Periodic Table |
 |
 |
 |
 |
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
The INTERNET Database of Periodic Tables
There are thousands of periodic tables in web space, but this is the only comprehensive database of periodic tables & periodic system formulations. If you know of an interesting periodic table that is missing, please contact the database curator: Mark R. Leach Ph.D. The database holds information on periodic tables, the discovery of the elements, the elucidation of atomic weights and the discovery of atomic structure (and much, much more).
Periodic Tables from the year 1937 :
| 1937 | Pozzi's Spiral Periodic Table |
| 1937 | Zmaczynski's Fan-Shaped System |
| 1937 | Technetium, Discovery of |
| 1937 | Geochemical Periodic Table (Goldschmidt Classification) |
| Year: 1937 | PT id = 25, Type = formulation spiral |
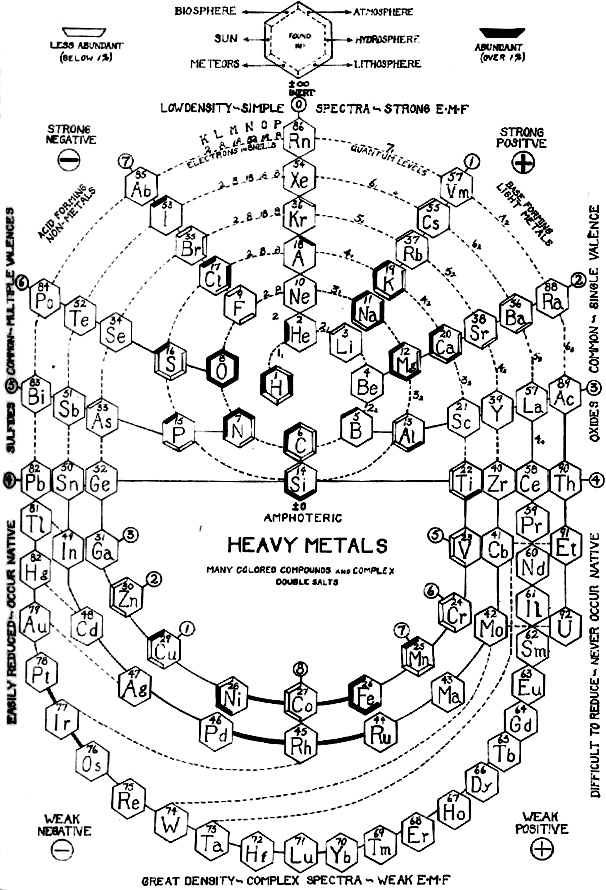
Pozzi Spiral Periodic Table
A spiral periodic table formulation constructed by E.C. Pozzi in 1937, from here.
Note the "Strong Positive, Strong Negative, Weak Positive and Weak Negative" corners:
| Year: 1937 | PT id = 286, Type = formulation |
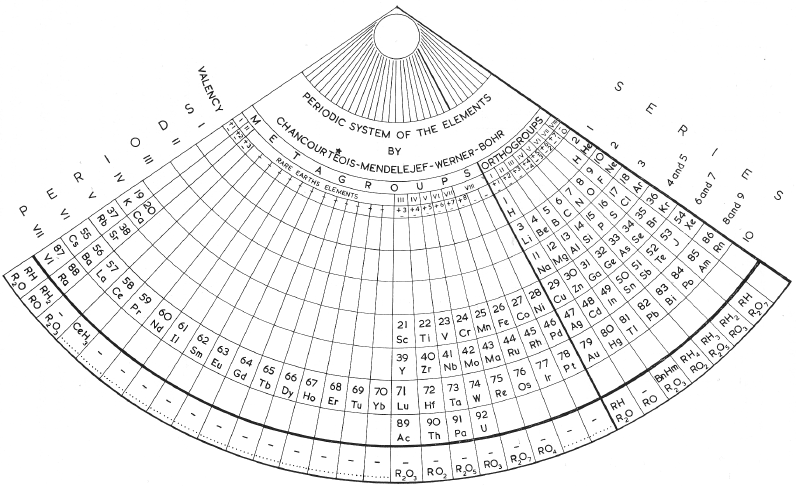
Zmaczynski's Fan-Shaped System
Zmaczynski's fan-shaped system of 1937 (from van Spronsen):
| Year: 1937 | PT id = 823, Type = element |
Discovery of Technetium
Tc ![]()
Technetium, atomic number 43, has a mass of 98 au.
Radioactive element: Tc is only found in tiny amounts in nature. Most samples are synthetic.
Technetium was first isolated in 1937 by C. Perrier and E. Segrè. The element had been predicted by Mendeleev in 1871 as eka-manganese.
| Year: 1937 | PT id = 1074, Type = data |
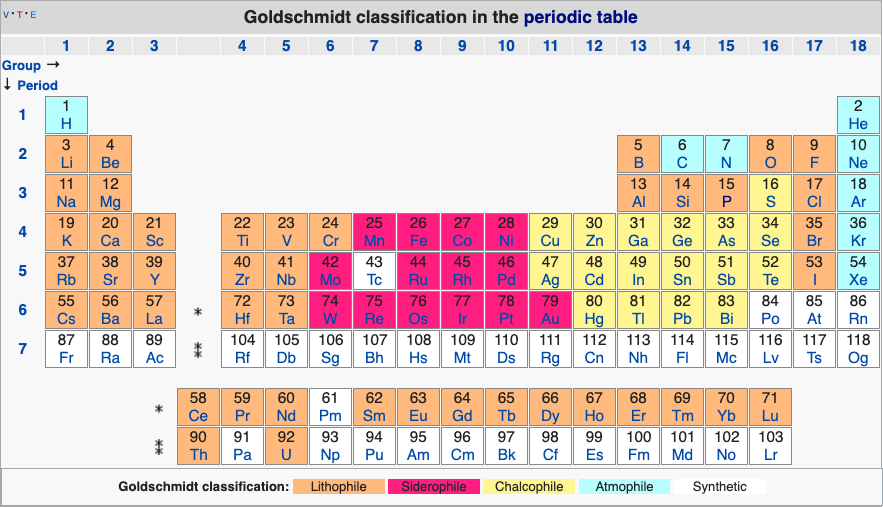
Geochemical Periodic Table (Goldschmidt Classification)
From Wikipedia: The Goldschmidt Classification is a gechemical periodic table which groups the chemical elements within the Earth according to their preferred host phases into:
- lithophile (rock-loving)
- siderophile (iron-loving)
- chalcophile (ore-loving or chalcogen-loving)
- atmophile (gas-loving)
- volatile (the element, or a compound in which it occurs, is liquid or gaseous at ambient surface conditions).
Some elements have affinities to more than one phase. The main affinity is given in the table below and a discussion of each group follows that table.
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
© Mark R. Leach Ph.D. 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com
This free, open access web book is an ongoing project and your input is appreciated.