Periodic Table |
 |
 |
 |
 |
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
The INTERNET Database of Periodic Tables
There are thousands of periodic tables in web space, but this is the only comprehensive database of periodic tables & periodic system formulations. If you know of an interesting periodic table that is missing, please contact the database curator: Mark R. Leach Ph.D. The database holds information on periodic tables, the discovery of the elements, the elucidation of atomic weights and the discovery of atomic structure (and much, much more).
Periodic Tables from the year 1947 :
| 1947 | Stedman's Design |
| 1947 | Stedman's Conic System |
| 1947 | Ageev's Crystalline Structures of The Elements |
| 1947 | Science Service: Two Periodic Tables |
| Year: 1947 | PT id = 85, Type = formulation spiral |
Stedman's Design
In his article Stedman says:
- The planar arrangement differs from the plan of his solid model, 'only as necessary to clarify the presentation'
- One the major features is that only two groups, at Si and Y, are considered to be 'truly' branched and that the latter 'is not usually considered in this manner'
- The smaller symbols, such as V under P, aren't necessary but are 'merely offered for consideration'
- Si shows a greater resemblance to Ge than it does to the closer Ti, while Y similarly shows greater resemblance to Lu than to La
- Stedman first drew his first version of this table sixteen years ago (= 1931)
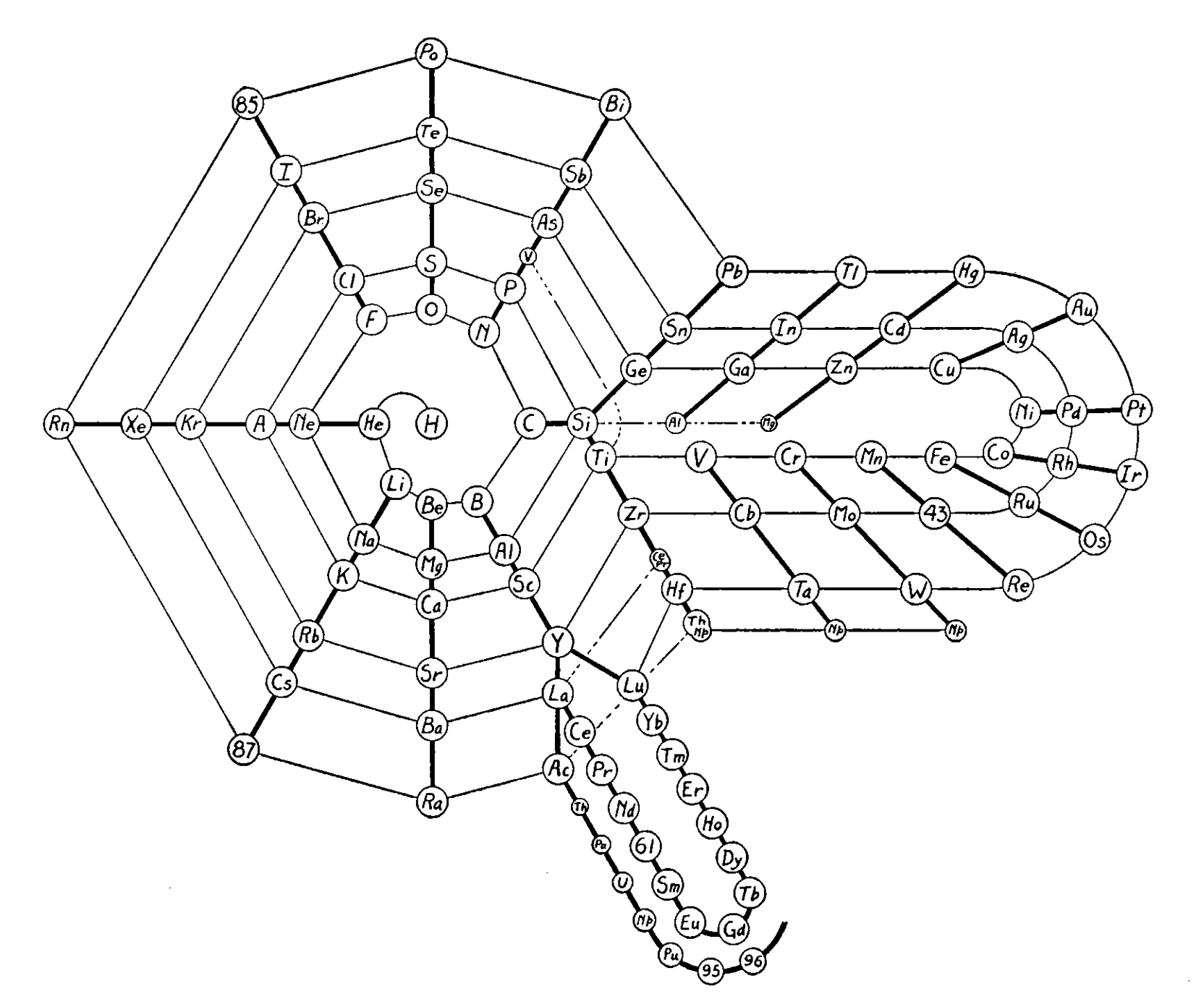
Thanks to René for the tip!
| Year: 1947 | PT id = 291, Type = formulation spiral 3D |
Stedman's Conic System
D. F. Stedman, A Periodic Arrangement of the Elements, Canadian Journal of Research, 1947, 25b(3): 199-210, https://doi.org/10.1139/cjr47b-023
Stedman's conic system from van Spronsen:
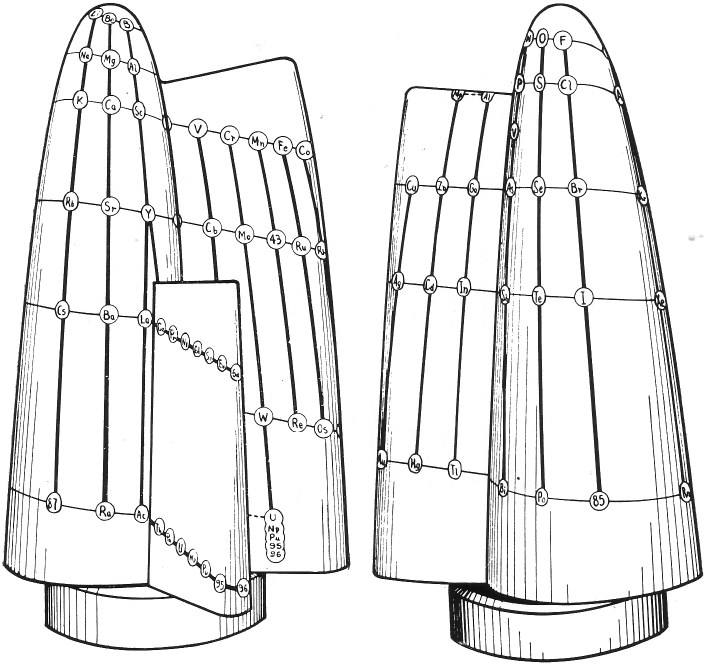
From c&en:
| Year: 1947 | PT id = 1166, Type = formulation |
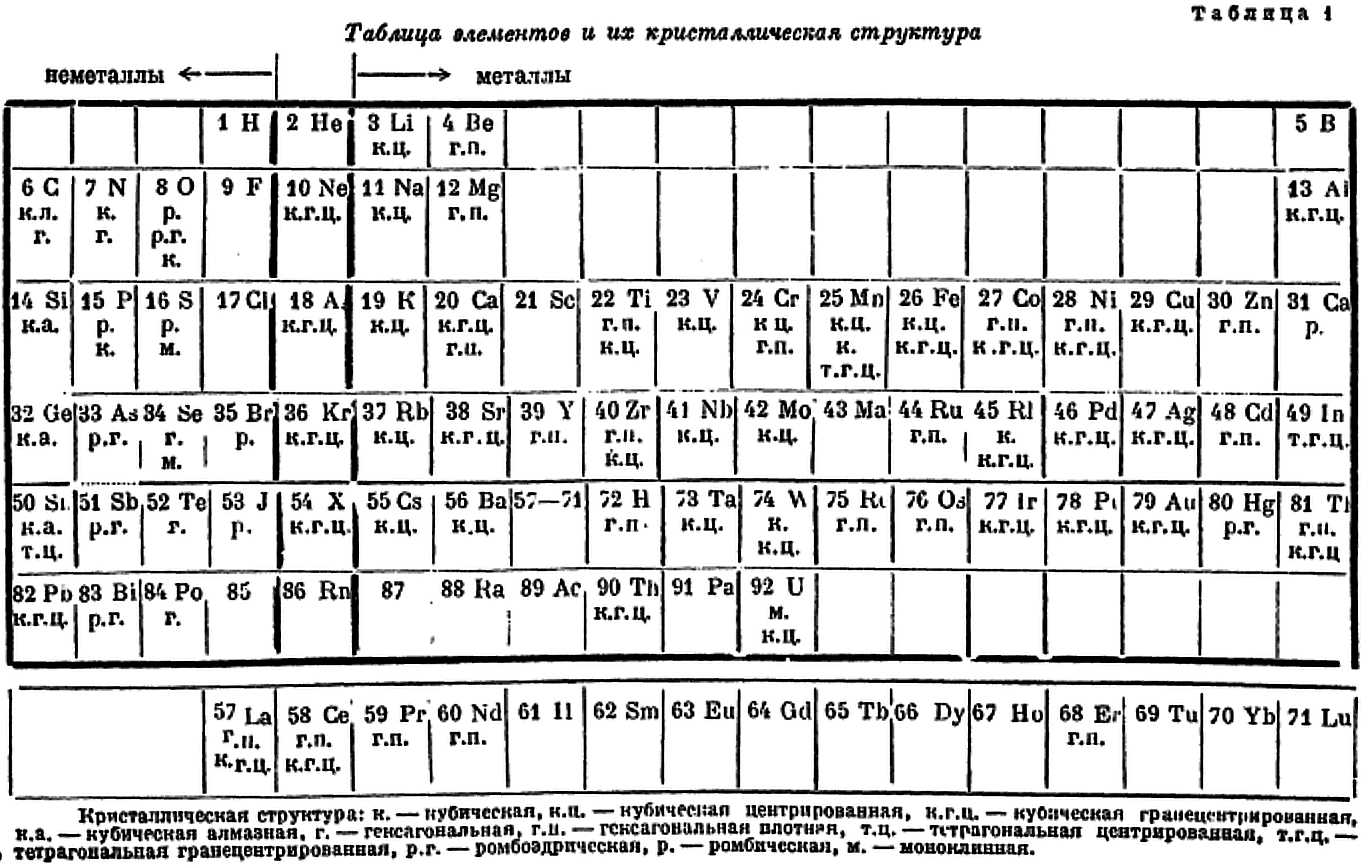
Ageev's Crystalline Structures of The Elements
Ageev NV 1947, The nature of the chemical bond in metal alloys (Izdvo Akad. Nauk SSSR, Moscow/Leningrad, p. 10
René Vernon writes:
"In this curious 18-column table, showing the crystalline structures of the elements, Ageev locates the predominately non-metallic groups on the left and the remaining groups on the right.
"It's odd that he located boron and aluminium on the far left over gallium, rather than over scandium. I suppose he did this so that gallium, indium, and thallium would not be mistaken for d-block metals.
"Reading from left to right then, Ageev's table could be said to be made up of five blocs:"
[1] the nonmetallic bloc
[2] the alkaline bloc
[3] the inner transition bloc
[4] the transition metal block
[5] a post-transition metallic bloc
| Year: 1947 | PT id = 1243, Type = formulation |
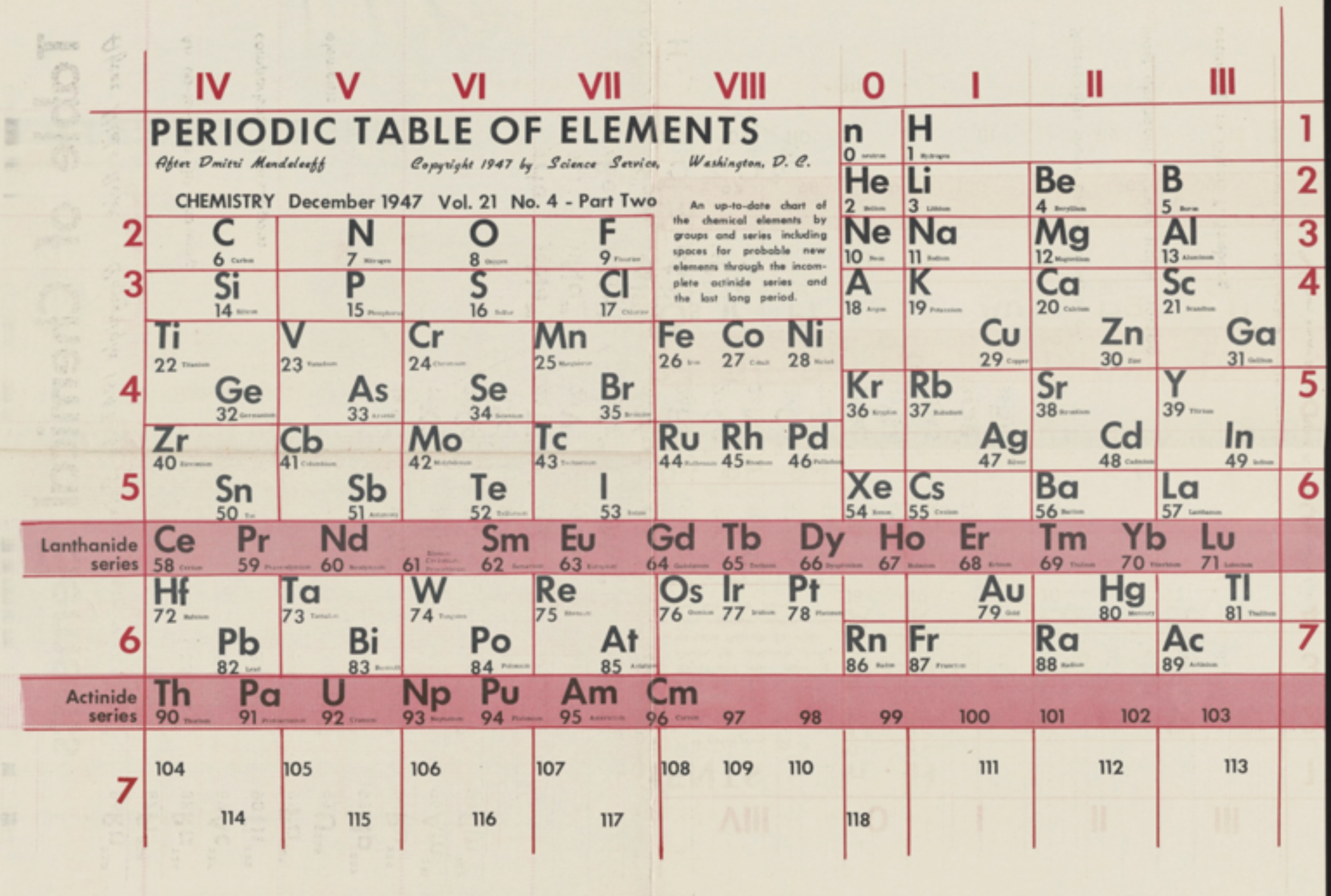
Science Service: Two Periodic Tables
A two-sided Science Service periodic table from 1947. The one is listed as "After Bohr", the other as "After Mendeleeff".
René Vernon writes:
"Here’s a slightly odd table (with two sides):
- The neutron is included in group 0.
- Argon is still A; niobium Cb
- There's a blank space for Pm (discovered 1945).
- The main groups are recognisable, with the exception of group 3 as B-Al-Sc-Y-La. The other side of the table lists B-Al as being analogous to Sc-Y-La, rather than Ga-In-Tl.
The former option works better than the latter in terms of the quantitative smoothness of chemico-physical trend lines going down the group."

 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
© Mark R. Leach Ph.D. 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com
This free, open access web book is an ongoing project and your input is appreciated.
