Periodic Table |
 |
 |
 |
 |
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
The INTERNET Database of Periodic Tables
There are thousands of periodic tables in web space, but this is the only comprehensive database of periodic tables & periodic system formulations. If you know of an interesting periodic table that is missing, please contact the database curator: Mark R. Leach Ph.D. The database holds information on periodic tables, the discovery of the elements, the elucidation of atomic weights and the discovery of atomic structure (and much, much more).
Periodic Tables from the year 1916 :
| 1916 | Harkins & Hall's Periodic Table |
| 1916 | Dushman's Periodic System of The Elements (after Mendelejeff) |
| 1916 | Sommerfeld's Periodic Table |
| 1916 | Sommerfeld and the Azimuthal and Magnetic Quantum Numbers |
| Year: 1916 | PT id = 77, Type = formulation 3D spiral |
Harkins & Hall's Periodic Table
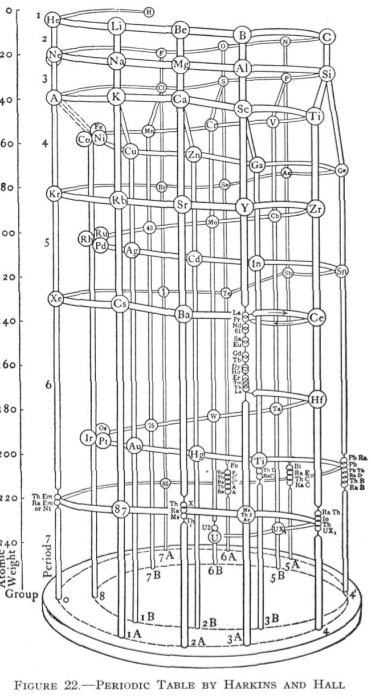
From Quam & Quam's 1934 review paper.pdf
| Year: 1916 | PT id = 541, Type = formulation |
Dushman's Periodic System of The Elements (after Mendelejeff)
Saul Dushman's article, "The Periodic Law", published in the General Electric Review (Vol. 18, pp. 614–621) in July 1915, provides a comprehensive overview of the status of the periodic law in the early 20th century.
Key Aspects of the Article:
- Historical Context: It reviews the development of the periodic classification, honouring the foundational work of Dmitri Mendeleev.
- Modernizing the Law: Writing just over a decade after the discovery of radioactivity and the electron, Dushman bridges the gap between Mendeleev’s empirical atomic weight law and emerging quantum-physical understandings of atomic structure.
- Radioactivity and Isotopes: The article discusses the impact of radioactivity research (including Soddy's work on isotopes) on the periodic table, explaining how many "new" radioactive elements were found to belong to the same place in the table.
- Atomic Number: It touches upon the significance of atomic numbers as a more fundamental ordering principle than atomic weight, a crucial transition in understanding atomic structure.
- Significance: It highlights the periodic law as the "most fundamental natural system of classification ever devised", marking a transition from chemical phenomenology to physical explanation.
The article is representative of the era's shift toward interpreting chemistry through atomic structure and the revolutionary discoveries of Moseley and Rutherford.

Thanks to Eric Scerri for the tip!
See the website EricScerri.com and Eric's Twitter Feed.
| Year: 1916 | PT id = 1214, Type = formulation |
Sommerfeld's Periodic Table
A periodic table by Arnold Sommerfeld, as an updated construction by Marks & Marks (2021).
John Marks writes:
"The reconstruction of Sommerfeld 1916 is derived from my reading of Henry Browse's translation of the third German edition of his Atomstruktur und Spektrallinien (Methuen 1923). Sommerfeld found the explanation of the greater (d– and f–) and lesser (s– and p–) periods in the solution of Kepler's ellipses using Schwarzschild's relativistic correction, communicated to him from the battlefront of WW1. Sommerfeld considered helium "an exception" but this is only an appearance deriving from defining periods as terminated by inert gases. In fact, the first period begins with hydrogen so the markers of periods are analogues of hydrogen, viz. the halogens."
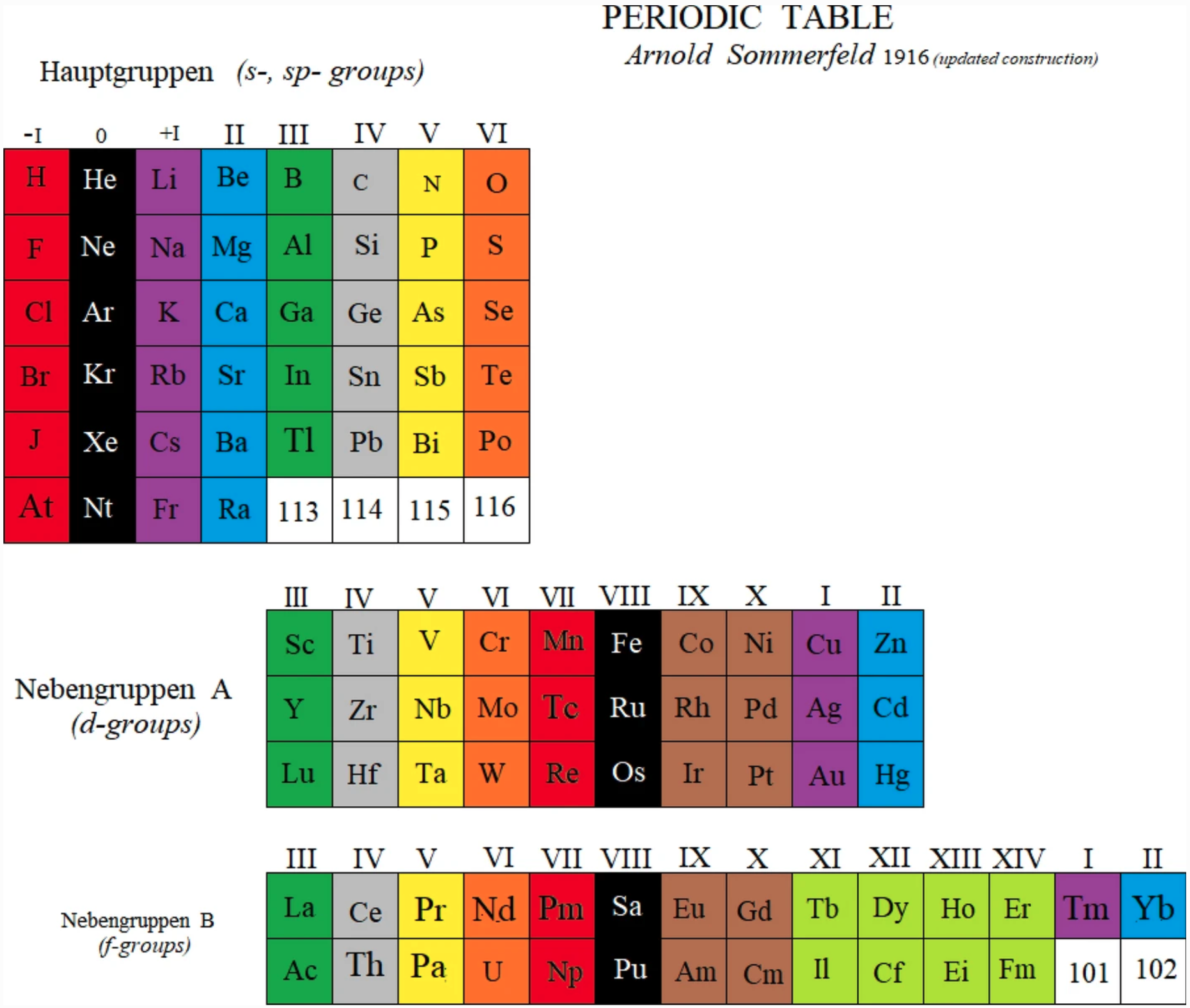
| Year: 1916 | PT id = 1371, Type = structure |
Sommerfeld and the Azimuthal and Magnetic Quantum Numbers
Sommerfeld, A. Zur Quantentheorie der Spektrallinien. Annalen der Physik, 51, 1–94 (1916).
"Arnold Sommerfeld was a German theoretical physicist who pioneered developments in both atomic and quantum physics. He also educated and mentored many students for the new era of theoretical quantum physics.
"Sommerfeld introduced the second (azimuthal) and the third (magnetic) quantum numbers ℓ and mℓ. (He also introduced the fine-structure constant and pioneered X-ray wave theory.)
"In quantum mechanics, the azimuthal quantum number ℓ is a quantum number for an atomic orbital that determines its orbital angular momentum and describes aspects of the angular shape of the orbital. The azimuthal quantum number is the second of a set of quantum numbers that describe the unique quantum state of an electron (the others being the principal quantum number n, the magnetic quantum number, mℓ, and the spin quantum number ms).
"Seven of Sommerfeld's doctoral students and postdoctoral supervisees: Werner Heisenberg, Wolfgang Pauli, Peter Debye, Hans Bethe, Linus Pauling, Isidor I. Rabi and Max von Laue went on to win Nobel Prizes in theoretical physics or chemistry. He also supervised at least 30 other famous physicists and chemists."

 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
© Mark R. Leach Ph.D. 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com
This free, open access web book is an ongoing project and your input is appreciated.