Periodic Table |
 |
 |
 |
 |
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
The INTERNET Database of Periodic Tables
There are thousands of periodic tables in web space, but this is the only comprehensive database of periodic tables & periodic system formulations. If you know of an interesting periodic table that is missing, please contact the database curator: Mark R. Leach Ph.D. The database holds information on periodic tables, the discovery of the elements, the elucidation of atomic weights and the discovery of atomic structure (and much, much more).
| Year: 2012 | PT id = 492, Type = data |
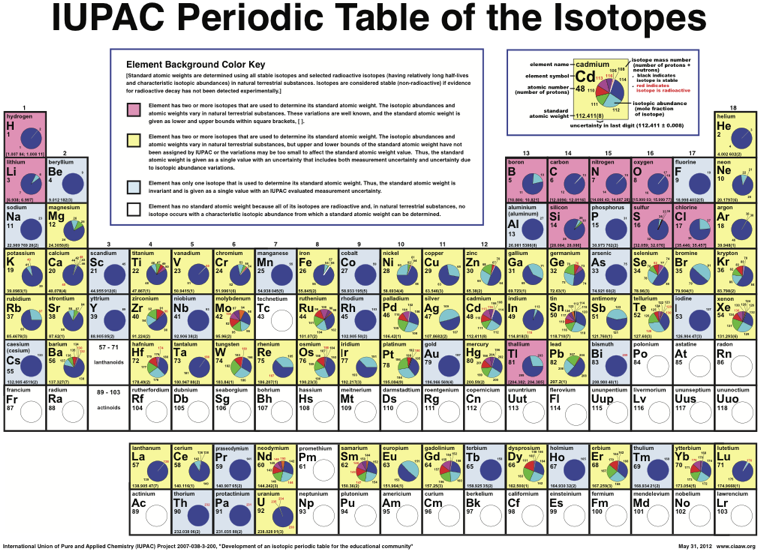
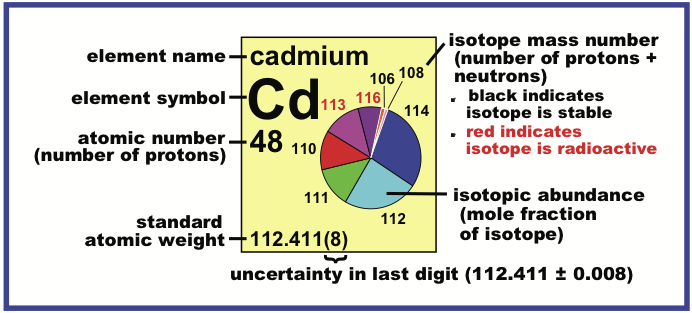
IUPAC Periodic Table of the Isotopes
The Periodic Table of the Isotopes, published by International Union of Pure and Applied Chemistry (IUPAC), is now available from the Commission on Isotopic Abundances and Atomic Weights, which is a commission under the Inorganic Division (Division II) of IUPAC.
The text identifies four types of atom, with respect to isotopes:
- Element has two or more isotopes that are used to determine its standard atomic weight. The isotopic abundances and atomic weights vary in natural terrestrial substances. These variations are well known, and the standard atomic weight is given as lower and upper bounds within square brackets, [ ].
- Element has two or more isotopes that are used to determine its standard atomic weight. The isotopic abundances and atomic weights vary in natural terrestrial substances, but upper and lower bounds of the standard atomic weight have not been assigned by IUPAC or the variations may be too small to affect the standard atomic weight value. Thus, the standard atomic weight is given as a single value with an uncertainty that includes both measurement uncertainty and uncertainty due to variations in isotopic abundances.
- Element has only one isotope that is used to determine its standard atomic weight. Thus, the standard atomic weight is
invariant and is given as a single value with an IUPAC evaluated measurement uncertainty.
- Element has no standard atomic weight because all of its isotopes are radioactive and, in natural terrestrial substances, no isotope occurs with a characteristic isotopic abundance from which a standard atomic weight can be determined.
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
© Mark R. Leach Ph.D. 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com
This free, open access web book is an ongoing project and your input is appreciated.