Periodic Table |
 |
 |
 |
 |
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
The INTERNET Database of Periodic Tables
There are thousands of periodic tables in web space, but this is the only comprehensive database of periodic tables & periodic system formulations. If you know of an interesting periodic table that is missing, please contact the database curator: Mark R. Leach Ph.D. The database holds information on periodic tables, the discovery of the elements, the elucidation of atomic weights and the discovery of atomic structure (and much, much more).
Periodic Tables from the year 1913:
| Year: 1913 | PT id = 13, Type = formulation element structure |
Moseley's Periodic Law and Atomic Number Z
Moseley, H. G. J. The High-Frequency Spectra of the Elements. Philosophical Magazine, 26, 1024–1034 (1913).
"Moseley's law is an empirical law concerning the characteristic X-rays emitted by atoms. The law was discovered and published by the English physicist Henry Moseley in 1913–1914. Until Moseley's work, "atomic number" was merely an element's place in the periodic table and was not known to be associated with any measurable physical quantity.
"In brief, Moseley's law states that the square root of the frequency, ν, of the emitted X-ray is (approximately) proportional to the atomic number":
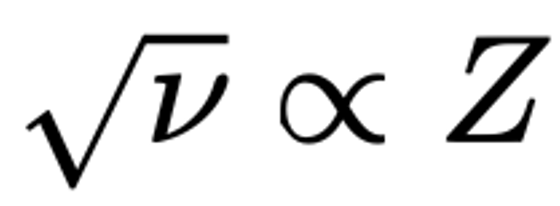
| Year: 1913 | PT id = 59, Type = formulation |
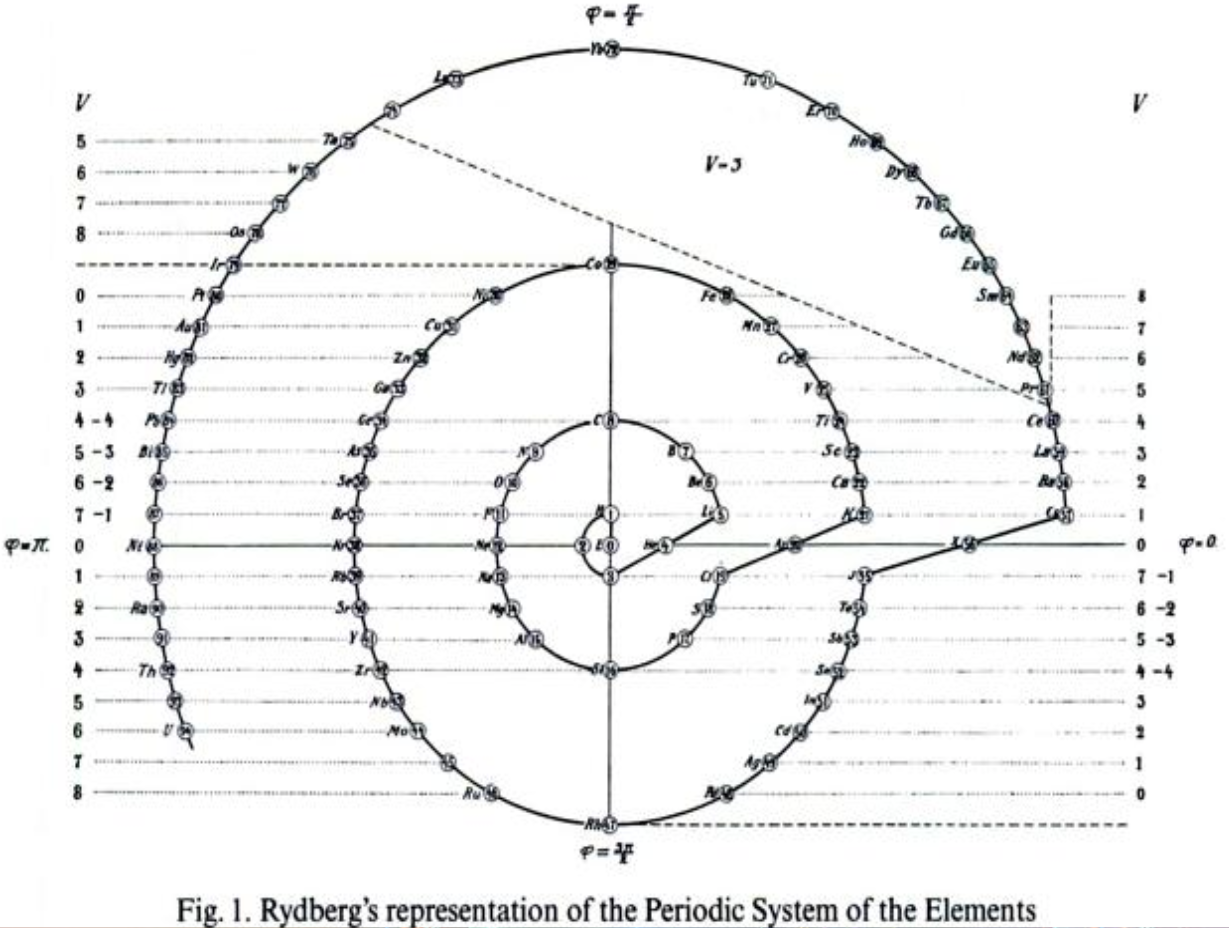
Rydberg's Table
René Vernon writes:
My source is the 1914 French translation of Rydberg’s 1913 German article.
- Rydberg 1913, Untersuchungen über das System der Grundstoffe, Lunds Univ. Årsskrift, (Acta Univers, Lundensis), vol. 9, no. 18, pp. 1-41
- — 1914, Recherches sur le système des éléments, Journal de Chimie Physique, vol. 12, pp. 585–639, https://doi.org/10.1051/jcp/1914120585

| Year: 1913 | PT id = 871, Type = element |
Discovery of Protactinium
Pa ![]()
Protactinium, atomic number 91, has a mass of 231.036 au.
Radioactive element: Pa is only found in tiny amounts in nature. Most samples are synthetic.
Protactinium was first observed or predicted in 1913 by O. H. Göhring and K. Fajans and first isolated in 1927 by A. von Grosse.
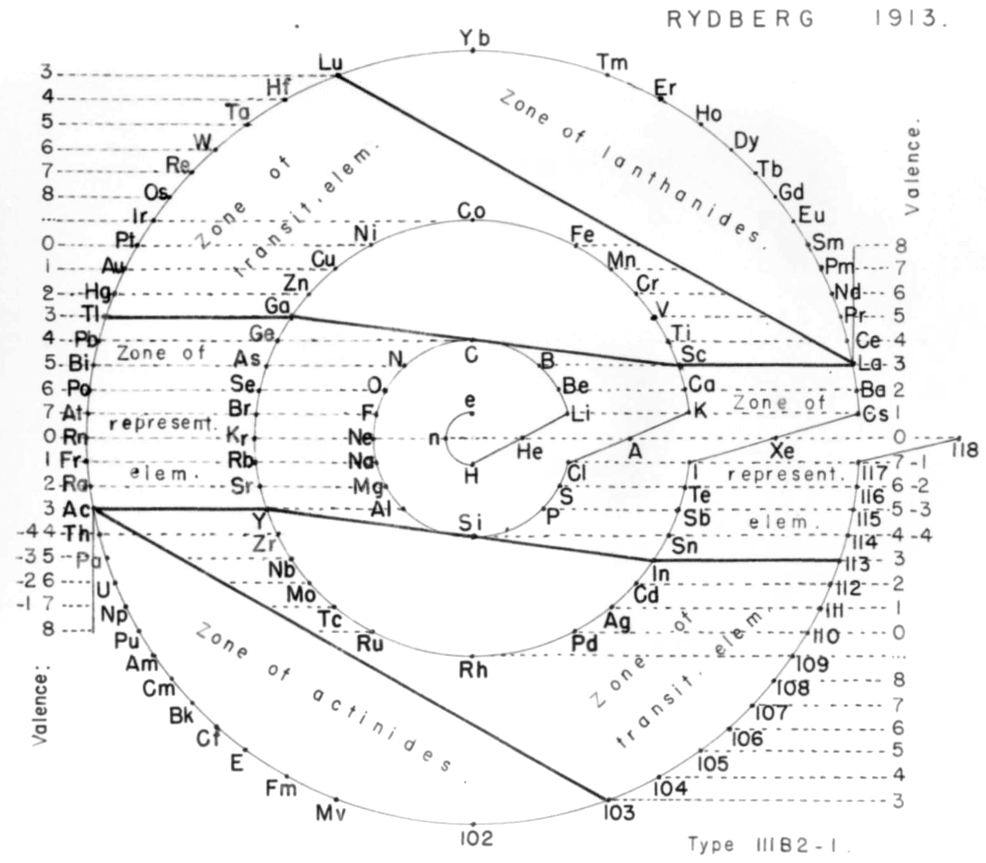
| Year: 1913 | PT id = 973, Type = formulation |
Rydberg's Periodic Table in style of Spiral with Four Revolutions
Periodic table in style of spiral with four revolutions circa 1913 (Original design) and 1957 (Date attributed to slide).
This table was originated by Swedish physicist Johannes Rydberg (1854-1919) in 1913 and classified by chemist Edward G. Mazurs as Type IIIB2-1 in his seminal work Types of Graphic Representation of the Periodic System of Chemical Elements (1957). The lower version of the table appears as Figure 63 on page 132 of Mazurs' 1957 publication.
| Year: 1913 | PT id = 1000, Type = formulation |
van den Broek's Periodic Table 3
From Wikipedia: Antonius Johannes van den Broek (1870-1926) was a Dutch amateur physicist notable for being the first who realized that the number of an element in the periodic table (now called atomic number) corresponds to the charge of its atomic nucleus. The 1911 inspired the experimental work of Henry Moseley, who found good experimental evidence for it by 1913. van den Broek envisaged the basic building block to be the 'alphon', which weighed twice as much as a hydrogen atom.
Read more in Chapter 4, Antonius Van Den Broek, Moseley and the Concept of Atomic Number by Eric Scerri. This chapter can be found in the book: For Science, King & Country: The Life and Legacy of Henry Moseley (Edited by Roy MacLeod, Russell G Egdell and Elizabeth Bruton).
van den Broek's periodic table of 1907: Annalen der Physik, 4 (23), (1907), 199-203
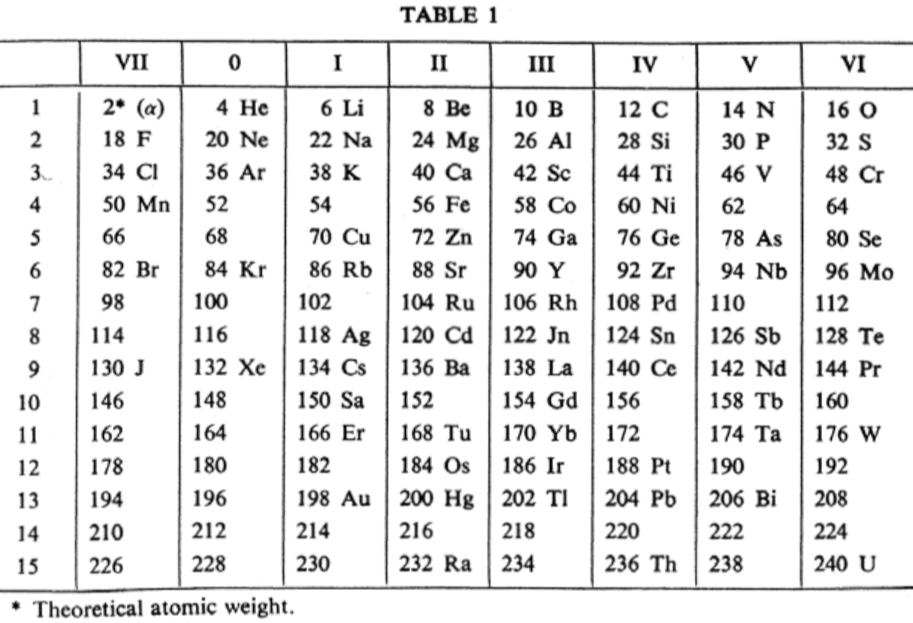
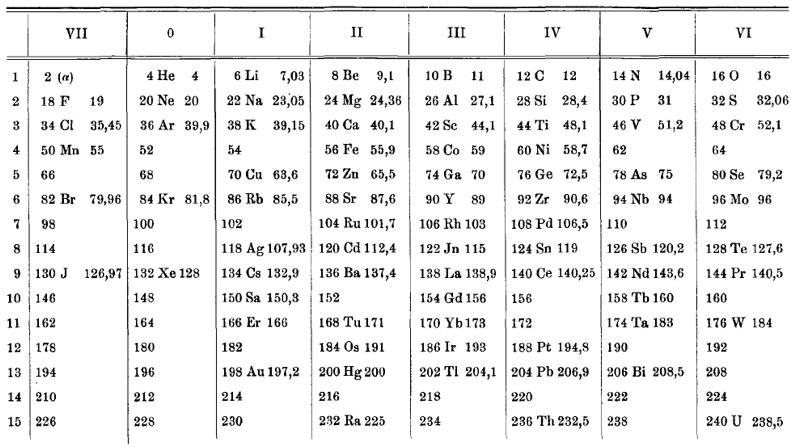
van den Broek's periodic table of 1911: Physikalische Zeitschrift, 12 (1911), 490-497); and also a paper in Nature the same year entitled: The Number of Possible Elements and Mendeléff's "Cubic" Periodic System, Nature volume 87, page 78 (20 July 1911)
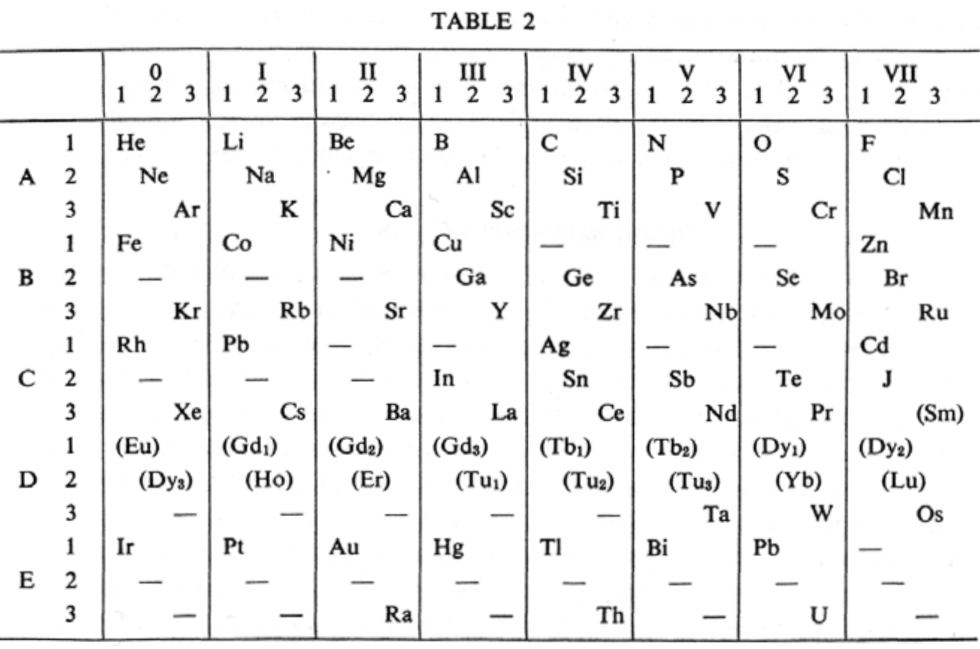
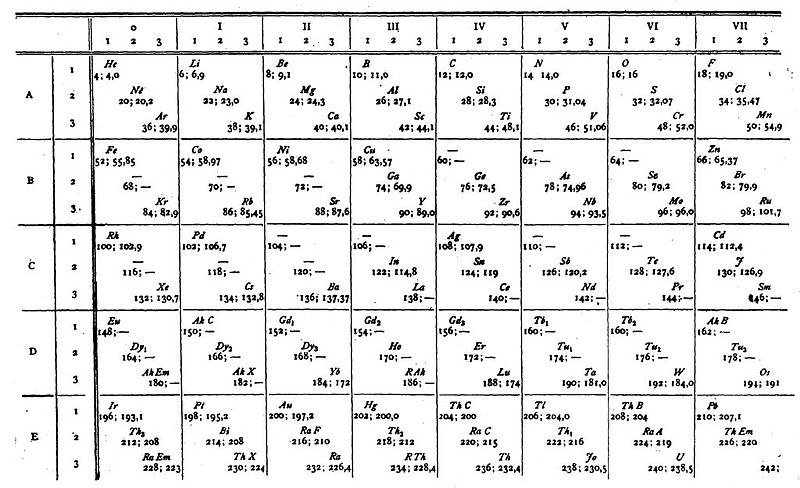
van den Broek's periodic table of 1913: Physikalische Zeitschrift, 14, (1913), 32-41
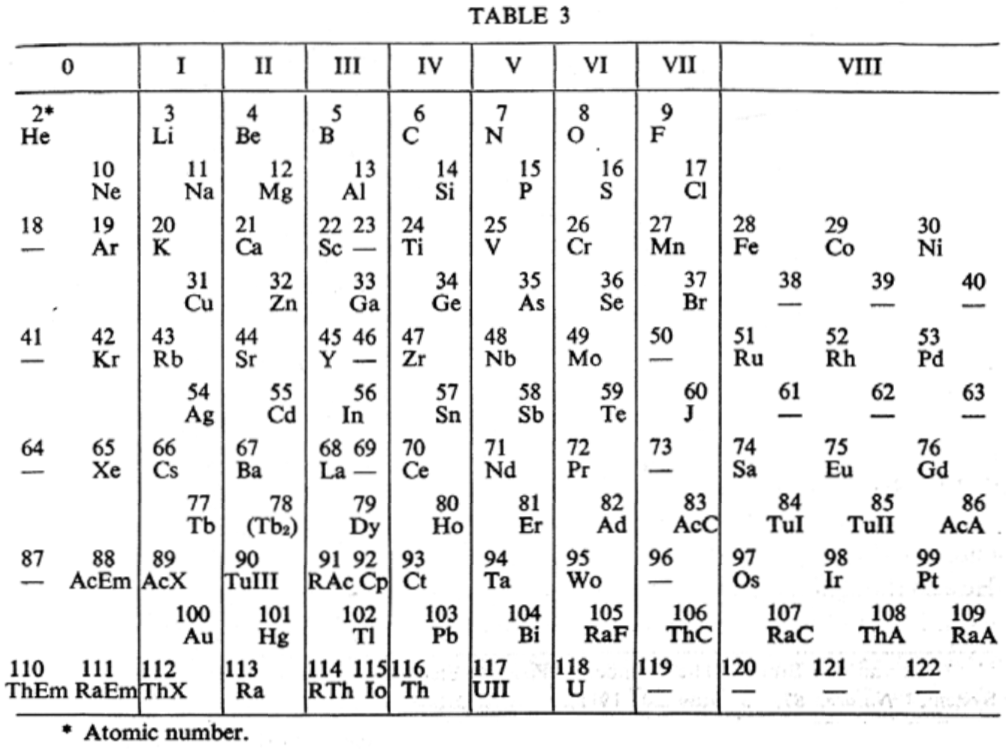

Thanks to Eric Scerri for the tip!
See the website EricScerri.com and Eric's Twitter Feed.
| Year: 1913 | PT id = 1370, Type = structure |
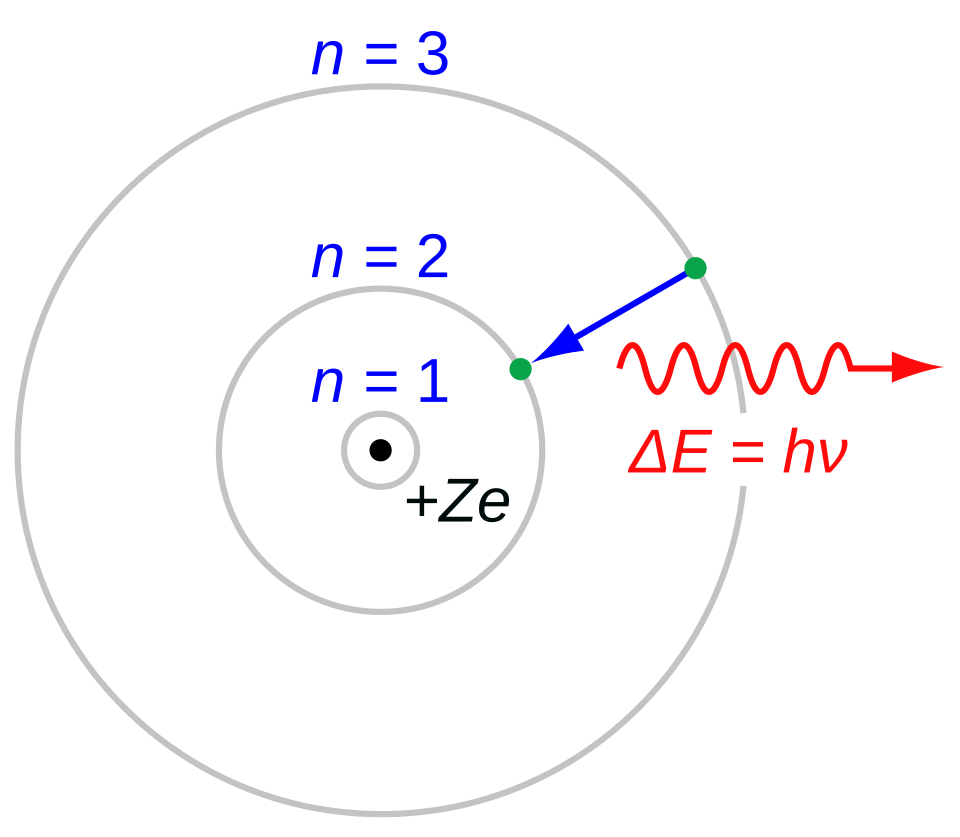
The Bohr Atom
Bohr, N. On the Constitution of Atoms and Molecules (Parts I–III). Philosophical Magazine, 26, 1–25; 476–502; 857–875 (1913).
"In the Bohr model (or the Rutherford–Bohr model) of the hydrogen atom (Z = 1), the negatively charged electron confined to an atomic shell encircles a small, positively charged atomic nucleus. When an electron jumps between orbits, is accompanied by an emitted or absorbed amount of electromagnetic energy (where E = hν). The orbits in which the electron may travel are shown as grey circles; their radius increases as n2, where n is the principal quantum number. The 3 ? 2 transition produces the first line of the Balmer series, and for hydrogen (Z = 1) it results in a photon of wavelength 656 nm (red light).
"The Bohr atom consists of a small, dense atomic nucleus surrounded by orbiting electrons. It is analogous to the structure of the Solar System, but with attraction provided by electrostatic force rather than gravity, and with the electron energies quantised (assuming only discrete values). The Bohr model incorporated some early quantum concepts. Developed from 1911 to 1918 by Niels Bohr and building on Ernest Rutherford's discovery of the atom's nucleus, the model supplanted the plum pudding model of J. J. Thomson only to be replaced by the quantum atomic model in the 1920s.
"The Bohr model's key success lies in explaining the Rydberg formula for hydrogen's spectral emission lines. While the Rydberg formula had been known experimentally, it did not gain a theoretical basis until the Bohr model was introduced. Not only did the Bohr model explain the reasons for the structure of the Rydberg formula, it also provided a justification for the fundamental physical constants that make up the formula's empirical results."
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
© Mark R. Leach Ph.D. 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com
This free, open access web book is an ongoing project and your input is appreciated.