Periodic Table |
 |
 |
 |
 |
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
The INTERNET Database of Periodic Tables
There are thousands of periodic tables in web space, but this is the only comprehensive database of periodic tables & periodic system formulations. If you know of an interesting periodic table that is missing, please contact the database curator: Mark R. Leach Ph.D. The database holds information on periodic tables, the discovery of the elements, the elucidation of atomic weights and the discovery of atomic structure (and much, much more).
Periodic Tables from the year 1962 :
| 1962 | Scott & Kendal Periodic Table |
| 1962 | Rinck & Feschotte's Tableau Périodique Proposé |
| Year: 1962 | PT id = 1177, Type = formulation |
Scott & Kendal Periodic Table
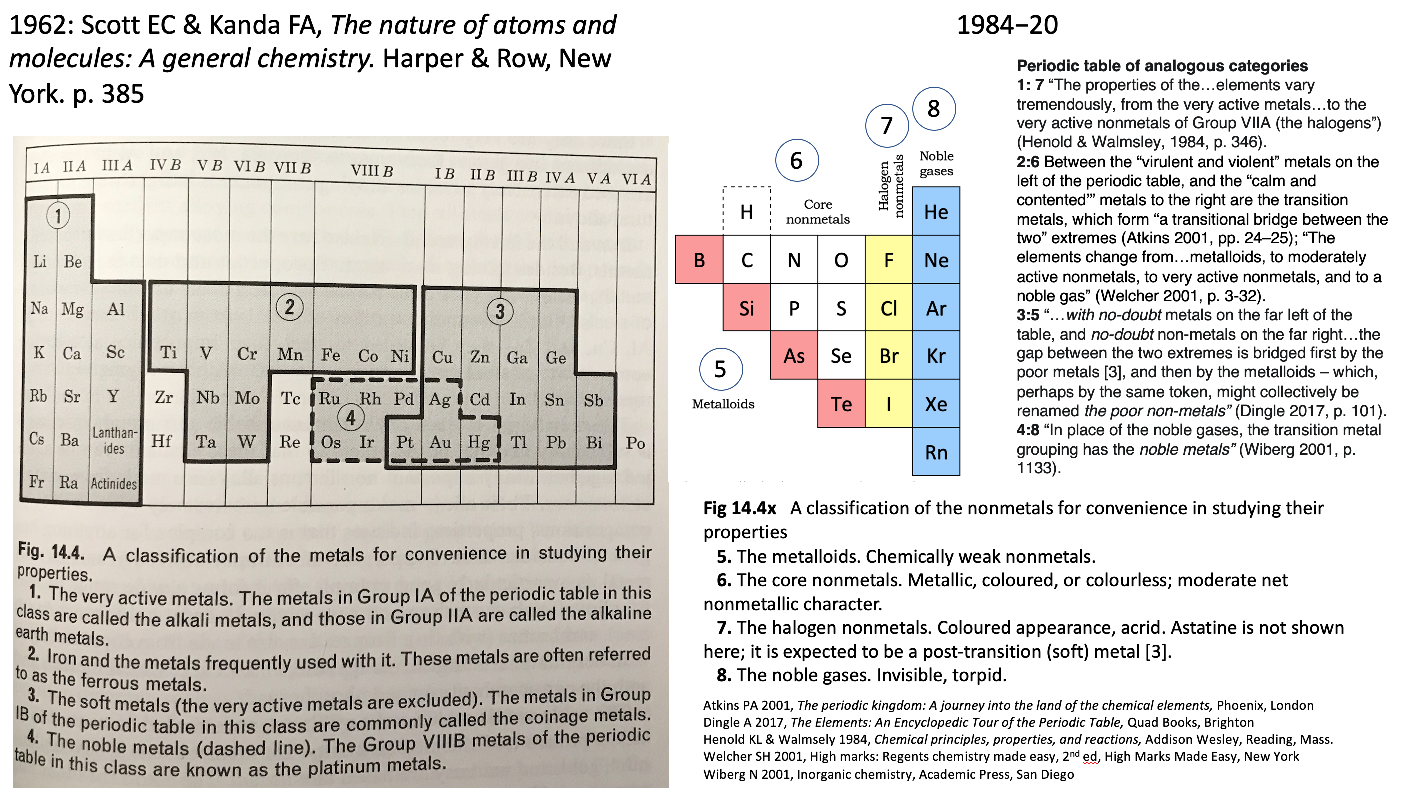
René Vernon shows an extract from Scott E.C. & Kendal F.A., The Nature of Atoms & Molecules: A General Chemistry. Harper & Row, New York, 1962 pp 385, categorising the metals.
Rather than providing a holistic treatment of the nonmetals, the authors take a group-by-group approach.
Items of interest: Al over Sc; the split between groups 3 and 3; and the inclusion of Pt with the soft metals.
On the right is my add-on for the nonmetals, plus extracts from the literature speaking to the analogies between the four metal and four nonmetal categories.
| Year: 1962 | PT id = 1398, Type = formulation |
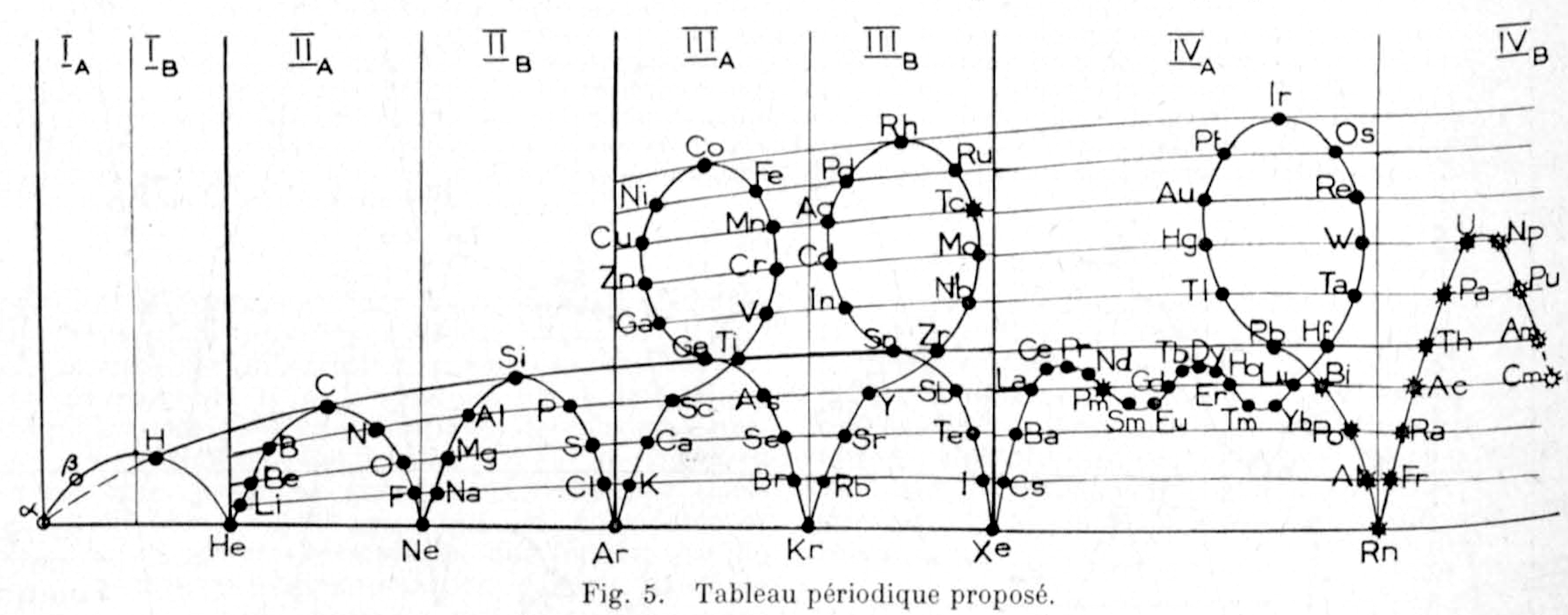
Rinck & Feschotte's Tableau Périodique Proposé
Rinck, E and Feschotte, P. Periodic Classification and The Proust Law. Bull. soc. chim. France, vol. Vol: No. 4, Apr. 1962.
Abstract (Google Translate from French):
Advances in our understanding of the solid state have led to the identification of numerous crystalline phases whose composition is not defined by Proust's law. The rigorous validity of this law, however, served as the starting point for atomic theory: it continues to be used in the measurement of atomic weights and remains valid for the vast field of organic chemistry. The search for the limits of Proust's law's validity leads to certain peculiarities of the metallic state that are closely linked to the periodic table of elements.
A new arrangement in the periodic table, allowing for the first time the integration of rare earth elements and giving hydrogen a very special place, accounts for a distinction between true metals and earth metals. This distinction is necessary because Proust's law, valid for combinations between metalloids and earth metals, is no longer always followed when these same metalloids combine with true metals. Finally, this law loses all meaning in alloys of true metals. The exceptions to this rule are explained by the specialization of chemical properties that occurs when moving from short to long half-lives. Hydrogen and the metals of short half-lives are considered undifferentiated elements, potentially possessing contradictory chemical and even physical properties. Using a perspective borrowed from embryology thus allows for a better understanding of the relationships between these properties.
René Vernon, who provided this PT, writes that "R&F's table shows two extra elements marked as alpha and beta.
Thanks to René for the tip!
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
© Mark R. Leach Ph.D. 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com
This free, open access web book is an ongoing project and your input is appreciated.