Periodic Table |
 |
 |
 |
 |
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
The INTERNET Database of Periodic Tables
There are thousands of periodic tables in web space, but this is the only comprehensive database of periodic tables & periodic system formulations. If you know of an interesting periodic table that is missing, please contact the database curator: Mark R. Leach Ph.D. The database holds information on periodic tables, the discovery of the elements, the elucidation of atomic weights and the discovery of atomic structure (and much, much more).
Periodic Tables from the year 1956:
| Year: 1956 | PT id = 976, Type = formulation |
Remy's Long Period Form Periodic Table
From H. Remy's 1956, Treatise on Inorganic Chemistry, Vol. 1, (Introduction and main groups of the periodic table), Elsevier, Amsterdam, p. 4, is what Remy calls a "Long-Period Form of the Natural System of the Elements".
This is a semi-lanthanide/actinide formulation, with Th-Pa-U shown as 6d metals, and the remaining actinides (Np, etc.) shown as transuranic counterparts to Pm, etc. The layout of Remy's table was based on ideas by Haissinsky in competition with Seaborg's formulation of 1945.
In the appendix there is a second "Table II" version of this formulation with shorter periods.
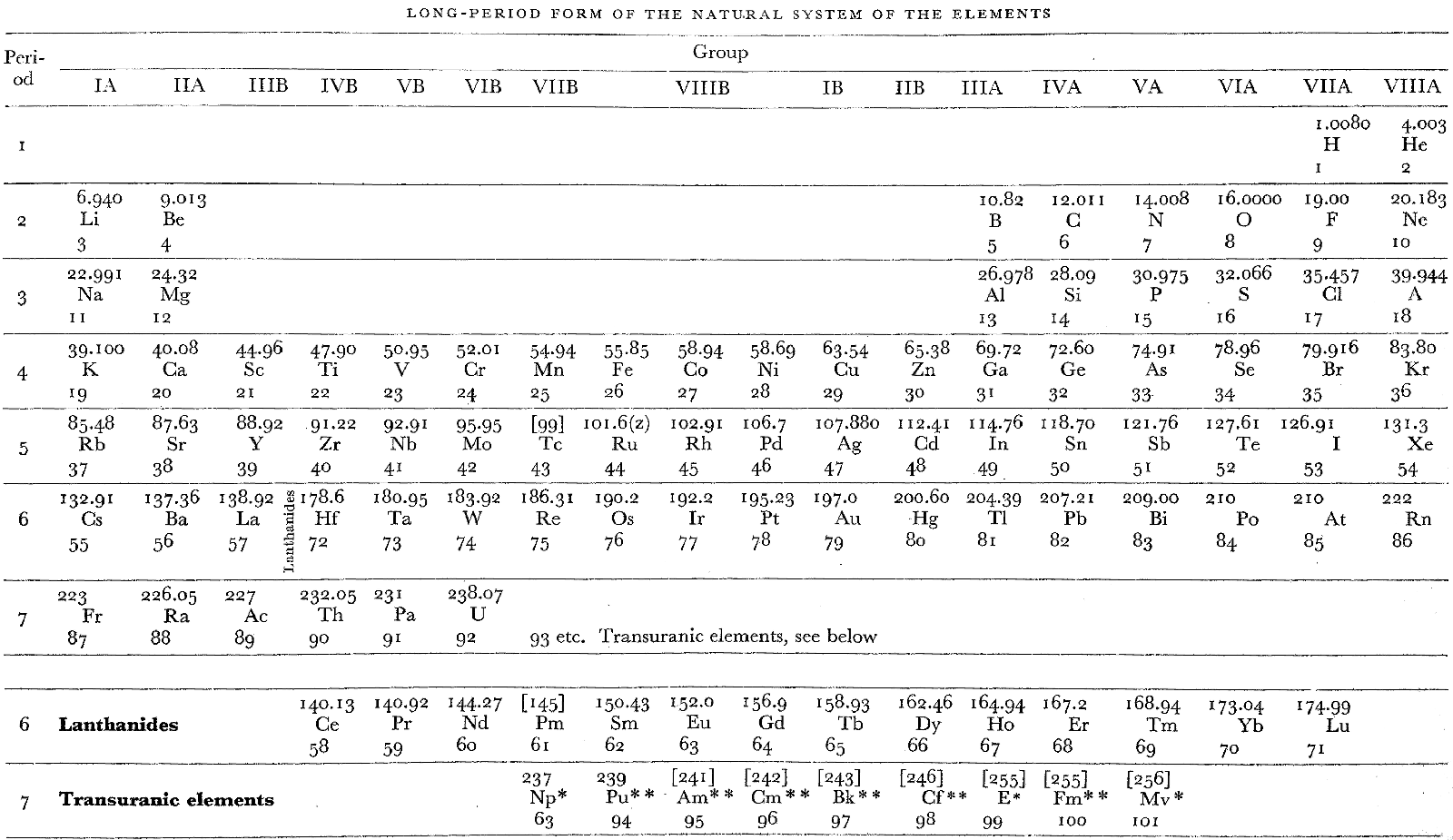
Thanks to René for the tip!
| Year: 1956 | PT id = 1009, Type = formulation |
Walker & Curthoys' New periodic Table Based of Stability of Atomic Orbitals
By W. R. Walker and G. C. Curthoys, A new periodic table based on the energy sequence of atomic orbitals, J. Chem. Educ., 1956, 33 (2), p 69.
The abstract states:
"Since the theory of atomic and molecular orbitals has proven to be of such value in interpreting the data of inorganic chemistry, it is hoped that a new periodic table based on the energy sequence of atomic orbitals will be an aid to the further systematizing of chemical knowledge."
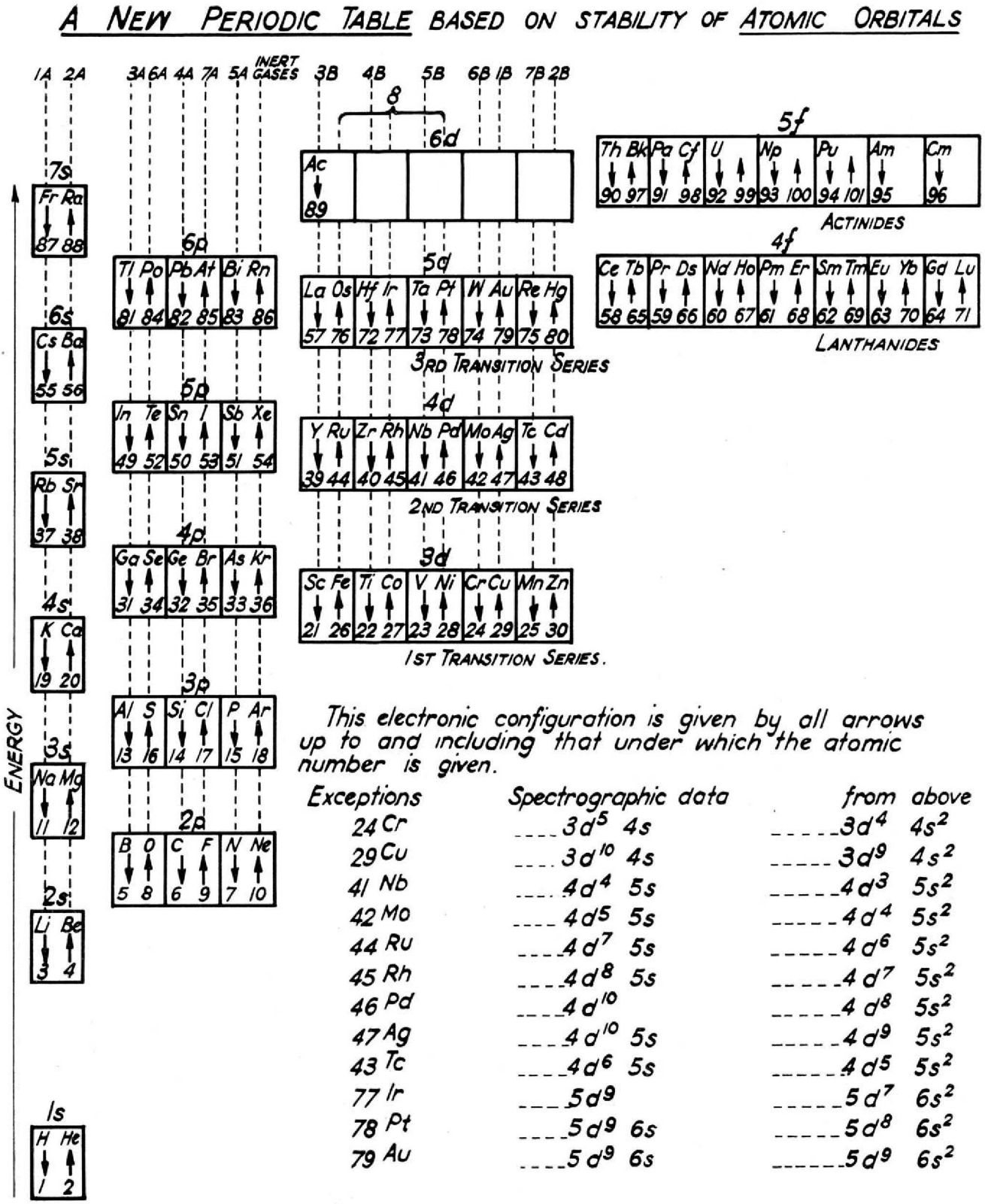
Thanks to René for the tip!
| Year: 1956 | PT id = 1216, Type = formulation |
Sistema Periodico de Los Elementos (after Antropoff)
Mario Rodríguez Peña, PhD translates the spanish text on the Archive.org website:
"Periodic System of Elements, type Antropoff., 1956 Antropoff's periodic table was designed in Bonn (Germany) in 1926: https://www.meta-synthesis.com/webbook/35_pt/pt_database.php?PT_id=26 It was disused after the WWII (1945) in most of the countries, except Spain. This was dated in 1956 because Mendelevium (101) was discovered and accepted by IUPAC in 1955 and in 1957, the element symbols of Argon (18), Xenon (54), Einstenium (99) and Mendelevium itself changed to the current Ar, Xe, Es and Md, respectively."
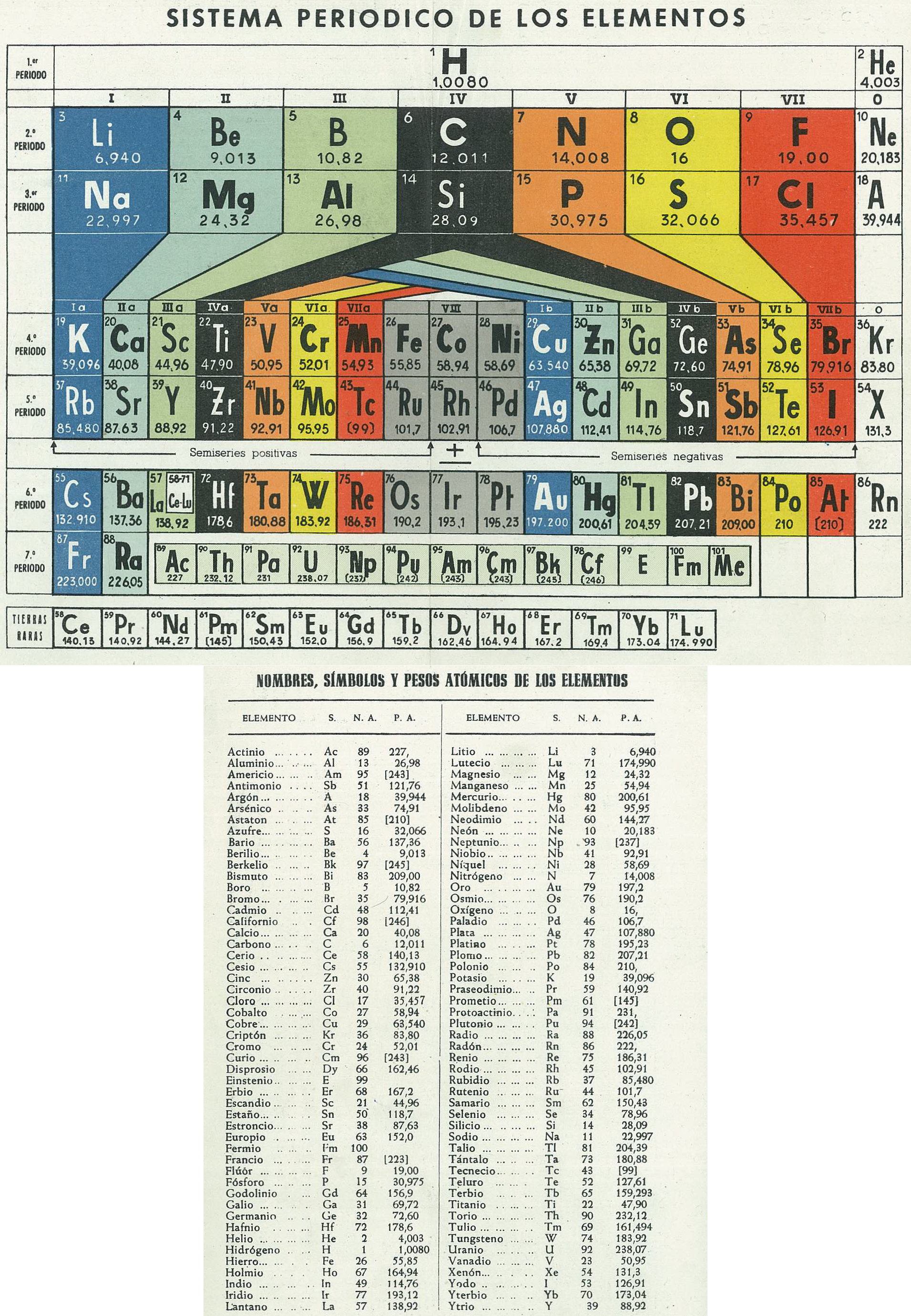
| Year: 1956 | PT id = 1227, Type = formulation |
Remy's Periodic Table II: The Short Period Presentation
Next to Remy's Long Form Periodic Table (H. Remy, Treatise on Inorganic Chemistry, Vol. 1, Introduction and main groups of the periodic table, Elsevier, Amsterdam, 1956, p. 4) is what Remy calls a "Short Period presentation" shown in the appendix, pages 838-939. The author comments:
"The form of presentation used in Table II in which the elements of the Long Periods are divided into two series, so that the short Periods determine the horizontal breadth of the system, is known as the Short Period presentation, as contrasted with the Long Period presentation in which the elements of the Long Periods are each time included in a single series.
"The Short Periods can then be broken up accordingly. Mendeléeff had already used the short-periodic and long-periodic mode of tabulation. The adjacent Table I sets it out as a form which is based directly on that already used by Mendeléeff, but completed by the insertion of the elements discovered subsequently."
Thanks to Mark Winter of WebElements for the tip!
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
© Mark R. Leach Ph.D. 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com
This free, open access web book is an ongoing project and your input is appreciated.
