Periodic Table |
 |
 |
 |
 |
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
The INTERNET Database of Periodic Tables
There are thousands of periodic tables in web space, but this is the only comprehensive database of periodic tables & periodic system formulations. If you know of an interesting periodic table that is missing, please contact the database curator: Mark R. Leach Ph.D. The database holds information on periodic tables, the discovery of the elements, the elucidation of atomic weights and the discovery of atomic structure (and much, much more).
| Year: 2013 | PT id = 567, Type = formulation |
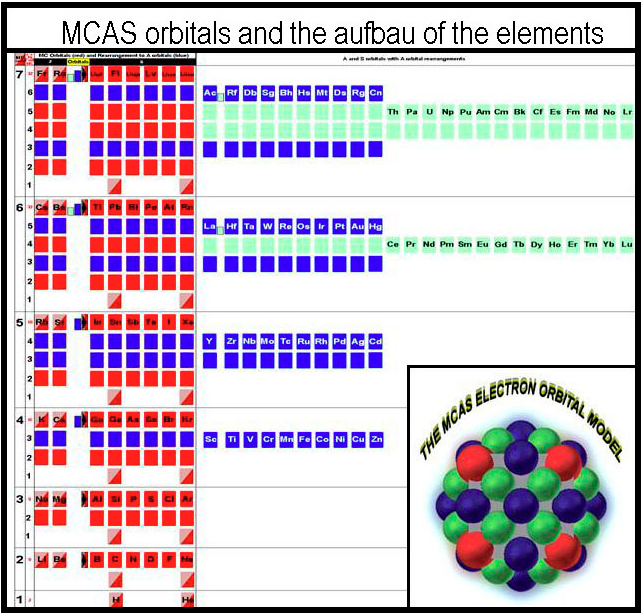
MCAS Electron Orbital Filling
From Joel M Williams:
"While the periodic table arrangement is usefully interpreted in columns of similar behavior, it is erroneous to imply that the underlying orbitals are all the same for all the elements in the columns of a block. Sub-orbital information has been excluded! From the standpoint of chemistry, the rule of eight would have provided better imagery on which to build an orbital system than was Bohr's orb turned-sphere. A sphere is useless from a chemical standpoint. Hybridization should not have to occur to explain the simplest of molecules. Simplicity would have the electrons occupying orbital spaces that are similar in shape. Only three orbital types are actually needed to describe the electron packing of the elements. Octahedral, square-planar, and pyramidal coordination complexes of the transition elements follow logically without the need to hybridize. This brief paper describes a rational packing of electrons around a nucleus that ends up mimicking the familiar periodic table when compressed to similar behavior."
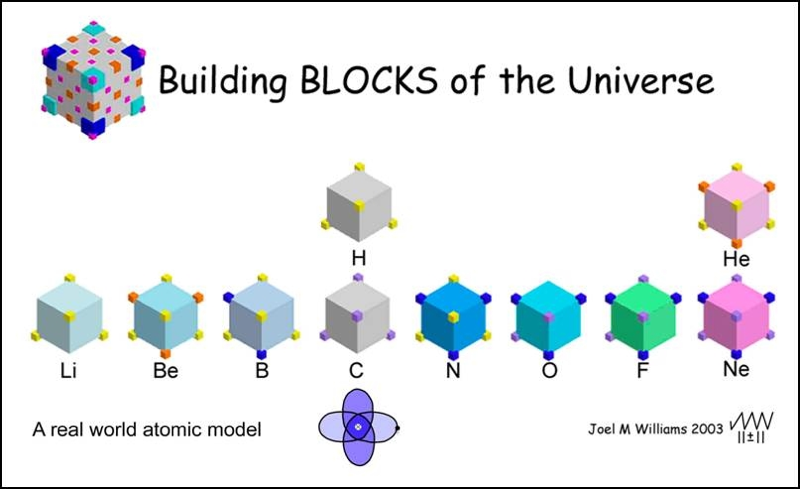
Modeling the MCAS Way describes this concept of "building blocks" and can be found here.
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
© Mark R. Leach Ph.D. 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com
This free, open access web book is an ongoing project and your input is appreciated.