Periodic Table |
 |
 |
 |
 |
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
The INTERNET Database of Periodic Tables
There are thousands of periodic tables in web space, but this is the only comprehensive database of periodic tables & periodic system formulations. If you know of an interesting periodic table that is missing, please contact the database curator: Mark R. Leach Ph.D. The database holds information on periodic tables, the discovery of the elements, the elucidation of atomic weights and the discovery of atomic structure (and much, much more).
Periodic Tables from the year 1930 :
| Year: 1930 | PT id = 154, Type = formulation spiral |
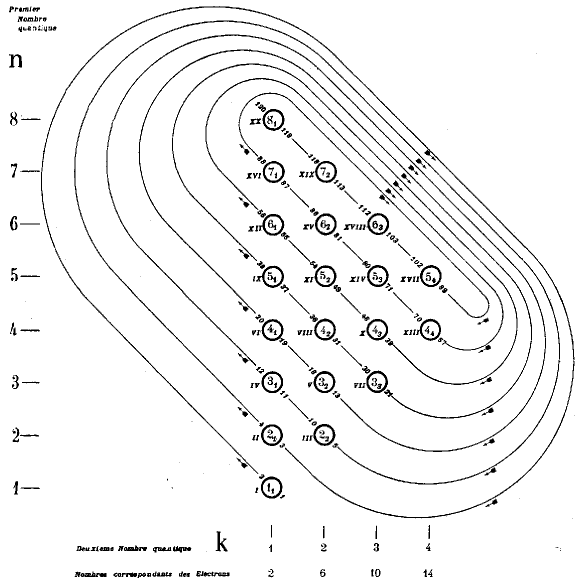
Janet's Shell Filling Diagram
Janet produced six papers, in French, which are almost unobtainable as he had them privately printed and didn't distribute them properly. The shell-filling diagram dated from November 1930, six years before Madelung. Note that Janet uses Bohr's radial quantum number, k, which is l+1. In the text he formulates the n+k-1 rule. Information supplied by Philip Stewart.
| Year: 1930 | PT id = 696, Type = formulation |
Gardner & Mazzucchelli's Periodic System Elaborated as Electronic Configuration
From Edward G. Mazurs' 1974 (2nd edition) Graphic Representations of the Periodic System During One Hundred Years, University of Alabama Press:
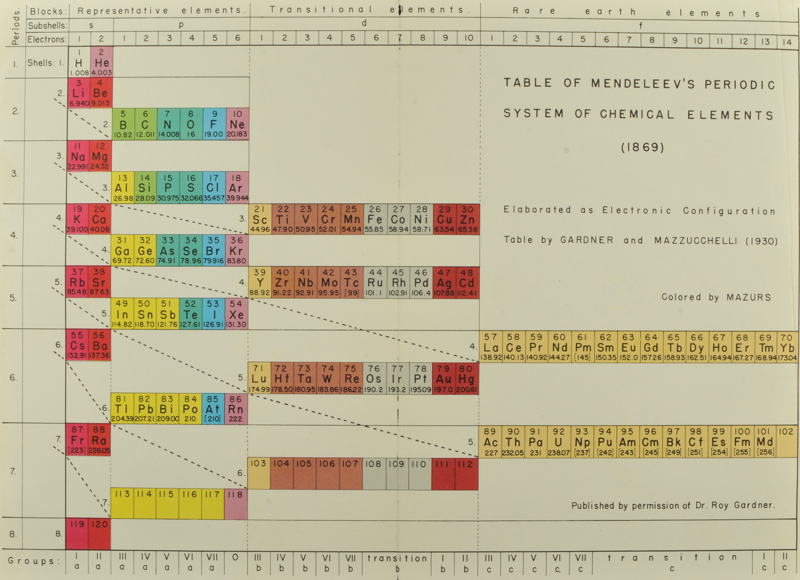
Thanks to Philip Stewart for the tip!
| Year: 1930 | PT id = 1264, Type = formulation |
Gardner's Table of Electronic Configurations of the Elements
A table of electronic configurations of the elements. Nature 125, 146 (1930). https://doi.org/10.1038/125146a0
Abstract:
"MR. ROY GARDNER gave an interesting paper on A Method of Setting out the Classification of the Elements at a recent meeting of the New Zealand Institute. The paper included the accompanying Table, which shows the distribution of electrons into groups corresponding to the principal quantum numbers for all the elements and at the same time preserves the most essential features of the two-dimensional arrangement of Mendeleef. Elements having the same complete groups (that is, all stable groups of 8 or 18) are placed in the same horizontal row, and the vertical columns include elements with the same number of electrons in the incomplete outer groups. The electronic configurations are those given by Sidgwick ("Electronic Theory of Valency", 1927). An asterisk marks elements for which the 'normal' atom is thought to have only one electron in the outermost group, but as practically all these give divalent ions, the point is of minor interest chemically. Distribution of electrons into k-subgroups is unnecessary; these have at present little significance for chemical purposes, and in any case the subgroups are considered to be filled in order to the maxima 2, 6, and 10."
René Vernon writes:
In this table Gardner emphasises the existence of four types of elements:
- those with all "groups" complete
- those with one incomplete group
- those with two incomplete groups (transition elements)
- those with three incomplete groups (rare earth elements)
The upper limits of existence of covalencies of 8, 6, and 4 are marked by heavy horizontal lines.
Note:
- there are nine groups of d-block elements [as we would now call them], and but 13 f-block elements
- La and Lu are treated as d-block elements
- while Yb is counted as an f-block element it was later realised (1937) that the 4f shell is full at Yb, hence it is not clear where Gardner would have placed it (Yb)—seemingly in the 0 column

| Year: 1930 | PT id = 1381, Type = structure review |
Quantum Atoms
Dirac, P. A. M. The Principles of Quantum Mechanics. Oxford University Press (1st ed. 1930; 2nd ed. 1935). Wikipeda entry on this work.
von Neumann, J., Mathematische Grundlagen der Quantenmechanik (Mathematical Foundations of Quantum Mechanics), 1932, Springer, Berlin, Germany. Wikepedia entry on this work.
By the 1930s, the mathematics of quantum mechanics was mature, as exemplified by these two text books. Dirac explicitly develops methods for atoms, molecules, radiation, and many-particle systems. Von Neumann formulates a fully general mathematical framework applicable to arbitrarily complex systems (though with few concrete examples).
"The Principles of Quantum Mechanics is an influential monograph written by Paul Dirac and first published by Oxford University Press in 1930. In this book, Dirac presents quantum mechanics in a formal, logically consistent, and axiomatic fashion, making the book the first of its kind. It is based on matrices and operators rather than wave–particle duality. Its 82 sections contain 785 equations with no diagrams. Nor does it have an index, a bibliography, or a list of suggestions for further reading. The first half of the book lays down the foundations of quantum mechanics while the second half focuses on its applications. Dirac did not pursue a historical approach to the subject. Nor did he discuss at length the philosophy of quantum mechanics."
"Von Neumann formalised quantum mechanics using the concept of Hilbert spaces and linear operators. He acknowledged the previous work by Paul Dirac on the mathematical formalisation of quantum mechanics, but was skeptical of Dirac's use of delta functions. He wrote the book in an attempt to be even more mathematically rigorous than Dirac. It was von Neumann's last book in German, afterwards he started publishing in English."
To read Dirac's The Principles of Quantum Mechanics click this link.

 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
© Mark R. Leach Ph.D. 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com
This free, open access web book is an ongoing project and your input is appreciated.