sequence' difficulity is nicely explained if the orbitals are arranged in a slightly different way:
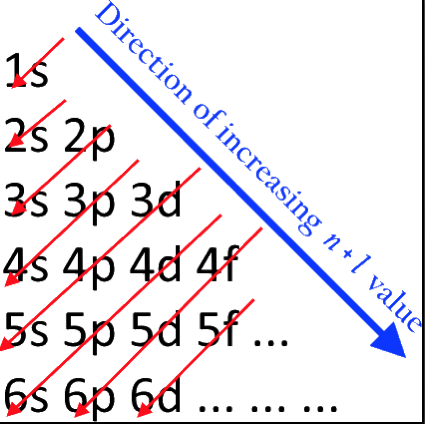
The aufbau principle states that in the ground state of an atom or ion, electrons fill atomic orbitals of the lowest available energy levels before occupying higher levels. For example, the 1s shell is filled before the 2s subshell is occupied. In this way, the electrons of an atom or ion form the most stable electron configuration possible.
The order in which these orbitals are filled is given by the n + ![]() rule, also known as the Madelung rule (after Erwin Madelung), the Janet rule or the diagonal rule.
rule, also known as the Madelung rule (after Erwin Madelung), the Janet rule or the diagonal rule.
Orbitals with a lower n + ![]() value are filled before those with higher n +
value are filled before those with higher n + ![]() values. In this context, n represents the principal quantum number and ? the azimuthal quantum number. The values
values. In this context, n represents the principal quantum number and ? the azimuthal quantum number. The values ![]() = 0, 1, 2, 3 correspond to the s, p, d and f orbital lables.
= 0, 1, 2, 3 correspond to the s, p, d and f orbital lables.
Julio Gutiérrez Samanez writes:
"I send you the diagram below that reconciles quantum mechanics (diagram for filling the electronic cells) with the Janet table or LSPT. Explaining the duplication of periods with the duplication of the quantum number n, and the introduction of Tao (T) spin of the level or spin of the period, which explains the parity of the symmetric periods."
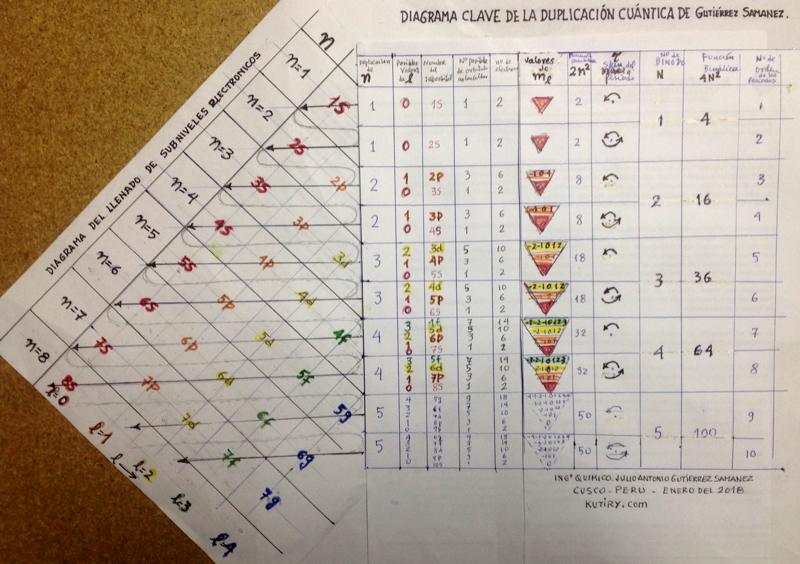
| Year: 2009 | PT id = 261, Type = data misc |
Orbitron Gallery of Atomic Orbitals
The Orbitron gallery of atomic orbitals is a poster available from Mark Winter's Web Elements:
The orbitron web page is here.
| Year: 2018 | PT id = 933, Type = data misc |
Organic Chemist's Periodic Table (another one)
The Periodic Table as seen by an Organic Chemist... a T-Shirt by REDBUBBLE:
Thanks to Marcus Lynch for the tip!
| Year: 2005 | PT id = 242, Type = formulation misc non-chem spiral |
Painting of The Elements
From Gabrielle David's website, here, a painting called Elements, inspired by Melinda Green's Periodic Fractal formulation of 1995:
- The tiniest ball in the center is hydrogen, the next helium, lithium, etc.
- Colors indicate the chemical group.
| Year: 2010 | PT id = 277, Type = formulation misc non-chem |
Periodic Arch of The Elements
Cynthia K. Whitney of Galilean Electrodynamics writes: "In his paper Explaining the periodic table, and the role of chemical triad, Eric Scerri mentioned the existence of at least four different candidate places for Hydrogen: Group 1 (alkali metals - Lithium, etc.), Group 17 (halogens - Fluorine, etc.), Group 14 (Carbon, etc.), or off the Periodic Table entirely, because it is so odd! The four-fold multiplicity (and maybe more) of candidate places for Hydrogen triggered in me the following thought: the excessive multiplicity of candidate places may have to do with the rectangular nature of the Periodic Tables under consideration there." Read more in this pdf file.

| Year: 2018 | PT id = 957, Type = misc |
Periodic Table Song (2018 UPDATE!)
An AsapSCIENCE song + video of the chemical elements. This is a nice alternative to the well known Tom Lehrer Elements song from 1959.
| Year: 2020 | PT id = 1121, Type = misc |
Periodic Table Challenge
IUPAC have developed a Periodic Table Challenge. Answer PT questions at Beginner, Intermediate or Advanced level.

| Year: 2024 | PT id = 1301, Type = misc |
Periodic Table of Food Initiative (PTFI)
Imagine a world where farmers choose to grow specific foods to combat food insecurity and diet-related chronic diseases using practices that are also good for the planet. A world where people everywhere are enabled to select customized diets that support their vitality. This future harnesses the power of food not only as a solution to hunger, but as an essential resource to support the well-being of communities and the environment.
The Periodic Table of Food Initiative (PTFI) is accelerating this future to empower data-driven solutions to our most pressing food system challenges: climate change, biodiversity loss, and malnutrition.
Visit the website: https://foodperiodictable.org


| Year: 2020 | PT id = 1321, Type = misc |
Periodic Table of Rock, Metal And Other Elements
From an Etsy Shop: Periodic Table of Music Poster Print




Thanks to Marcus for the tip!
| Year: 2019 | PT id = 1246, Type = misc element |
Periodic Table of the Elements Coloring Book
Periodic Table of the Elements Coloring Book
Project managing and chemistry overseen by Yann Brouillette (Faculty, Chemistry, Dawson College). Element representations and cover by Dawson College Illustration & Design students (2nd year)* overseen by Meinert Hansen (Faculty, Illustration & Design, Dawson College).
Thanks to René for the tip!
| Year: 2024 | PT id = 1294, Type = data misc |
Periodic Table Regions
Permanent link to the comic: https://xkcd.com/2913/
Image URL (for hotlinking/embedding): https://imgs.xkcd.com/comics/periodic_table_regions.png
Thanks to Marcus for the tip!
| Year: 2009 | PT id = 330, Type = misc |
Periodic Table Table
A Periodic Table Table from Wake Forest Univerity, North Carolina:
| Year: 2024 | PT id = 1328, Type = formulation misc |
pgf-PeriodicTable: Create Custom Periodic Tables
A website that enables the construction of publication quality periodic tables.
Read the package documentation.

| Year: 2016 | PT id = 722, Type = misc |
Philatelic Table of The Elements
Larry French writes:
"I created and first displayed [this Philatelic Table of the Elements] at the ACS National Meeting in San Diego.
"The table has been assembled with each element is represented by a single (or in a few cases a pair) of postage stamps. The table offers a platform for discussions of people, places, sources and applications associated with 114 elements. A total of 73 stamp issuing entities are represented. The table runs from hydrogen, with a North Vietnamese stamp celebrating the test of first Chinese H bomb, to livermorium, represented by a Soviet issue marking the 25th anniversary of the Nuclear Research Institute at Dubna. The table travels from Bolivia's Salar de Uyuni (lithium) to the Enewetak Atoll of the Marshall Islands (einsteinium) and spotlights environmental impacts of phosphate extraction in Nauru and lead mining in Peru. Discoverers and inventors from Moissan and Soddy to Auer and the Curies are met along the way. A range of applications including cesium formate brines in North Sea oil and gas drilling, indium in solar energy conversion, lanthanum in electric cars and technetium in positron emission tomographic medical imaging is included.
"Eventually, my aim is to produce a book which includes an essay for each element and stamp. I have made significant headway with the writing but there is much still to be done."
Larry French
Baker Professor of Chemistry
St. Lawrence University
Click here for full size version
| Year: 2016 | PT id = 770, Type = data misc |
Pictures & Words
A couple of periodic tables from Keith Enevoldsen with information shown in Pictures & Words:
Thanks to Eric Scerri for the tip!
See the website EricScerri.com and Eric's Twitter Feed
| Year: 2005 | PT id = 467, Type = misc |
Pictures, Periodic Table of The Elements
By Keith Enevoldsen, a Periodic Table of The Elements in Pictures, and here.
| Year: 2018 | PT id = 956, Type = misc data |
Places of the Periodic Table
An interactive, searchable Google map of places associated with the developers of the periodic table and with the chemical elements with links to further information brought to you by Carmen Giunta and James Marshall, with the encouragement of the ACS Division of the History of Chemistry (HIST), to mark the International Year of the Periodic Table (IYPT). This is an interactive searchable map of places associated with the developers of the periodic table and with the chemical elements with links to further information.
Examples include places where elements were discovered or synthesized, mineral sources of elements, places where discoverers of chemical periodicity worked, and places for which elements were named. Each entry contains links to further information about the person, place, or event described. The type of site is indicated (for example, lab, residence, mineral source, etc.), as well as whether (to the best of our knowledge) the historical site still exists at the location. For more information on the type of site, please consult this key to the map's fields. The map is intended for educational and informational purposes only, and is not meant as a travel guide. If you wish to visit a site on this map, please consult other resources to confirm access, and use common sense. (Read more here.)
Thanks to Eric Scerri for the tip! See the website EricScerri.com and Eric's Twitter Feed.
| Year: 1984 | PT id = 29, Type = misc |
Planiverse Periodic Table
The Planiverse is set in a 2-D universe that somehow enters into resonance with ours, enabling a computing professor (which Dewdney is) and his class to follow the adventures and scientific education of the hero Yendred (Dewdney backwards).
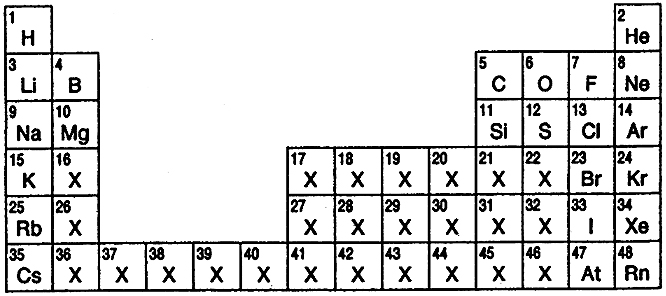
| Year: 2008 | PT id = 265, Type = misc |
Polymer Periodic Table
"The Periodic Table of the elements by Mendeleev was a historic achievement in chemistry and enabled chemists to see the relationship between structure and properties of the basic elements. Polymers also have a strong relationship between structure and properties and this Periodic Table of Polymers is a first attempt to provide a simple codification of the basic polymer types and structures. The diversity of polymer types makes it impossible to include all of the variations in one simple table and this table only includes the most common polymers. At this stage the Table only includes the most common thermoplastics but it will be extended in the future to include thermosets and potentially rubbers and alloys/blends."
| Year: 2007 | PT id = 89, Type = misc |
Periodic Table Stamp, from Spain

| Year: 1975 | PT id = 744, Type = misc review |
Primo Levi's Elements
Primo Levi's elements, from his book The Periodic Table:
Thanks to Eric Scerri for the tip!
See the website EricScerri.com and Eric's Twitter Feed
| Year: 2015 | PT id = 715, Type = misc non-chem |
Protein Complexes, Periodic Table of
The Periodic Table of Protein Complexes, developed by researchers in the UK and published in the in the journal Science (Dec 11, 2015), offers a new way of looking at the enormous variety of structures that proteins can build in nature. More importantly, it suggests which ones might be discovered next and how entirely novel structures could be engineered. Created by an interdisciplinary team led by researchers at the Wellcome Genome Campus and the University of Cambridge, the Table provides a valuable tool for research into evolution and protein engineering.
Read more on Kurzweil and Wild Types (ASBMB Today).
Thanks to Bob Bruner for the tip!
| Year: 2002 | PT id = 531, Type = misc |
Protein Structure Periodic Tables
From a paper by W. R. Taylor, A 'Periodic Table' for Protein Structures, Nature, 2002 Apr 11;416(6881):657-60
Abstract:
Current structural genomics programs aim systematically to determine the structures of all proteins coded in both human and other genomes, providing a complete picture of the number and variety of protein structures that exist. In the past, estimates have been made on the basis of the incomplete sample of structures currently known. These estimates have varied greatly (between 1,000 and 10,000; see for example refs 1 and 2), partly because of limited sample size but also owing to the difficulties of distinguishing one structure from another. This distinction is usually topological, based on the fold of the protein; however, in strict topological terms (neglecting to consider intra-chain cross-links), protein chains are open strings and hence are all identical. To avoid this trivial result, topologies are determined by considering secondary links in the form of intra-chain hydrogen bonds (secondary structure) and tertiary links formed by the packing of secondary structures. However, small additions to or loss of structure can make large changes to these perceived topologies and such subjective solutions are neither robust nor amenable to automation. Here I formalize both secondary and tertiary links to allow the rigorous and automatic definition of protein topology.
This work has been developed by Efrosini Moutevelis and Derek N. Woolfson in their paper A Periodic Table of Coiled-Coil Protein Structures, J. Mol. Biol. (2009) 385, 726–732.
Abstract:
Coiled coils are protein structure domains with two or more ?-helices packed together via interlacing of side chains known as knob-into-hole packing. We analysed and classified a large set of coiled-coil structures using a combination of automated and manual methods. This led to a systematic classification that we termed a "periodic table of coiled coils", which we have made available here. In this table, coiled-coil assemblies are arranged in columns with increasing numbers of α-helices and in rows of increased complexity. The table provides a framework for understanding possibilities in and limits on coiled-coil structures and a basis for future prediction, engineering and design studies.
| Year: 2012 | PT id = 490, Type = misc |
QR Coded Audio Periodic Table of the Elements
The QR coded Audio Periodic Table of the Elements by Vasco D. B. Bonifa?cio, REQUIMTE, Chemistry Department, Faculdade de Cie?ncias e Tecnologia, Universidade Nova de Lisboa, 2829-516 Caparica, Portugal. Email: vbb@fct.unl.pt.
From the paper in The Journal of Chemical Education "A quick response coded audio periodic table of the elements (QR-APTE) was developed using free online resources. The potential of QR-APTE was tested using a smart phone and is envisaged to become a truly powerful tool to teach chemistry to blind and visually impaired students under a mobile-learning environment":
| Year: 2019 | PT id = 1041, Type = misc |
Quantum Victoria Periodic Art
In 2019 Quantum Victoria, an Australian specialist STEM education centre for primary and secondary students and teachers, commissioned a series of 51 images illustrating the birth of the Universe through elements of significance from the Periodic Table.
These will be permanently installed at Quantum Victoria and launched in July to celebrate IYPT2019 - the 150th anniversary of the discovery of the Periodic Table in 1869.
All designs are available as limited edition prints in sizes from A3 to one metre across, printed with archival inks on 100% cotton paper.
Click here or on the image below to access the website and larger versions of the illustrations.
| Year: 1908 | PT id = 995, Type = formulation misc |
Ramsay's Periodic Table
William Ramsay with a section of his 1904 periodic table as a portrait in Vanity Fair.
From the Science History Institute:
In 1892 Ramsay's curiosity was piqued by Lord Rayleigh's observation that the density of nitrogen extracted from the air was always greater than nitrogen released from various chemical compounds. Ramsay then set about looking for an unknown gas in air of greater density, which – when he found it – he named argon.
While investigating for the presence of argon in a uranium-bearing mineral, he instead discovered helium, which since 1868 had been known to exist, but only in the sun. This second discovery led him to suggest the existence of a new group of elements in the periodic table. He and his coworkers quickly isolated neon, krypton, and xenon from the earth's atmosphere.

| Year: 2007 | PT id = 226, Type = misc non-chem |
Rap Periodic Table by NOVI NOV
NOVI NOV writes:
"I infused the Periodic Table into a rap. It was for an old mixtape. I used to record all my music through my camcorder and would delete the video...and mix just the vocals. I kept some of those video files...enjoy"
| Year: 2011 | PT id = 423, Type = misc |
Rapping the Elements
Oort Kuiper writes:
"Many people have heard of Tom Lehrer's 'The Elements' song. One day I decided to search for it online to memorise some stuff about the elements and found out that Daniel 'Harry Potter' Radcliffe had recently recited it on TV. I wondered what he (and the viewers) might have learnt about the elements by listening to it but shock horror... after listening I realised the song hadn't actually told me anything about The Periodic Table, except what's on it! So I decided to do my own song, specifically about The Periodic Table."
| Year: 2010 | PT id = 325, Type = formulation misc data |
Recipe For A Human Shirt
By Sean Fallon and available from Fashionably Geek, A Recipe For Humans Shirt:
| Year: 2016 | PT id = 955, Type = misc data |
Rejected Element Names, Periodic Table of
A periodic table of rejected element names by Andy Brunning's Compound Interest:
Thanks to Eric Scerri for the tip!
See the website EricScerri.com and Eric's Twitter Feed.
| Year: 2025 | PT id = 1337, Type = misc |
RIP Tom Lehrer (The Elements Song) - Periodic Table of Videos
| Year: 2007 | PT id = 225, Type = misc non-chem |
Rock, Periodic Table of
From OscTV:
| Year: 2013 | PT id = 568, Type = data misc |
RSC Visual Elements Periodic Table: Alchemy
From the RSC Website: "Alchemists are often described as the first chemists. They developed an extraordinary language (rather than the chemical symbols we use today) to describe all manner of things, from chemical reactions to philosophical tenets. Click on ‘What is Alchemy?’ to learn about the three aims of the alchemists. Click on each of the alchemical symbols for more information and to see alternative symbols."
| Year: 2016 | PT id = 1123, Type = formulation misc |
Russian Orthodox Elementary System of Unity of the Periodicity of the Electroatoms of the Universe
By Bence Szalai: Russian Orthodox Elementary System of Unity of the Periodicity of the Electroatoms of the Universe
See the 2D version here and the 3d vesion here (in Ukrainian)
| Year: 2009 | PT id = 189, Type = formulation misc |
Russian Periodic Table
A modern Russian periodic table using the Mendeleeve formulation:
An older version of the same formulation (date unknown, 1950s?), from here:
| Year: 2025 | PT id = 1343, Type = non-chem misc |
S Ar Ca Sm: The Elements Required to Deal With Stupidity
S Ar Ca Sm: The Elements Required to Deal With Stupidity

Thanks to Barry for the tip!
| Year: 2020 | PT id = 1149, Type = misc review formulation |
Scerri's Periodic Table of Books About The Periodic Table & The Chemical Elements
From Eric Scerri, a periodic table of books about the periodic table & the chemical elements... many by Eric Scerri himself.
Eric Scerri, UCLA, Department of Chemistry & Biochemistry. See the website EricScerri.com and Eric's Twitter Feed.
There is no particular connection between each of the elements and the book associated with it in the table, with the exception of: H, He, N, Ti, V, Nb, Ag, La, Au, Ac, U, Pu & Og.
The following is a list of references for each of the 118 books featured on Periodic Table of Books About The Periodic Table & The Chemical Elements. Books published in languages other than English are. They include the Catalan, Croatian, French, German, Italian, Norwegian & Spanish languages:
| 1 | H | J. Ridgen, Hydrogen, the Essential Element, Harvard University Press, Cambridge, MA, 2002. |
| 2 | He | W.M. Sears Jr., Helium, The Disappearing Element, Springer, Berlin, 2015. |
| 3 | Li | K. Lew, The Alkali Metals, Rosen Central, New York, 2009. |
| 4 | Be | S. Esteban Santos, La Historia del Sistema Periodico, Universidad Nacional de Educación a Distancia, Madrid, 2009. (Spanish) |
| 5 | B | E.R. Scerri. The Periodic Table, Its Story and Its Significance, 2nd edition, Oxford University Press, New York, 2020. |
| 6 | C | U. Lagerkvist, The Periodic Table and a Missed Nobel Prize, World Scientific, Singapore, 2012. |
| 7 | N | W.B. Jensen, Mendeleev on the Periodic Law: Selected Writings, 1869–1905, Dover, Mineola, NY, 2005. |
| 8 | O | M. Kaji, H. Kragh, G. Pallo, (eds.), Early Responses to the Periodic System, Oxford University, Press, New York, 2015. |
| 9 | F | E. Mazurs, Graphic Representation of the Periodic System During One Hundred Years, Alabama University Press, Tuscaloosa, AL, 1974. |
| 10 | Ne | T. Gray, The Elements: A Visual Exploration of Every Known Atom in the Universe, Black Dog & Leventhal, 2009. |
| 11 | Na | N.C. Norman, Periodicity and the s- and p-Block Elements, Oxford University Press, Oxford, 2007. |
| 12 | Mg | M. Gordin, A Well-Ordered Thing, Dimitrii Mendeleev and the Shadow of the Periodic Table, 2nd edition, Basic Books, New York, 2019. |
| 13 | Al | S. Kean, The Disappearing Spoon, Little, Brown & Co., New York, 2010. |
| 14 | Si | P.A. Cox, The Elements, Oxford University Press, Oxford, 1989. |
| 15 | P | J. Emsley, The 13th Element: The Sordid Tale of Murder, Fire, and Phosphorus, Wiley, New York, 2002. |
| 16 | S | P. Parsons, G. Dixon, The Periodic Table: A Field Guide to the Elements, Qurcus, London, 2014. |
| 17 | Cl | P. Levi, The Periodic Table, Schocken, New York, 1995. |
| 18 | Ar | B.D. Wiker, The Mystery of the Periodic Table, Bethlehem Books, New York, 2003. |
| 19 | K | H. Alderesey-Williams, Periodic Tales, Viking Press, 2011. |
| 20 | Ca | P. Strathern, Mendeleyev's Dream, Hamish-Hamilton, London, 1999. |
| 21 | Sc | D. Scott, Around the World in 18 Elements, Royal Society of Chemistry, London, 2015. |
| 22 | Ti | E. W. Collings, Gerhard Welsch, Materials Properties Handbook: Titanium Alloys, ASM International, Geauga County, Ohio, 1994. |
| 23 | V | D. Rehder, Bioinorganic Vanadium Chemistry, Wiley-Blackwell, Weinheim, 2008. |
| 24 | Cr | K. Chapman, Superheavy, Bloomsbury Sigma, New York, 2019. |
| 25 | Mn | E.R. Scerri, E. Ghibaudi (eds.), What is an Element? Oxford University Press, New York, 2020. |
| 26 | Fe | M. Soon Lee, Elemental Haiku, Ten Speed Press, New York, 2019. |
| 27 | Co | J. Emsley, Nature's Building Blocks, An A-Z Guide to the Elements, Oxford University Press, Oxford, 2001. |
| 28 | Ni | T. James, Elemental, Robinson, London, 2018. |
| 29 | Cu | E.R. Scerri, The Periodic Table, Its Story and Its Significance, Oxford University Press, New York, 2007. |
| 30 | Zn | H. Rossotti, Diverse Atoms, Oxford University Press, Oxford, 1998. |
| 31 | Ga | P. Ball, A Very Short Introduction to the Elements, Oxford University Press, 2004. |
| 32 | Ge | I. Asimov, The Building Blocks of the Universe, Lancer Books, New York, 1966. |
| 33 | As | J. Browne, Seven Elements that Changed the World, Weidenfeld and Nicholson, London, 2013. |
| 34 | Se | N. Raos, Bezbroj Lica Periodnog Sustava Elemenata, Technical Museum of Zagreb, Croatia, 2010. (Croatian) |
| 35 | Br | P. Strathern, The Knowledge, The Periodic Table, Quadrille Publishing, London, 2015. |
| 36 | Kr | A. Ede, The Chemical Element, Greenwood Press, Westport, CT, 2006. |
| 37 | Rb | A. Stwertka, The Elements, Oxford University Press, Oxford, 1998. |
| 38 | Sr | E.R. Scerri, A Tale of Seven Elements, Oxford University Press, New York, 2013. |
| 39 | Y | H.-J. Quadbeck-Seeger, World of the Elements, Wiley-VCH, Weinheim, 2007. |
| 40 | Zr | M. Fontani, M. Costa, M.V. Orna (eds.), The Lost Elements, Oxford University Press, New York, 2015. |
| 41 | Nb | M. Seegers, T. Peeters (eds.), Niobium: Chemical Properties, Applications and Environmental Effects, Nova Science Publishers, New York, 2013. |
| 42 | Mo | E.R. Scerri, Selected Papers on the Periodic Table, Imperial College Press, Imperial College Press, London and Singapore, 2009. |
| 43 | Tc | A. Dingle, The Periodic Table, Elements with Style, Kingfisher, Richmond, B.C. Canada, 2007. |
| 44 | Ru | G. Rudorf, Das periodische System, seine Geschichte und Bedeutung für die chemische Sysytematik, Hamburg-Leipzig, 1904. (German) |
| 45 | Rh | I. Nechaev, G.W. Jenkins, The Chemical Elements, Tarquin Publications, Publications, Norfolk, UK, 1997. |
| 46 | Pd | P. Davern, The Periodic Table of Poems, No Starch Press, San Francisco, 2020. |
| 47 | Ag | C. Fenau, Non-ferrous metals from Ag to Zn, Unicore, Brussells, 2002. |
| 48 | Cd | J. Van Spronsen, The Periodic System of the Chemical Elements, A History of the First Hundred Years, Elsevier, Amsterdam, 1969. |
| 49 | In | M. Tweed, Essential Elements, Walker and Company, New York, 2003. |
| 50 | Sn | M.E. Weeks, Discovery of the Elements, Journal of Chemical Education, Easton PA, 1960. |
| 51 | Sb | P. Wothers, Antimony Gold Jupiter's Wolf, Oxford University Press, Oxford, 2019. |
| 52 | Te | W. Zhu, Chemical Elements in Life, World Scientific Press, Singapore, 2020. |
| 53 | I | O. Sacks, Uncle Tungsten, Vintage Books, New York, 2001. |
| 54 | Xe | E.R. Scerri, (ed.), 30-Second Elements, Icon Books, London, 2013. |
| 55 | Cs | M. Jacob (ed.), It's Elemental: The Periodic Table, Celebrating 80th Anniversary, Chemical & Engineering News, American Chemical Society, Washington D.C., 2003. |
| 56 | Ba | J. Marshall, Discovery of the Elements, Pearson Custom Publishing, New York,1998. |
| 57 | La | K. Veronense, Rare, Prometheus Books, Amherst, New York, 2015. |
| 58 | Ce | N. Holt, The Periodic Table of Football, Ebury Publishing, London, 2016. |
| 59 | Pr | S. Alvarez, C. Mans, 150 Ans de Taules Périodiques a la Universitat de Barcelona, Edicions de la Universitat de Barcelona, Barcelona, 2019. (Catalan) |
| 60 | Nd | L. Garzon Ruiperez, De Mendeleiev a Los Superelementos, Universidad de Oviedo, Oviedo, 1988. (Spanish) |
| 61 | Pm | P. Ball, A Guided Tour of the Ingredients, Oxford University Press, Oxford, 2002. |
| 62 | Sm | S. Esteban Santos, La Historia del Sistema Periodico, Universidad Nacional de Educación a Distancia, Madrid, 2009. (Spanish). |
| 63 | Eu | A. E. Garrett, The Periodic Law, D. Appleton & Co., New York, 1909. |
| 64 | Gd | M.S. Sethi, M. Satake, Periodic Tables and Periodic Properties, Discovery Publishing House, Delhi, India, 1992. |
| 65 | Tb | M. Eesa, The cosmic history of the elements: A brief journey through the creation of the chemical elements and the history of the periodic table, Createspace Independent Publishing Platform, 2012. |
| 66 | Dy | P. Depovere, La Classification périodique des éléments, De Boeck, Bruxelles, 2002. (French). |
| 67 | Ho | F. Habashi, The Periodic Table & Mendeleev, Laval University Press, Quebec, 2017. |
| 68 | Er | W.J. Nuttall, R. Clarke, B. Glowacki, The Future of Helium as a Natural Resource, Routledge, London, 2014. |
| 69 | Tm | R.D. Osorio Giraldo, M.V. Alzate Cano, La Tabla Periodica, Bogota, Colombia, 2010. (Spanish). |
| 70 | Yb | P.R. Polo, El Profeta del Orden Quimico, Mendeleiev, Nivola, Spain, 2008. (Spanish). |
| 71 | Lu | E.R. Scerri, A Very Short Introduction to the Periodic Table, 2nd edition, Oxford University Press, Oxford, 2019. |
| 72 | Hf | D.H. Rouvray, R.B. King, The Mathematics of the Periodic Table, Nova Scientific Publishers, New York, 2006. |
| 73 | Ta | P. Thyssen, A. Ceulemans, Shattered Symmetry, Oxford University Press, New York, 2017. |
| 74 | W | P.W. Atkins, The Periodic Kingdom, Basic Books, New York, NY, 1995. |
| 75 | Re | D.G. Cooper, The Periodic Table, 3rd edition. Butterworths, London, 1964. |
| 76 | Os | E. Lassner, W.-D. Schubert, Tungsten: Properties, Chemistry, Technology of the Element, Alloys, and Chemical Compounds, Springer, Berlin, 1999. |
| 77 | Ir | J.C.A. Boeyens, D.C. Levendis, Number Theory and the Periodicity of Matter, Springer, Berlin, 2008. |
| 78 | Pt | R. Hefferlin, Periodic Systems and their Relation to the Systematic Analysis of Molecular Data, Edwin Mellen Press, Lewiston, NY, 1989. |
| 79 | Au | R.J. Puddephatt, The Chemistry of Gold, Elsevier, Amsterdam, 1978. |
| 80 | Hg | D.H. Rouvray, R.B. King, The Periodic Table Into the 21st Century, Research Studies Press, Baldock, UK, 2004. |
| 81 | Tl | R.E. Krebs, The History and Use of Our Earth's Chemical Elements, Greenwood Publishing Group, Santa Barbara, CA, 2006. |
| 82 | Pb | E. Torgsen, Genier, sjarlataner og 50 bøtter med urin - Historien om det periodiske system, Spartacus, 2018. (Norwegian). |
| 83 | Bi | K. Buchanan, D. Roller, Memorize the Periodic Table, Memory Worldwide Pty Limited, 2013. |
| 84 | Po | D. Morris, The Last Sorcerers, The Path from Alchemy to the Periodic Table, Joseph Henry Press, New York, 2003. |
| 85 | At | T. Jackson, The Elements, Shelter Harbor Press, New York, 2012. |
| 86 | Rn | R.J.P. Williams, J.J.R. Frausto da Silva, The Natural Selection of the Chemical Elements: The Environment and Life's Chemistry, Clarendon Press, Oxford, 1997. |
| 87 | Fr | G. Rudorf, The Periodic Classification and the Problem of Chemical Evolution, Whittaker & Co., London, New York, 1900. |
| 88 | Ra | L. Van Gorp, Elements, Compass Point Books, Manakato, MN, 2008. |
| 89 | Ac | G.T. Seaborg, J.J. Katz, L.R. Morss, Chemistry of the Actinide Elements, Springer, Berlin, 1986. |
| 90 | Th | G. Münzenberg, Superheavy Elements - Searching for the End of the Periodic Table, Manipal Universal Press, India, 2018. |
| 91 | Pa | A. Castillejos Salazar, La Tabla Periòdica: Abecedario de la Quimica, Universidad Autonoma de Mexico, D.F. Mexico, 2005. (Spanish). |
| 92 | U | T. Zoellner, Uranium, Penguin Books, London, 2009. |
| 93 | Np | J. Barrett, Atomic Structure and Periodicity, Royal Society of Chemistry, London, 2002. |
| 94 | Pu | J. Bernstein, Plutonium, Joseph Henry, Washington DC, 2007. |
| 95 | Am | S. Hofmann, Beyond Uranium, Taylor & Francis, London, 2002. |
| 96 | Cm | H.M. Davis, The Chemical Elements, Ballantine Books, New York, 1961. |
| 97 | Bk | P.González Duarte, Les Mils Cares de la Taula Periòdica, Universitat Autonoma de Barcelona, Bellaterra Barcelona, 2005 (Catalan). |
| 98 | Cf | R. Rich, Periodic Correlations, Benjamin, New York, 1965. |
| 99 | Es | E. Rabinowitsch, E. Thilo, Periodisches System, Geschichte und Theorie, Stuttgart, 1930. (German). |
| 100 | Fm | P.K. Kuroda, The Origin of the Chemical Elements, and the Oklo Phenomenon, Springer-Verlag, Berlin, 1982. |
| 101 | Md | G. Villani, Mendeleev, La Tavola Periodica Degli Elementi, Grandangolo, Milan, 2016. (Italian). |
| 102 | No | J. Russell, Elementary: The Periodic Table Explained, Michael O'Mara, London, 2020. |
| 103 | Lr | P. Enghag, Encyclopedia of the Elements, Wiley-VCH, Weinheim, 2004. |
| 104 | Rf | R.J. Puddephatt, The Periodic Table of the Elements, Oxford University Press, Oxford, 1972. |
| 105 | Db | L. Ohrström, The Last Alchemist in Paris, Oxford University Press, New York, 2013. |
| 106 | Sg | N.N. Greenwood, E. Earnshaw, Chemistry of the Elements, 2nd edition, Elsevier, Amsterdam, 1997. |
| 107 | Bh | R. Luft, Dictionnaire des Corps Simples de la Chimie, Association Cultures et Techniques, Nantes, 1997. (French) |
| 108 | Hs | Science Foundation Course Team, The Periodic Table and Chemical Bonding, The Open University, Milton Keynes, 1971. |
| 109 | Mt | W.W. Schulz, J. Navratil, Transplutonium Elements, American Chemical Society, Washington D.C., 1981. |
| 110 | Ds | I. Nechaev, Chemical Elements, Lindsay Drummond, 1946. |
| 111 | Rg | F. Hund, Linienspektren und Periodisches System Der Elemente, Springer, Berlin, 1927. |
| 112 | Cn | F.P. Venable, The Development of the Periodic Law, Chemical Publishing Co., Easton, PA, 1896. |
| 113 | Nh | O. Baca Mendoza, Leyes Geneticas de los Elementos Quimicos. Nuevo Sistema Periodico, Universidad Nacional de Cuzco, Cuzco, Peru, 1953 (Spanish). |
| 114 | Fl | B. Yorifuji, Wonderful Life with the Elements, No Starch Press, San Francisco, 2012. |
| 115 | Mc | D.I. Mendeléeff, The Principles of Chemistry, American Home Library, New York, 1902. |
| 116 | Lv | A. Lima-de-Faria, Periodic Tables Unifying Living Organisms at the Molecular Level: The Predictive Power of the Law of Periodicity, World Scientific Press, Singapore, 2018. |
| 117 | Ts | H.B. Gray, J.D. Simon, W.C. Trogler, Braving the Elements, University Science Books, Sausalito, CA, 1995. |
| 118 | Og | E.R. Scerri, G. Restrepo, Mendeleev to Oganesson, Oxford University Press, New York, 2018. |
| Year: 2020 | PT id = 1153, Type = review formulation misc |
Scerri's Periodic Table of Books About The Periodic Table & The Chemical Elements by ERS
From Eric Scerri, a periodic table of books about the periodic table & the chemical elements... by Eric Scerri, including translations.
Eric Scerri, UCLA, Department of Chemistry & Biochemistry. See the website EricScerri.com and Eric's Twitter Feed.
| Year: 2019 | PT id = 1005, Type = formulation misc 3D spiral |
Schaltenbrand's Helical Gathering of the Elements
From the RSC Website:
"A glistering, shining spiral made of silver, gold, platinum, palladium and a diamond forms the show-stopping apex of the tribute from the University of Cambridge's St Catharine's college to the International Year of the Periodic Table.
"Commissioned to match George Schaltenbrand's 1920 design for a helical gathering of the elements – albeit extended to all 118 current elements – and signed by Yuri Oganessian, it is almost certainly the most expensive periodic table in the world."


Thanks to Eric Scerri for the tip!
See the website EricScerri.com and Eric's Twitter Feed.
| Year: 2010 | PT id = 668, Type = misc |
Science Museum Lockers
From Kotaku:
While visiting the Nagoya City Science Museum, Twitter user Kantaku noticed something very cool, the coin lockers.
The name of each element is written below each symbol in Japanese, allowing visitors to store their belongings in Helium, Calcium, Oxygen, Potassium and more.
The number of each locker corresponds to the element. So, locker 21 is Scandium as it's the twenty-first element on the periodic table. Locker 3? It's Lithium, like it is on the periodic table, and so on. Dibs on Krypton!
Thanks to Eric Scerri for the tip!
See the website EricScerri.com and Eric's Twitter Feed
| Year: 2019 | PT id = 1067, Type = formulation 3D spiral misc |
Scott Van Note Periodic Table Sculpture
On the Saatchi Art website, a 3D periodic table Sculpture by Scott Van Note.
Sculpture: Metal (Bronze). Ten made for the local ASM international chapter.
Loops and changes of direction show electron shell filling. S,P,D,F with S just a change of direction. Continuous spiral from top to bottom. New loops introduce as the electron shell would. Does not show the out-of-order shell filling.
Keywords: periodic, science, sculpture, functional, nerd
Thanks to Roy Alexander for the tip!
| Year: 1969 | PT id = 1270, Type = formulation data misc |
Seel-Klechkovskii Version of Madelung's Rule for Orbital Filling
Seel F., Bild der Wissenschaft, 6, 44 (1969), a monthly popular scientific journal.
- Klechkovskii VM, Dokl. Akad. Nauk. SSSR, 135. 855 (1980). [In Russian]
- Klechkovskii VM, Zh. Eksperim. i. Teoret. Fiz., 41. 465 (1961). [In Russian]
Thanks to René for the tip!
| Year: 1948 | PT id = 231, Type = formulation misc |
Segrè Chart of Elements & Isotopes
The Segrè chart of elements and isotopes arranges atomic nuclei by numbers or protons and numbers of neutrons and is a table of nuclides. There are various ways the axes can be arranged. From elsewhere in this chemogenesis web book:
And from Wikipedia:
| Year: 2023 | PT id = 1287, Type = formulation element data misc spiral |
Semicircular Hybrid Chart of the Nuclides
Nawa Nagayasu has produced a new version of the Segrè Chart of the Nuclides.
Nawa writes:
"The chart has the number of neutrons on the [curved] horizontal axis and the number of protons (atomic number) on the vertical axis. I used the IAEA colour coding [scheme]. JAEA's half-life ranks are indicated by simple numbers, not rounded frames.
"In order to fit the whole chart into a semicircle, the axis representing the number of neutrons was made a spiral-like curve. For clarity, the number of neutrons is shown in the middle of each curve."
Yuri Oganessian has commented:
"Nawa Nagayasu is an original and talented designer. After all, it is not easy to work with 118 elements, but now also with isotopes, of which there are more than 3000. The fan design looks attractive and this is very important. This will make people, especially school age, guess the numbers that are written there. So they will gradually delve into the content of the Table, a truly brilliant creation."
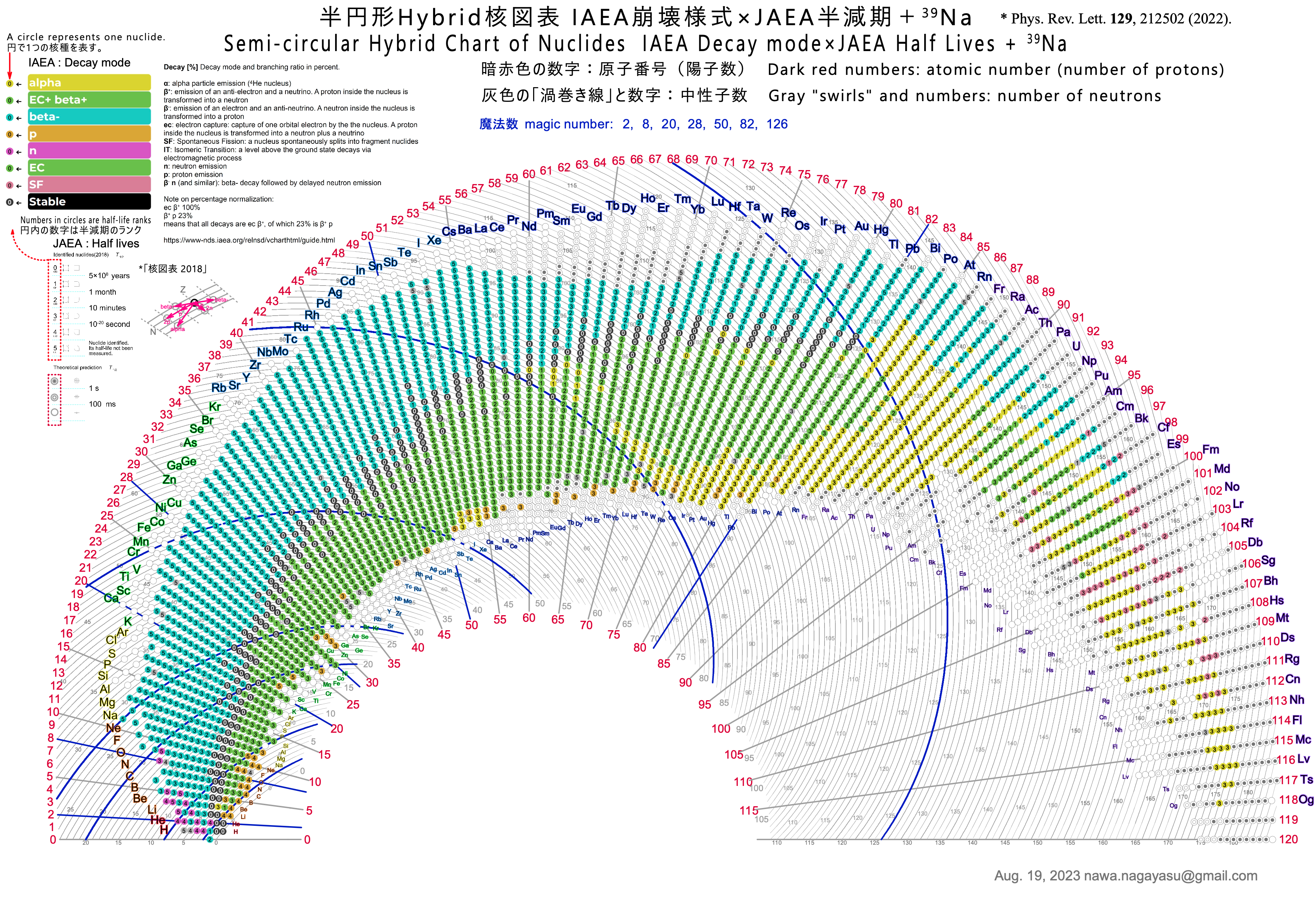
| Year: 2019 | PT id = 1031, Type = misc |
Setting The Table
The journal Science gives "a visual brief history" of the periodic table, with some neat graphics showing the PT grew and changed with time. (You will need to visit the webpage to see the cool graphics inaction):
Thanks to Eric Scerri for the tip!
See the website EricScerri.com and Eric's Twitter Feed
| Year: 2013 | PT id = 590, Type = formulation non-chem misc |
Shapes Periodic Table
By ScienceIsGolden.com comes the Periodic Table of Shapes. The site is worth clicking around, as there is a lot of good PT stuff to find:
| Year: 2013 | PT id = 609, Type = misc |
Simplest Periodic Table
No numbers, just dots by London-based graphic designer Alison Haigh.
Each element is represented by a visualization of its electronic structure, rather than by numbers and letters:
| Year: 1998 | PT id = 376, Type = misc non-chem |
The Simpsons Periodic Table
A Periodic Table from the Simpsons... look closely and it is not quite as expected...

Lisa Gets an "A" Season Ten (1998-1999) - 23 Episodes [204-226] Episode 210 Original Airdate on FOX: 22-Nov-1998
Skinner: We can buy =real= periodic tables instead of these promotional ones from Oscar Meyer.
Krabappel: Who can tell me the atomic weight of bolognium?
Martin: Ooh ... delicious?
Krabappel: Correct. I would also accept snacktacular.
| Year: 2000 | PT id = 707, Type = misc |
Sistema Peryodico
A famous Spanish periodic table with puns/jokes on the element names. (Click here for a larger version.)
- For example, in Spanish hydrogen is "Hidrogen" which is made up from the root words "water" and "genius"... hence the genie from Aladdin.
- Likewise, the Spanish for chromium, "Cromo" is the same as the word for stamp... hence a picture of a stamp.
This is one for Spanish speaking chemists!
| Year: 2005 | PT id = 312, Type = data misc |
Smart Elements
Smart Elements, at smart-elements.com, is a company selling physical samples of chemical elements for research, education & collection.
- High purity Elements for Science, Laboratory and Education
- High-End element samples for collectors, museums, lectures and exhibitions
- Free picture service for educational purposes
- Professional advisory service
- Purchase of Elements
Smart Elements sell numerous examples of all the naturally occuring elements. For example they sell 26 copper, Cu, products including samples in acrylic blocks, vials and bottles:
| Year: 2008 | PT id = 221, Type = misc non-chem |
Snelson Atom
"Kenneth Snelson's Portrait of an Atom is a multi-media artwork that [attempts to] describe the atom's electronic architecture. If you happen to have a rapid prototype printer this STL file can be downloaded free for creating a desktop model at any preferred size of the Snelson atom."

| Year: 2013 | PT id = 577, Type = misc |
Spectraphonic Periodic Table
Relax and enjoy the Spectraphonic Periodic Table of the Elements, the first and only periodic table where you may hear the characteristic light signature (spectra) of each element dropped forty octaves into the auditory range. Hear the sounds of the atoms. Experience the building blocks of reality... of the Universe... of You.:
| Year: 2013 | PT id = 632, Type = misc data |
Spider Chart of The Periodic Table of Chemical Elements
A Spider Chart linking together various ideas about the Periodic Table of the Chemical Elements by Roy Alexander (of Alexander Arrangement fame).
Click here to embiggen the image:
| Year: 2019 | PT id = 1247, Type = misc |
Spinor 150th Periodic Table Anniversary
Spinor Magazine's 150th Periodic Table Anniversary front cover:

| Year: 2008 | PT id = 175, Type = formulation spiral misc |
Spiral Periodic Table
A spiral periodic table available as a poster, binder, cup, T-shirt, etc. by Vectoria:
| Year: 2025 | PT id = 1334, Type = formulation spiral non-chem misc 3D |
Spiral-Struktur-Periodensystem der Elemente
Martin Röder writes:
https://archive.org/details/spiral-struktur-periodensystem
This is a new periodic table of the elements, which classifies the elements as follows:
1. according to the structural groups of occult chemistry
2. on the basis of positive charges (proton number)
3. according to the number of primary atoms (mass)Detailed description under "PDF" in "Article - Spiral-Structure-Periodic System 250621.pdf"
Overview chart to print out: "Spiral-Structure-Periodic System of the Elements (with structure groups of Occult Chemistry) 250621.pdf".
2D and 3D animations under: "ANIMATED GIF" Graphics and diagrams under: "PNG" video with 3D animation



| Year: 2018 | PT id = 930, Type = misc |
Stamps Commemorating Yuri Organeson
Stamps Commemorating Yuri Organeson, Issued by Armenia, Dec 28th, 2017

| Year: 2013 | PT id = 647, Type = misc |
Stardust Periodic Table of The Elements
Inspired by Carl Sagan, Stardust Elements introduces a display case of the periodic table of the elements with real high purity samples:
| Year: 2019 | PT id = 1109, Type = misc |
Students, Periodic Table of
Translated from Catalan: Representation of the periodic table made by the students of Baccalaureate of the INS Santa Coloma de Farners in commemoration of the international year of the Periodic Table (2019). Photo: Emma Masó

Thanks to Eric Scerri – who appears – for the tip!
See the website EricScerri.com and Eric's Twitter Feed.
| Year: 2018 | PT id = 971, Type = data misc |
Superconductivity of Hydrides Periodic Table
Scientists from Moscow Institute of Physics and Technology and Skoltech have demonstrated the high-temperature superconductivity of actinium hydrides and discovered a general principle for calculating the superconductivity of hydrides based on the periodic table alone. The results of their study were published in The Journal of Physical Chemistry Letters.
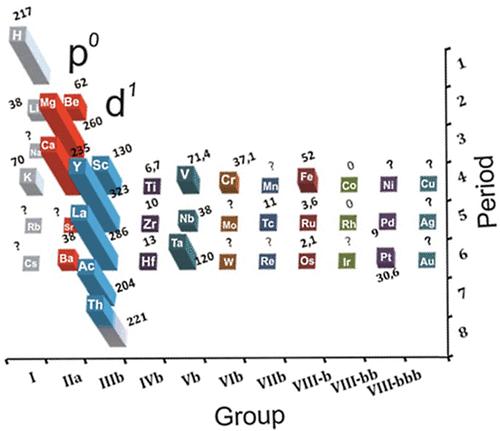
Thanks to Eric Scerri for the tip! See the website EricScerri.com and Eric's Twitter Feed.
| Year: 2009 | PT id = 273, Type = misc |
Sweater With Periodic Table
A sweater with a periodic table and stitch pattern details, as seen on the This and That blog:
| Year: 2014 | PT id = 649, Type = misc non-chem |
Table Lab
The Table Lab with several Periodic Tables:
Animal, Cat, Christmas, Crayon, Dinosaur, Dog, Farm, Mixology, Sushi Bar & USA... as well as Classic:
Thanks to Eric Scerri for the tip!
See the website EricScerri.com and Eric's Twitter Feed
| Year: 2014 | PT id = 633, Type = misc |
Table of Organic Chemicals and Their Smells
A table of organic chemicals and their smells:
Thanks to Marcus Lynch for the tip!
| Year: 2019 | PT id = 1089, Type = data misc |
Term Symbol of the Chemical Elements
From Wikipedia (edited):
In quantum mechanics, the term symbol is an abbreviated description of the (total) angular momentum quantum numbers in a multi-electron atom. Each energy level of an atom with a given electron configuration is described by not only the electron configuration but also its own term symbol, as the energy level also depends on the total angular momentum including spin. The usual atomic term symbols assume LS coupling (also known as Russell-Saunders coupling or spin-orbit coupling). The ground state term symbol is predicted by Hund's rules.
Neutral atoms of the chemical elements have the same term symbol for each column in the s-block and p-block elements, but may differ in d-block and f-block elements, if the ground state electron configuration changes within a column.
For (far more) information refer to the Wikipedia Term Symbol page.
And from T.F. Yen, Chemistry for engineers, Imperial College Press, London, p. 53 (2008)
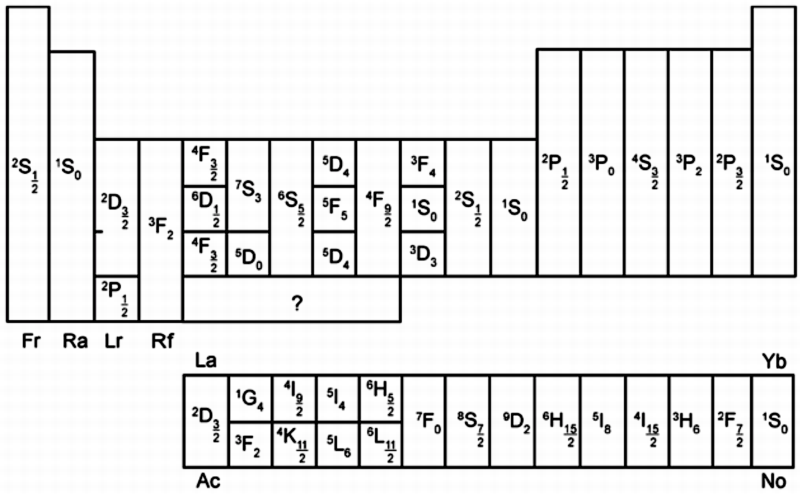
| Year: 2017 | PT id = 758, Type = misc |
Tetris Version of the Periodic Table
From a Piled Higher and Deeper [Ph.D. Comics] video, here, a Tetris version of the Periodic Table:
| Year: 2026 | PT id = 1347, Type = misc |
The Approximate Chemical Formula of the (Observable) Universe
From xkcd, The Approximate Chemical Formula of the (Observable) Universe:

Thanks to Marcus for the tip!
| Year: 2008 | PT id = 992, Type = misc |
Twin Vortex Theory
By consulting engineer Anthony H. Davis comes the Twin Vortex Theory of nuclear structure. Read more in the attached PDF and watch the embedded video below:
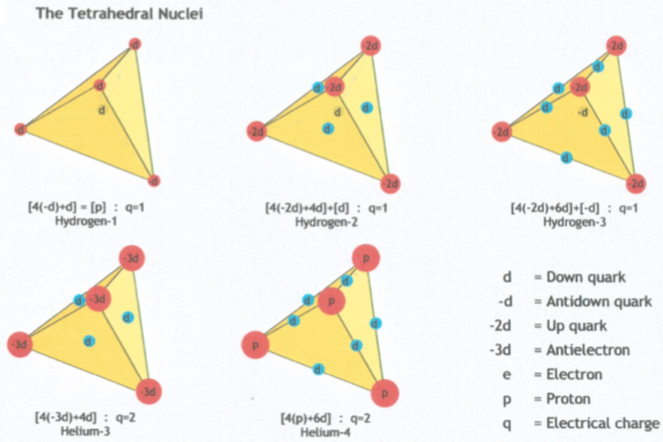
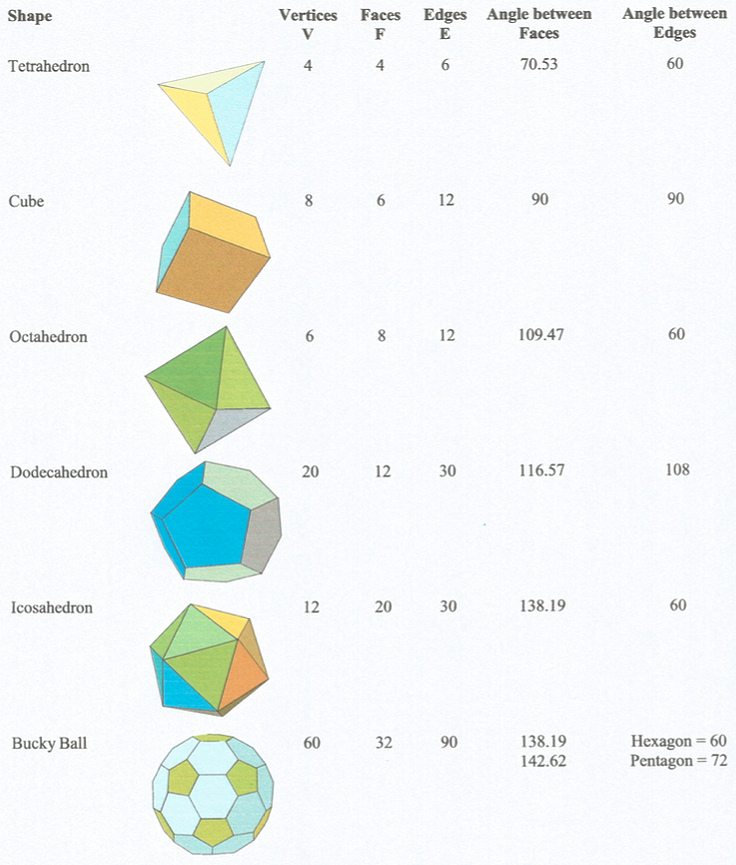
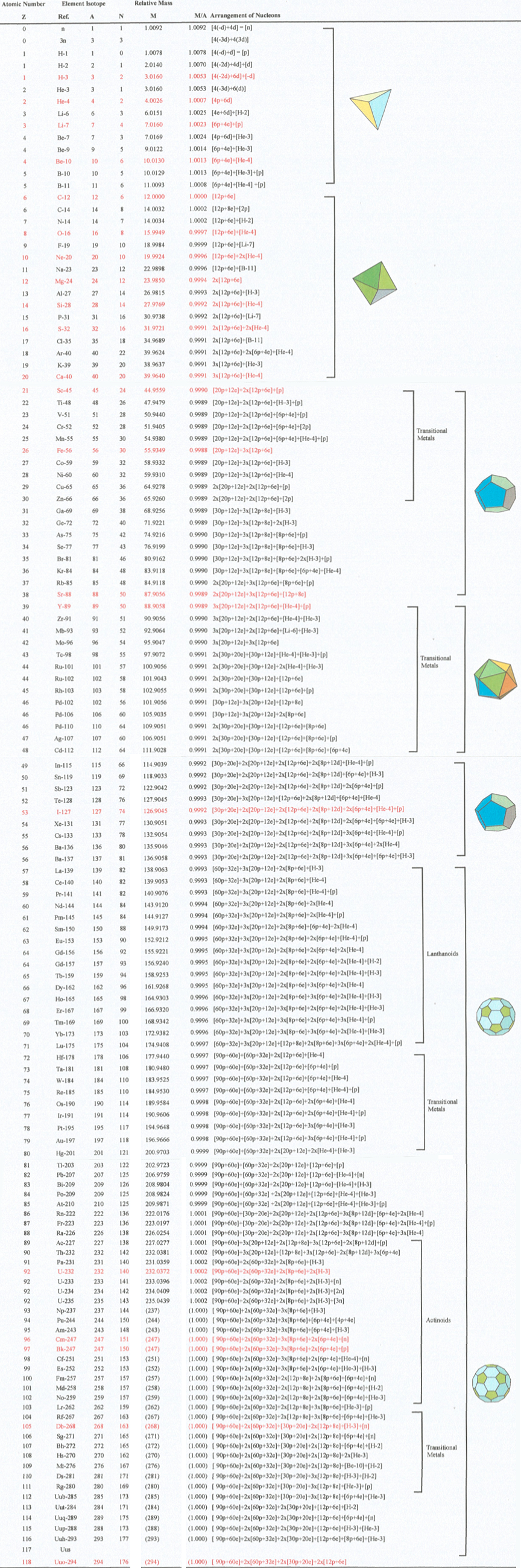
| Year: 2013 | PT id = 557, Type = non-chem misc |
Twitter Handle Periodic Table
From Stuart Cantrill of Chemical Connections:
So, you're a chemist and you've finally decided to find out what all the fuss is about with this thing called Twitter. You decide to sign up, but, for whatever reason, you don't fancy using your own name. Maybe an element; that would be cool wouldn't it?
You are a chemist after all. Maybe you work with Grubbs' catalyst a lot, and you like the idea of being @ruthenium. Or perhaps Stille/Suzuki/Heck couplings are your thing and so @palladium seems appropriate.
Not into metals? Well why not @fluorine, @helium or @bromine?
Well, I'm sorry to report that all of those are taken, but there are 114 named elements (we're ignoring those ununelementium placeholder names) to choose from. Surely some of the more exotic elements must be there for the taking?
Well, no. Gone. All of 'em.
Thought you'd sneak in and claim one of the two newest additions to the periodic table @flerovium or @livermorium? Sorry, you've been beaten.
Here is the periodic table of Twitter, with all the accounts linked:
| Year: 2004 | PT id = 112, Type = misc |
Periodic Tables in Two Hundred Languages
Periodic Tables of the Elements in Two Hundred Languages:
| Year: 2010 | PT id = 993, Type = misc |
UCL Lecturers, Periodic Table Of
This table attempts to chart the evolution of the department from its inception in 1826 to the present day through the members of academic staff who have taught here. The names appear in order of appointment. In the interests of clarity, the f-block has been suppressed in its entirety. This move in no way reflects Departmental Policy. UCL Chemistry does not discrimate against particular azimuthal quantum numbers and has no comment to make regarding elements with n > 6.
Click on a name to get details of the time they spent here and perhaps even see a picture of the person. With time we hope to include biographical information, anecdotes, lists of key publications, pictures of mountains they climbed, and other goodies from our archives. An alphabetical list is also available. Disclaimer: Positions in the chart are provisional and no conclusions concerning the moral fibre of any individual should be drawn on the basis of their position in a group or by virtue of a diagonal relationship to anyone else.
Produced by Dr Andrea Sella, with thanks to Ms Tina Simon for the design idea.
| Year: 2019 | PT id = 1042, Type = data misc |
Ultimate Periodic Table by Goodfellow
While the 'ultimate' periodic table by Goodfellow may not appear to be very ultimate, it does actually a possess a very rare property: Goodfellow is a materials company that supplies most of the chemical elements for industrial and research use.
By clicking on the element palladium, various facts about, and properties of, Pd are shown. Additionally, Goodfellow can supply:
| Year: 2013 | PT id = 611, Type = formulation misc |
Underground Map of the Elements
By Dr Mark Lorch of the University of Hull, an Underground Map of the Elements.
From here: "My son loves trains. So I came up with a train related twist to an inspection of the periodic table. We sat and cut up a copy of the table and then rearranged each element as a 'station' on an underground rail system. Each line represents a characteristic shared by the elements on that line":
Thanks to Eric Scerri for the tip!
See the website EricScerri.com and Eric's Twitter Feed.
| Year: 2007 | PT id = 307, Type = formulation misc |
University of Jaén (Spain) Wall Mural Periodic Table
From November of 2007 a large Periodic Table placed on the main facade of Sciences Building in the University of Jaén (Spain) welcome everybody.
The table was made in honor of Mendeleev on the 100 aniversary of his death and on the occasion of the Spanish Year of Science according to the concept and design of the Spanish Chemist Antonio Marchal Ingrain, who was inspired in a postage stamp launched that year in Spain.
The artistic mural is composed of 117 tiles of 20 x 30 cm, one for each of the elements known to date, reaching a final dimensions of 2.8 x 3.6 meters. Apart from the traditional information with which students are familiar, such as the atomic number, atomic mass and the chemical symbol of the element, each of the ceramics incorporate information concerning the meaning of its name in Latin or Greek, the year and the name of the person or group of people who discovered it or isolated.
Dr. Antonio Marchal, UNIVERSITY OF JAÉN, SPAIN

| Year: 2017 | PT id = 939, Type = misc |
University of Murcia's Oversize Periodic Table
From C&EN:
The faculty at the University of Murcia in Spain has a giant periodic table of the elements emblazoned on the facade of the school's chemistry building.
Covering 150 m2, the table displays 118 elements identified by their symbol, atomic number, and atomic mass. "It could be the world's largest permanent periodic table placed on a wall", Pedro Lozano Rodriguez, dean of the department of chemistry at the school, tells Newscripts.
A number of local companies chipped in to place the oversize chart on the side of the building, including the energy firm Repsol, the brewer Estrella de Levante, and personal care products maker Tahe Productos Cosméticos. Lozano says the display will serve as part of an introductory chemistry lesson for incoming freshmen.

| Year: 2020 | PT id = 1295, Type = misc |
University of UNAM Periodic Table
Mexico City, University of UNAM Chemistry Department

Thanks to Marcus for the tip!
| Year: 2016 | PT id = 717, Type = formulation misc |
Valentine Periodic Table
A Valentine Periodic Table by Claude Bayeh:

| Year: 2017 | PT id = 745, Type = misc |
Venn Diagram of the Chemical Elements and the United States
A rather nice Venn diagram showing the intersection of the chemical element symbols and the States of the Union (based on an origional found at I Love Charts):
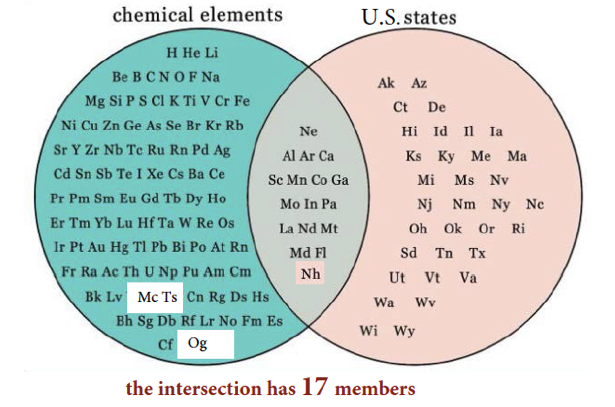
Thanks to Eric Scerri for the tip!
See the website EricScerri.com and Eric's Twitter Feed
| Year: 2018 | PT id = 950, Type = data misc |
Waterloo Periodic Table Project/Projet Tableau Périodique
To celebrate the International Year of Chemistry (IYC), Chem 13 News magazine together with the University of Waterloo's Department of Chemistry and the Faculty of Science encouraged chemistry educators and enthusiasts worldwide to adopt an element and artistically interpret that element.
The project created a periodic table as a mosaic of science and art. Students from all Canadian provinces and territories, 20 U.S. states and 14 countries researched, created and designed the elemental tiles. We created a poster, wall mural and a mobile app. The app includes the creative process behind each tile along with basic atomic properties of the element. The free app work to truly highlight the artistic expression of the Periodic Table Project. Thank you to all the teachers and students who participated in the collaborative Periodic Table Project.
Read more on the University of Waterloo website.
Click here image to enlarge the PT below.
| Year: 2022 | PT id = 1236, Type = data misc element |
Which Element is the Best?
The That Chemist YouTube channel asks "Which Element is the Best? Elements 1-20 & Elements 21-40"
That Chemist is a synthetic organic chemist and his bias is in that direction although he gives a variety of examples:
| Year: 2014 | PT id = 637, Type = misc |
Winter Olympics Opening Ceremony Periodic Table
From the 2014 Winter Olympics Opening ceremony, a Russian periodic table. See the whole video on RuTube.
Thanks to Eric Scerri for the tip!
See the website EricScerri.com and Eric's Twitter Feed.
| Year: 2012 | PT id = 543, Type = misc review |
Wonderful Life with the Elements
From the Japanese artist Bunpei Yorifuji comes Wonderful Life with the Elements, an illustrated guide to the periodic table that gives chemistry a friendly face, available from Amazon.
In this super periodic table, every element is a unique character whose properties are represented visually: heavy elements are fat, man-made elements are robots, and noble gases sport impressive afros. Every detail is significant, from the length of an element's beard to the clothes on its back. You'll also learn about each element's discovery, its common uses, and other vital stats like whether it floats - or explodes - in water.
There is also a full review with more images from Wired.
| Year: 2021 | PT id = 1207, Type = misc |
World's Largest Periodic Table Created on ECU's Science Building
From architectureanddesign.com.au:
"The new science building at Edith Cowen University (ECU), Joondalup Campus in Perth stands out for its striking façade, which features the world's largest periodic table. The thoughtful design by Silver Thomas Hanley Architects responded to the brief to deliver a bold and sophisticated architectural statement in the urban setting. The façade featuring the periodic table celebrates the building's purpose as a centre of scientific research and learning. Based on the university vice chancellor Professor Steve Chapman's idea, the periodic table is an enormous 662 square metres, spanning the entire front façade of the building."

Thanks to Eric Scerri for the tip!
See the website EricScerri.com and Eric's Twitter Feed.
| Year: 2010 | PT id = 377, Type = formulation misc data |
World's Smallest Periodic Table
The World's Smallest Periodic Table:
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
© Mark R. Leach Ph.D. 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com
This free, open access web book is an ongoing project and your input is appreciated.