Periodic Table |
 |
 |
 |
 |
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
The INTERNET Database of Periodic Tables
There are thousands of periodic tables in web space, but this is the only comprehensive database of periodic tables & periodic system formulations. If you know of an interesting periodic table that is missing, please contact the database curator: Mark R. Leach Ph.D. The database holds information on periodic tables, the discovery of the elements, the elucidation of atomic weights and the discovery of atomic structure (and much, much more).
Periodic Tables from the year 1902 :
| 1902 | Brauner's Table |
| 1902 | Erdmann's Spiral Table |
| 1902 | Blitz's Periodensystem der Elemente |
| 1902 | Actinium, Discovery of |
| 1902 | Armstrong's Classification of The Elements |
| Year: 1902 | PT id = 58, Type = formulation |
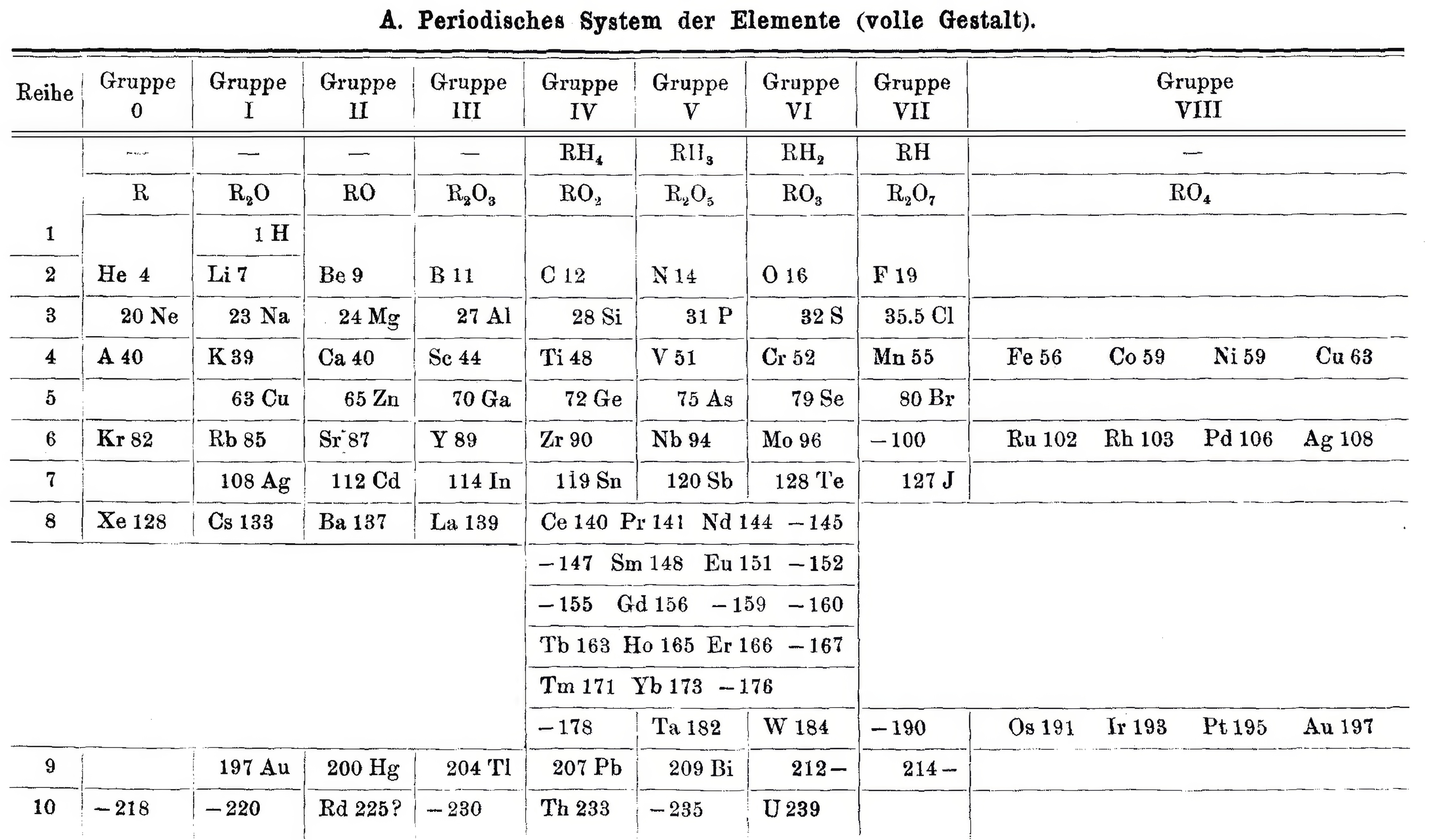
Brauner's Table
From Quam & Quam's 1934 review paper.pdf:
This table is practically identical with the Mendelceff short table, except that new elemeuts, atomic numbers, group zero, and more exact atomic weights are introduced. Through a thorough study of the rare-earth elements, Brauner concluded that all should be placed in a miniature table following La and preceding the space now occupied by Hf.
| Year: 1902 | PT id = 71, Type = formulation spiral |
Erdmann's Spiral Table

From Quam & Quam's 1934 review paper.pdf
| Year: 1902 | PT id = 365, Type = formulation |
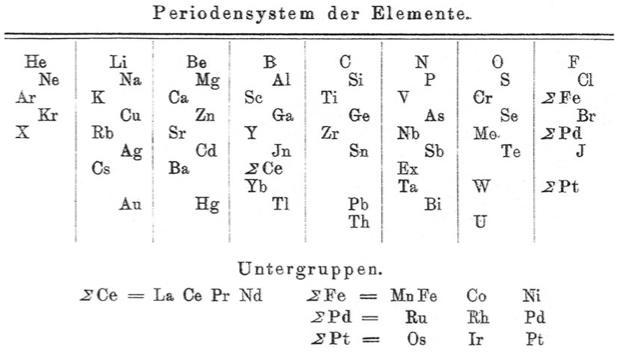
Blitz's Periodensystem der Elemente
Periodic Table of Biltz (1902) with an intraperiodic accommodation of the rare earths. Reproduced from Biltz, H., 1902. Ber. 35 (562), 4242:
| Year: 1902 | PT id = 869, Type = element |
Discovery of Actinium
Ac ![]()
Actinium, atomic number 89, has a mass of 227 au.
Radioactive element.
Actinium was first isolated in 1902 by F. O. Giesel.
| Year: 1902 | PT id = 1403, Type = formulation |
Armstrong's Classification of The Elements
Henry Edward Armstrong; The classification of the elements. Proc. R. Soc. 30 September 1902; 70 (459-466): 86–94. https://doi.org/10.1098/rspl.1902.0012
Abstract:
Although no direct evidence acceptable to chemists has been adduced which in any way justifies the belief that the elements are decomposible, it is impossible to resist the conclusion that they are genetically related, so closely in many respects do they resemble a series of related compounds, especially when regarded from the point of view of the organic chemist. The generalisation known as the Periodic Law is in itself a justification of this view: the manner in which interrelationship becomes manifest when they are classified in accordance with its canons, being probably the strongest of all the arguments which can be cited as tending to show that the elements are compounds—but compounds very different from those with which we are accustomed to deal. Even in the form in which it was put forward by Mendeleeff, however, the periodic generalisation is but a first approximation: and the great Russian has himself pointed out that it needs improvement and development. As chemists are beginning to recognise this, I venture to submit a scheme of classification which I have been led to draw up in writing an article for the forthcoming Supplement to the ‘Encyclopedia Britannica.’ The article, I may say, was sent to press in May, 1900 and the first proof before me is dated November 20, 1900.

 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
© Mark R. Leach Ph.D. 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com
This free, open access web book is an ongoing project and your input is appreciated.