Periodic Table |
 |
 |
 |
 |
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
The INTERNET Database of Periodic Tables
There are thousands of periodic tables in web space, but this is the only comprehensive database of periodic tables & periodic system formulations. If you know of an interesting periodic table that is missing, please contact the database curator: Mark R. Leach Ph.D. The database holds information on periodic tables, the discovery of the elements, the elucidation of atomic weights and the discovery of atomic structure (and much, much more).
| Year: 2016 | PT id = 730, Type = formulation misc |
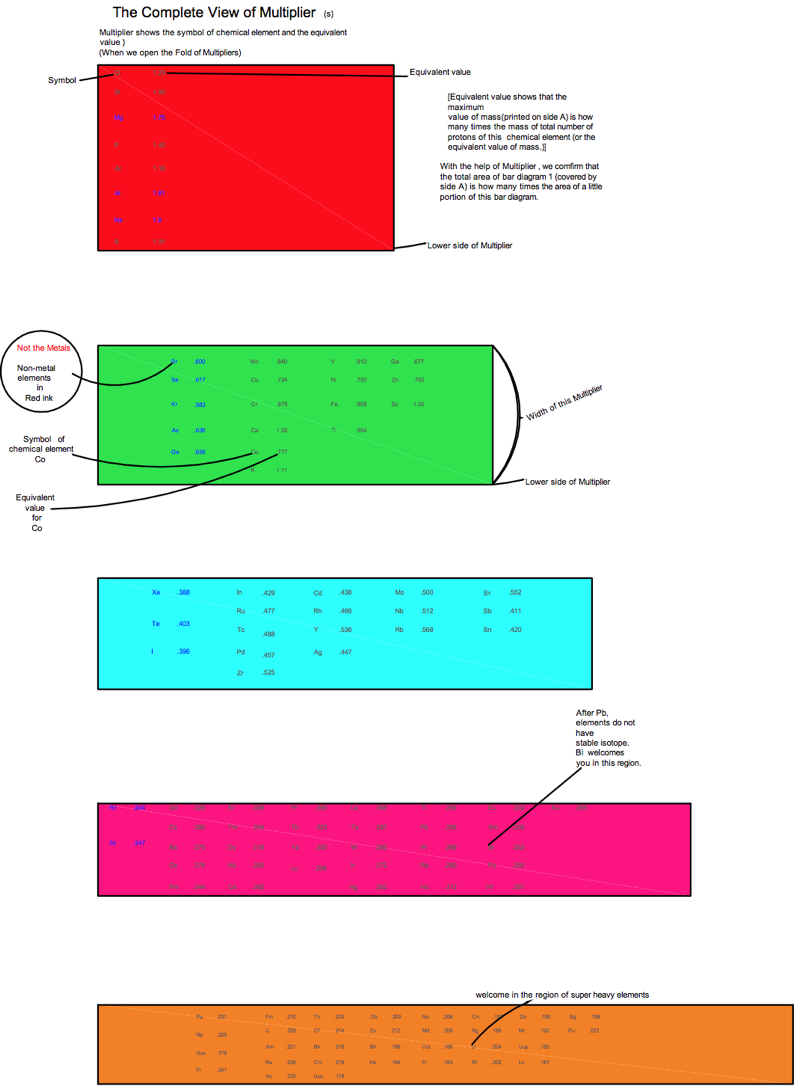
KAS Periodic Table
The KAS periodic table reproduces and depicts the nuclear properties of chemical elements. This periodic table depicts not only the trends of nuclear properties, but also reproduces their numerical values that remain very close to the experimental values (difference less than 4%).
The Segre Chart is based on the number of protons, Z, and the number of neutrons, N. It is like a library of nuclei and shows the recorded data only. The Segre Chart can not work when the number of neutrons is not given. But KAS Periodic Table works when the number of neutrons is not given.It does not require the number of neutrons to produce the results.This is a simple chart based on the number of protons of chemical element. We identify the following properties of elements:-
- Location that remains near the Neutron Dripline of element.
- Location that remains very close to stable or long-lived isotopes of the element. Location that remains near the Proton Dripline of element.
- In the case of superheavy elements, we identify which Compound Nuclei are involved in the Hot Fusion reaction and which Compound Nuclei are involved in the Cold Fusion reaction.
- We see the r-process path and assess the r-process abundance.
- The pattern of abundance of chemical elements.
- We identify which elements are the product of exothermal fusion.
- We identify the location of isotope on the basis of two-neutron separation energy.
- Nuclear binding energy trend. Beta decay trend.
- We see the Straight Line of Nuclear Stability.
- Empirical Law discovered.
- Periodicity in the nuclear properties.
- We can compare the nuclear properties of an element with the nuclear properties of almost all the chemical elements.
Read more here, here and here.
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
© Mark R. Leach Ph.D. 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com
This free, open access web book is an ongoing project and your input is appreciated.