Periodic Table |
 |
 |
 |
 |
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
The INTERNET Database of Periodic Tables
There are thousands of periodic tables in web space, but this is the only comprehensive database of periodic tables & periodic system formulations. If you know of an interesting periodic table that is missing, please contact the database curator: Mark R. Leach Ph.D. The database holds information on periodic tables, the discovery of the elements, the elucidation of atomic weights and the discovery of atomic structure (and much, much more).
Periodic Tables from the year 1945 :
| 1945 | Seaborg's Periodic Table of 1945 |
| 1945 | Krafft's Periodic Table (1945) |
| 1945 | Promethium, Discovery of |
| 1945 | Talpain's Gnomonic Classification of the Elements |
| Year: 1945 | PT id = 522, Type = formulation structure |
Seaborg's Periodic Table of 1945
From his Priestly Medal Address, The Periodic Table: Tortuous Path to Man-Made Elements printed in C&EN April 16, 1979 and reprinted in Modern Alchemy: Selected Papers of Glenn T. Seaborg (1994), page 181.
Seaborg describes how "the theory was advanced that [the] new elements heavier than than actinium might constitute a second series similar to the series of 'rare-earth' or 'lanthanide' elements":
| Year: 1945 | PT id = 578, Type = formulation |
Krafft's Periodic Table (1945)
From Ether and Matter, p. 86, Carl Frederick Krafft:
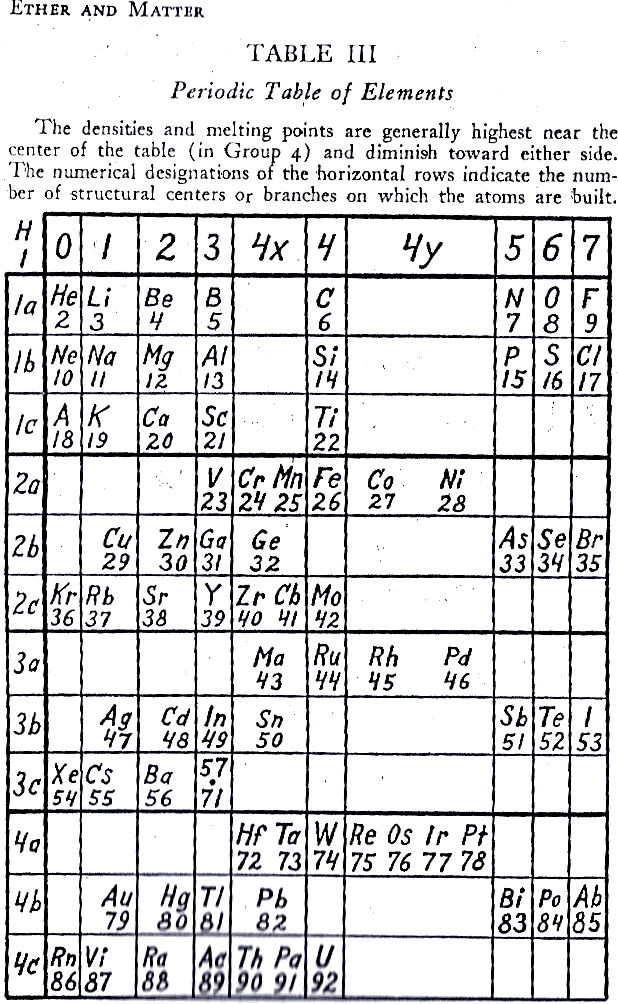
Thanks to Edmond Maurice Peyroux for the tip!
| Year: 1945 | PT id = 841, Type = element |
Discovery of Promethium
Pm ![]()
Promethium, atomic number 61, has a mass of 145 au.
Radioactive element: Pm is only found in tiny amounts in nature. Most samples are synthetic.
Promethium was first observed or predicted in 1942 by S. Wu, E.G. Segrè and H. Bethe and first isolated in 1945 by Charles D. Coryell, Jacob A. Marinsky, Lawrence E. Glendenin, and Harold G. Richter.
| Year: 1945 | PT id = 1118, Type = formulation 3D |
Talpain's Gnomonic Classification of the Elements
Talpain PL 1945, Gnomonic classification of elements, J.Phys. Radium 6, 176-181 (in French), https://doi.org/10.1051/jphysrad:0194500606017600
Talpain writes:
"To overcome the drawbacks presented by the various tables in rows and columns into which the classification of chemical elements is usually inserted, the author proposes a diagram in space, having the form of a double pyramid constructed according to a simple arithmetic law, inspired by Greek surveyors. Under these conditions, all the bodies belonging to the same chemical family are placed on the same column, and all those which have similar physical properties (magnetic, electrical, radioactive, crystallographic, rare earths, etc.) are grouped together. This same diagram also makes it possible to represent the electronic structure of the atoms, the quantified states of the electrons, the energy levels and the spectral lines of hydrogen. Perhaps spectroscopists will be able to use it to also represent the lines of other bodies."
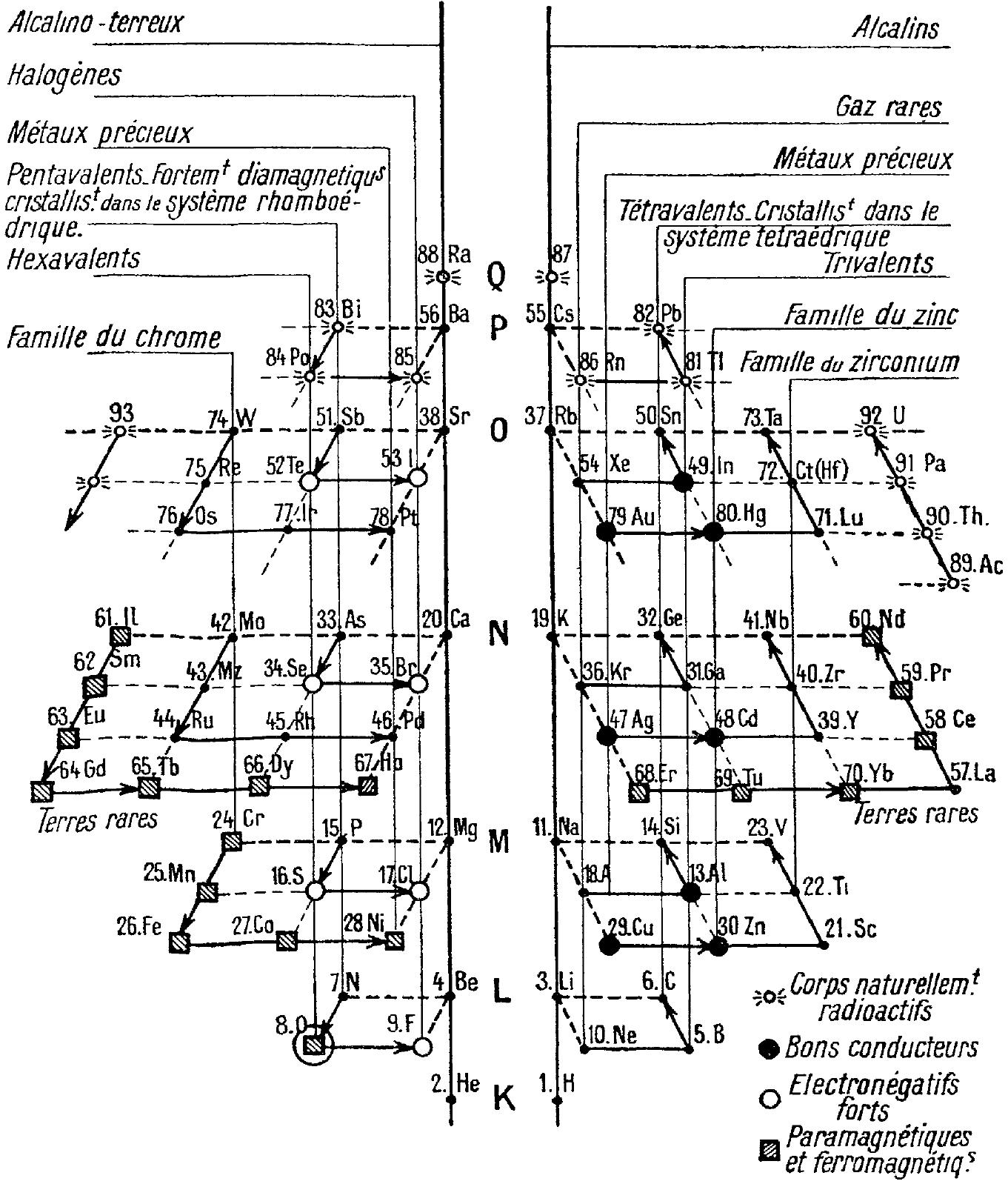
Thanks to René for the tip!
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
© Mark R. Leach Ph.D. 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com
This free, open access web book is an ongoing project and your input is appreciated.