Periodic Table |
 |
 |
 |
 |
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
The INTERNET Database of Periodic Tables
There are thousands of periodic tables in web space, but this is the only comprehensive database of periodic tables & periodic system formulations. If you know of an interesting periodic table that is missing, please contact the database curator: Mark R. Leach Ph.D. The database holds information on periodic tables, the discovery of the elements, the elucidation of atomic weights and the discovery of atomic structure (and much, much more).
| Year: 1996 | PT id = 238, Type = formulation misc |
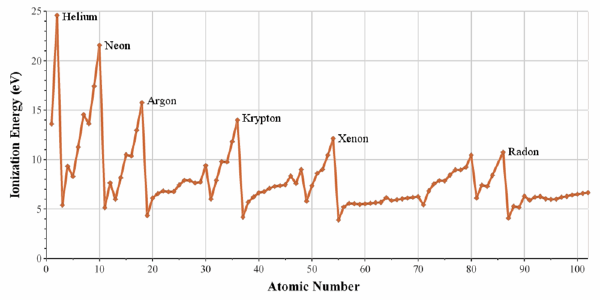
First Ionisation Energy of The Elements
Periodic trend for ionization energy, for example Mg → Mg+ + e–
Each period begins at a minimum for the alkali metals, and ends at a maximum for the noble gases. From Wikipedia:
Based on data from: Martin, W. C.; Wiese, W. L. (1996). Atomic, Molecular, & Optical Physics Handbook. American Institute of Physics. ISBN 156396242X.
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
© Mark R. Leach Ph.D. 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com
This free, open access web book is an ongoing project and your input is appreciated.