Periodic Table |
 |
 |
 |
 |
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
The INTERNET Database of Periodic Tables
There are thousands of periodic tables in web space, but this is the only comprehensive database of periodic tables & periodic system formulations. If you know of an interesting periodic table that is missing, please contact the database curator: Mark R. Leach Ph.D. The database holds information on periodic tables, the discovery of the elements, the elucidation of atomic weights and the discovery of atomic structure (and much, much more).
| Year: 1966 | PT id = 248, Type = formulation misc |
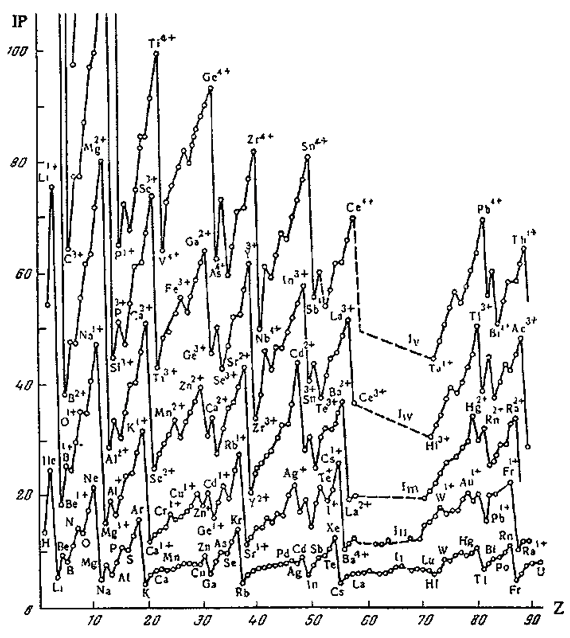
Periodic Table of Ions
From Concept of Chemical Periodicity: from Mendeleev Table to Molecular Hyper-Periodicity Patterns E. V. Babaev and Ray Hefferlin, here.
"One intriguing problem that arises from with the periodic table of atoms is the possibility of constructing periodic systems of ions, V. K. Grigorovich, Periodic Law of Mendeleev and Electronic Structure of Metals, Nauka Publ.: Moscow, 1966 (in Russian). An atom can be completely or partially ionized to a cation by removing electrons or transformed into an anion by the addition of new electrons. The energy required for a few consecutive ionisations of atoms is plotted against the atomic number. One can see that the curves are periodic, and hence it is possible to construct periodic tables for mono-, di-, and multi- charged cations. If we look at the dispositions of the maxima and minima of the curves and compare them with those for atoms, it becomes evident that the magic numbers of electrons for ions are the same as for neutral atoms. Therefore, the number of electrons (but not the charge of the nucleus) is responsible for the periodicity of ions."
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
© Mark R. Leach Ph.D. 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com
This free, open access web book is an ongoing project and your input is appreciated.