Periodic Table |
 |
 |
 |
 |
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
The INTERNET Database of Periodic Tables
There are thousands of periodic tables in web space, but this is the only comprehensive database of periodic tables & periodic system formulations. If you know of an interesting periodic table that is missing, please contact the database curator: Mark R. Leach Ph.D. The database holds information on periodic tables, the discovery of the elements, the elucidation of atomic weights and the discovery of atomic structure (and much, much more).
| Year: 2014 | PT id = 664, Type = formulation |
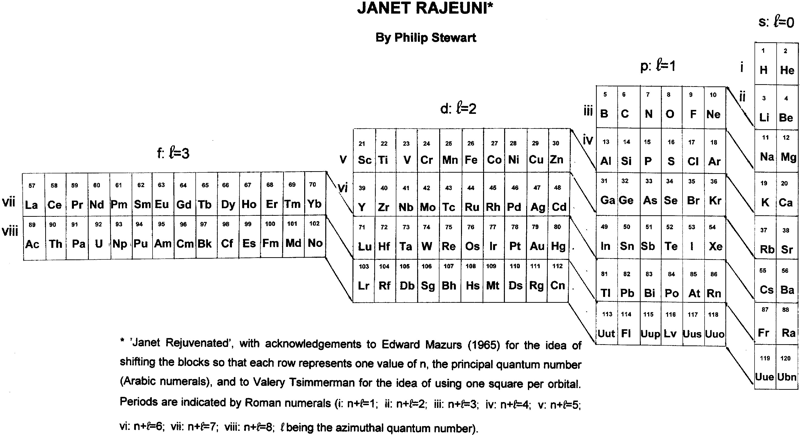
Janet Rajeuni
By Philip Stewart:
Janet Rejuvenated, with acknowledgement to Mazurs and to Valery Tsimmerman for the idea of using one square per orbital and of shifting the blocks so that each row represents one value of n, the principal quantum number.
The main objection people make to Janet is that He is placed at the head of the alkaline earth metals although it behaves as a noble gas. The essential answer is that electronic structure explains behaviour and not vice versa; like Ne (and unlike Ar, Kr, Xe and Rn), He has a complete shell. Similarly H, like C, is half way between a full and an empty shell, unlike the alkali metals and the halogens. I suggest a new argument: nobody finds it strange that the p block has a row of non-metals at its head (and that half its members are non-metals), so why not the s block?
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
© Mark R. Leach Ph.D. 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com
This free, open access web book is an ongoing project and your input is appreciated.