Periodic Table |
 |
 |
 |
 |
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
The INTERNET Database of Periodic Tables
There are thousands of periodic tables in web space, but this is the only comprehensive database of periodic tables & periodic system formulations. If you know of an interesting periodic table that is missing, please contact the database curator: Mark R. Leach Ph.D. The database holds information on periodic tables, the discovery of the elements, the elucidation of atomic weights and the discovery of atomic structure (and much, much more).
Periodic Tables from the year 1918 :
| 1918 | Hackh's Classification of the Elements |
| 1918 | Meyer's (Stephan) Periodisches System der Elemente |
| 1918 | Cherkesov: Two Periodic Tables |
| 1918 | One of Mendelejeff's Tables, Modified |
| Year: 1918 | PT id = 83, Type = formulation |
Hackh's Classification of the Elements
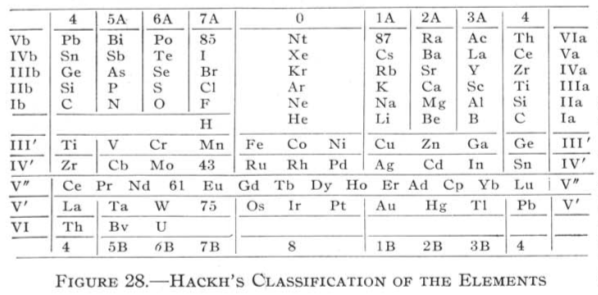
From Quam & Quam's 1934 review paper.pdf
| Year: 1918 | PT id = 367, Type = formulation |
Meyer's Periodisches System der Elemente
Periodic Table of Meyer (1918) with an intraperiodic accommodation of the rare earths. Reproduced from Meyer, S., 1918. Phys. Z. 19, 178.
Philip Stewart has provided a bit more detail:
Stefan Meyer (1872-1949) was an Austrian physicist, no relation of Julius Lothar Meyer. He had a special interest in 'rare earth' and radioactive elements. He published several versions of the periodic table. In this definitive version of 1918, note elements 69-72. Tu I is 'thulium I', Ad is Aldeberanium (Yb), Cp is Cassiopeium (Lu) and Tu II is 'thulium II' (Hf).:
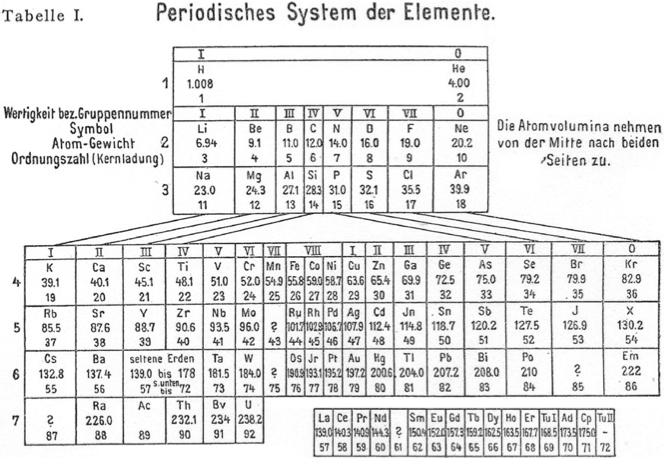
| Year: 1918 | PT id = 1260, Type = formulation |
Cherkesov: Two Periodic Tables
von Bichowsky FR, The place of manganese in the periodic system, J. Am. Chem. Soc. 1918, 40, 7, 1040–1046 Publication Date: July 1, 1918 https://doi.org/10.1021/ja02240a008
René Vernon writes:
"In this curious article, von Bichowsky, a physical chemist (1889-1951), mounted an argument for regarding Mn as belonging to group 8 (see table 1 below) rather than group 7 (table 2). His article has effectively been assigned to the dustbin of history, having apparently gathered zero citations over the past 103 years.
"Items of note in his 24-column table:
- While Mn, 43 and 75 are assigned to group 8 they remain in alignment with group 7. Se is shown as Sc
- 14 lanthanides, from Ce to Yb, make up group 3a; If La and Lu are included, there are 16 Ln
- Gd is shown as Cd
- Positions of Dy and Ho have been reversed
- Tm and Tm2
- Po shown as "RaF"
- Ra shown as "RaEm"
- Pa shown as Ux2
von Bichowsky made his argument for Mn in group 8, on the following grounds:
- by removing the Ln from the main body of the table all of the gaps denoted by the dashes (in table 2) were removed
- the eighth group links Cr with Cu; Mo with Ag; and W with Au
- the symmetry of the table is greatly increased
- the triads are replaced by tetrads and a group of 16 Ln which accords better with "the preference of the periodic system for powers of two"
- about eight chemistry-based differences between Ti-V-Cr and Mn, including where Mn shows more similarities to Fe-Co-Ni, for example:
- divalent Ti, V, Cr cations are all powerful reducing agents, Cr being one of the most powerful known; divalent Mn, Fe, Co, Ni are either very mild reducing agents as divalent Mn or Fe, or have almost no reducing power in the case of divalent Co or Ni;
- metal titanates, vanadates and chromates are stable in alkaline solution and are unstable in the presence of acid whereas permanganates are more stable in acid than alkali; their oxidizing power is also widely different.
I can further add:
- Mn, Fe, and Co, and to some extent Ni, occupy the "hydrogen gap" among the 3d metals, having no or little proclivity for binary hydride formation
- the +2 and +3 oxidation states predominate among the Mn-Fe-Co-Ni tetrad (+3 not so much for Mn)
- in old chemistry, Mn, Fe, Co, and Ni represented the "iron group" whereas Cr, Mo, W, and U belonged to the "chromium group": Struthers J 1893, Chemistry and physics: A manual for students and practitioners, Lea Brothers & Co., Philadelphia, pp. 79, 123
- Tc forms a continuous series of solid solutions with Re, Ru, and Os
Moving forward precisely 100 years, Rayner-Canham (2018) made the following observations:
- Conventional classification systems for the transition metals each have one flaw: "They organise the TM largely according to one strategy and they define the trends according to that organisation. Thus, linkages, relationships, patterns, or similarities outside of that framework are ignored."
- There are two oxide series of the form MnO and Mn3O4 which encompass Mn through Ni. Here the division is not clear cut since there are also the series Mn2O3 for Ti-Cr and Fe; and MnO2 for Ti to Cr.
- Under normal condition of aqueous chemistry, Mn favours the +2 state and its species match well with those of the following 3d member, Fe.
Rayner-Canham G 2018, "Organizing the transition metals" [a chapter in] in E Scerri & G Restrepo, Mendeleev to Oganesson: A multidisciplinary perspective on the periodic table, Oxford University Press, Oxford, pp. 195–205
I've also attached a modern interpretation of von Bichowsky’s table. It's curious how there are eight metals (Fe aside) capable of, or thought to be capable of, achieving +8. I am not sure that a table of this kind with Lu in group 3 is possible, without upsetting its symmetry."
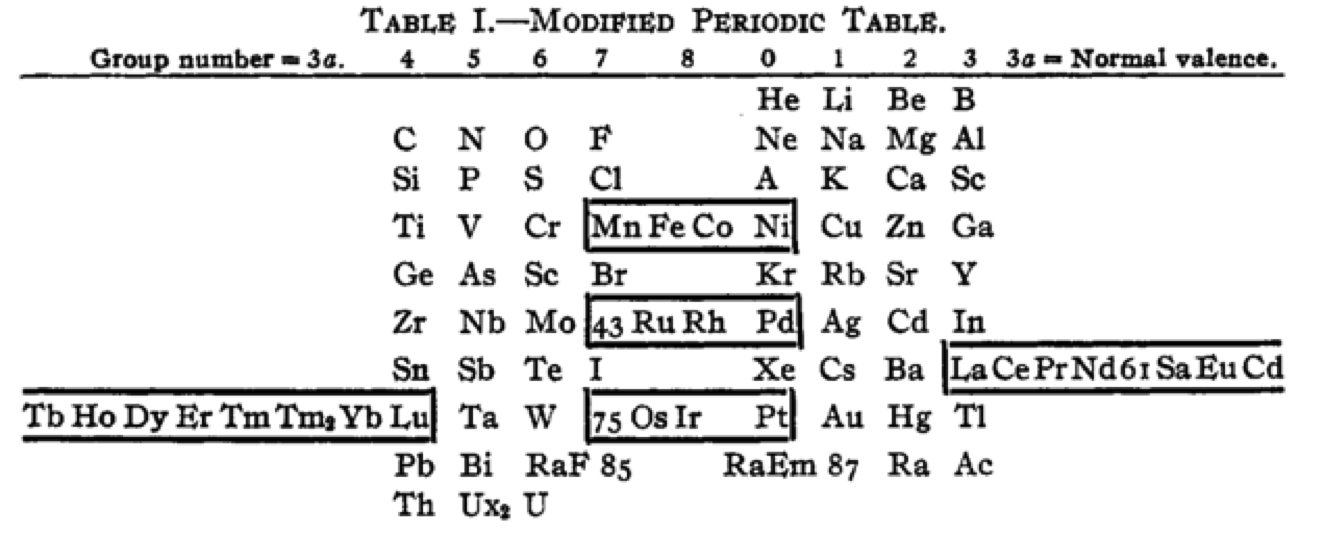


| Year: 1918 | PT id = 1300, Type = formulation |
One of Mendelejeff's Tables, Modified
From Smith A 1918, General Chemistry for Colleges, 2nd ed., The Century Co., New York, p. 299
René Vernon writes:
- H is missing, as are the noble gases.
- Consequently, the period numbers are out by one apiece.
- Seven groups are on the left and seven are on the right (the ever present allure of symmetry).
- After La, Ce is placed under Zr, and Nd is placed under columbium/technetium.
- According to Smith the rest of the lanthanide elements do not fit into any series, because their valences and other chemical properties do not permit most of them to be distributed over so many different groups.
- Po is expected to be a metal which is what it turned out to be Smith has anticipated that astatine will be a metal. Nine decades later, Hermann, Hoffmann & Ashcroft (2013) predicted the same thing: Hermann, A.; Hoffmann, R.; Ashcroft, N. W. (2013). Condensed astatine: Monatomic and metallic. Physical Review Letters, 111 (11), 116404-1–116404-5
- While he does not discuss it, Smith appears to have allowed for missing elements between Li and Gl and between Na and Mg.
- The three elements inside square brackets are those predicted by Mendeleev.
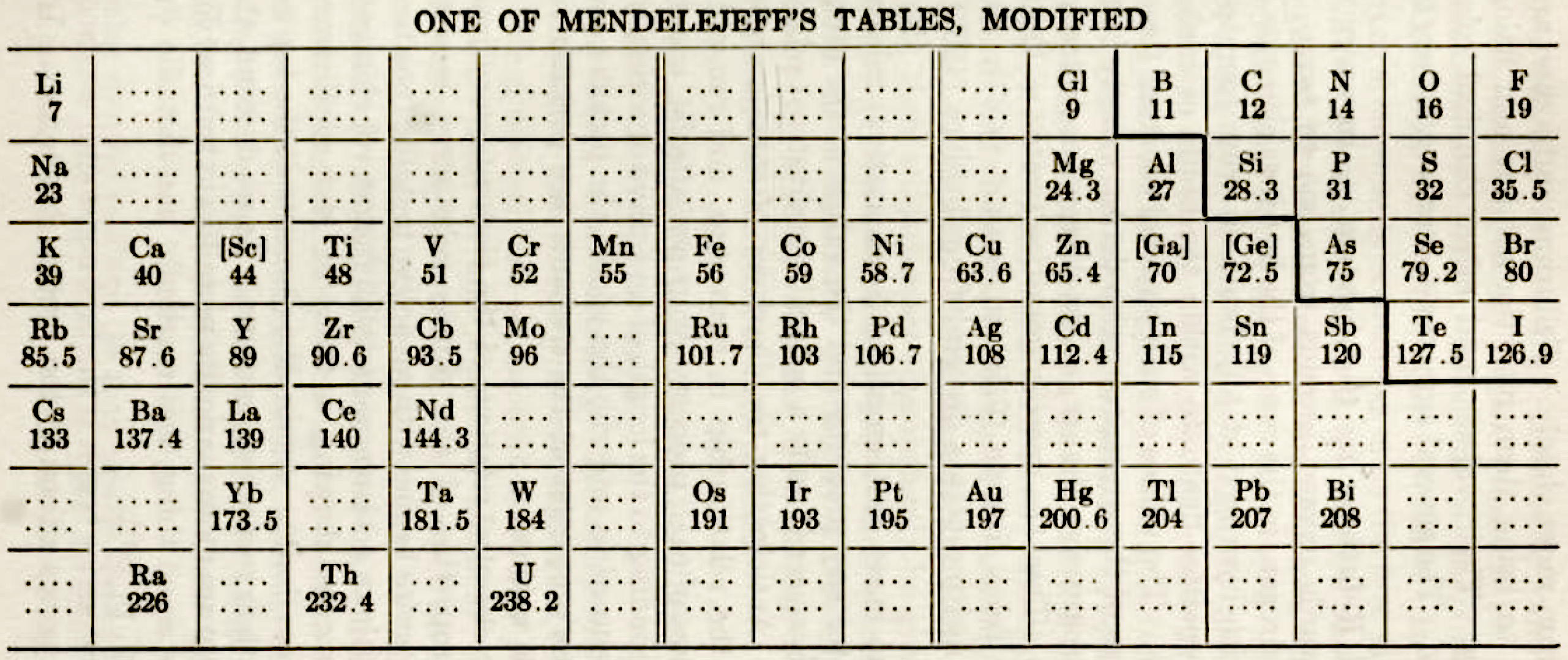
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
© Mark R. Leach Ph.D. 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com
This free, open access web book is an ongoing project and your input is appreciated.