Periodic Table |
 |
 |
 |
 |
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
The INTERNET Database of Periodic Tables
There are thousands of periodic tables in web space, but this is the only comprehensive database of periodic tables & periodic system formulations. If you know of an interesting periodic table that is missing, please contact the database curator: Mark R. Leach Ph.D. The database holds information on periodic tables, the discovery of the elements, the elucidation of atomic weights and the discovery of atomic structure (and much, much more).
| Year: 1907 | PT id = 752, Type = formulation |
van den Broek Periodic Table 1
From Wikipedia: Antonius Johannes van den Broek (1870-1926) was a Dutch amateur physicist notable for being the first who realized that the number of an element in the periodic table (now called atomic number) corresponds to the charge of its atomic nucleus. The 1911 inspired the experimental work of Henry Moseley, who found good experimental evidence for it by 1913. van den Broek envisaged the basic building block to be the 'alphon', which weighed twice as much as a hydrogen atom.
Read more in Chapter 4, Antonius Van Den Broek, Moseley and the Concept of Atomic Number by Eric Scerri. This chapter can be found in the book: For Science, King & Country: The Life and Legacy of Henry Moseley (Edited by Roy MacLeod, Russell G Egdell and Elizabeth Bruton).
van den Broek's periodic table of 1907: Annalen der Physik, 4 (23), (1907), 199-203
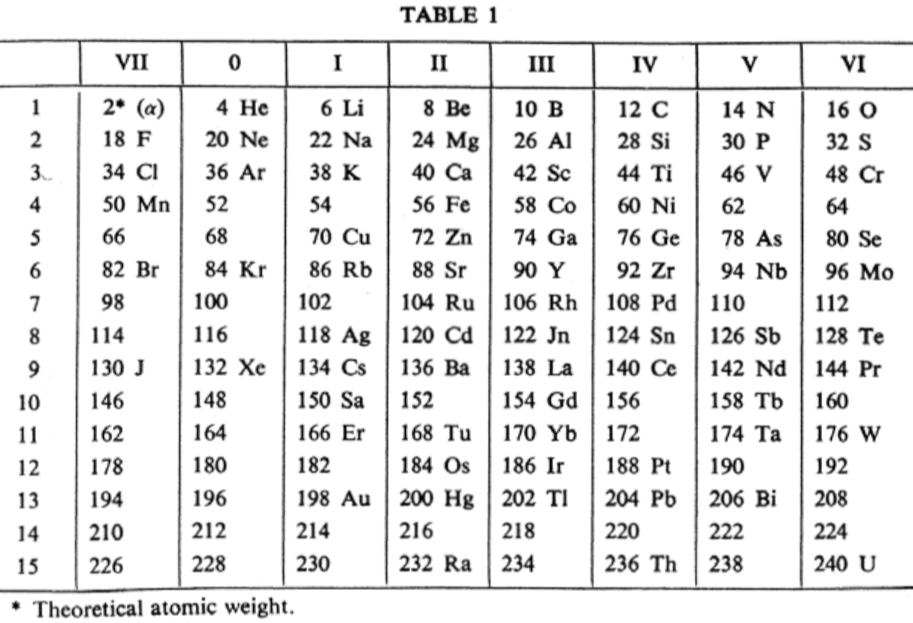
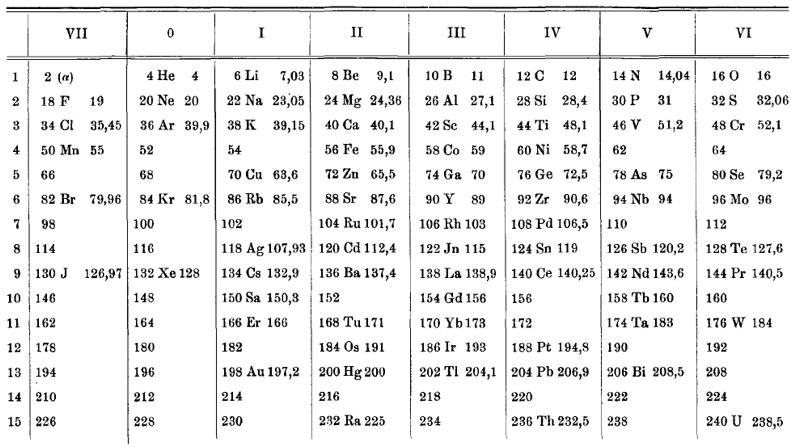
Thanks to Eric Scerri for the tip!
See the website EricScerri.com and Eric's Twitter Feed.
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
© Mark R. Leach Ph.D. 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com
This free, open access web book is an ongoing project and your input is appreciated.