Periodic Table |
 |
 |
 |
 |
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
The INTERNET Database of Periodic Tables
There are thousands of periodic tables in web space, but this is the only comprehensive database of periodic tables & periodic system formulations. If you know of an interesting periodic table that is missing, please contact the database curator: Mark R. Leach Ph.D. The database holds information on periodic tables, the discovery of the elements, the elucidation of atomic weights and the discovery of atomic structure (and much, much more).
Periodic Tables from the year 1808 :
| 1808 | Dalton's Elements |
| 1808 | Boron, Discovery of |
| 1808 | Magnesium, Discovery of |
| 1808 | Calcium, Discovery of |
| 1808 | Strontium, Discovery of |
| 1808 | Barium, Discovery of |
| Year: 1808 | PT id = 5, Type = formulation data element weight structure |
Dalton's Elements
Two pages from John Dalton's A New System of Chemical Philosophy in which he proposed his version of atomic theory based on scientific experimentation (see the scanned book, page 219):

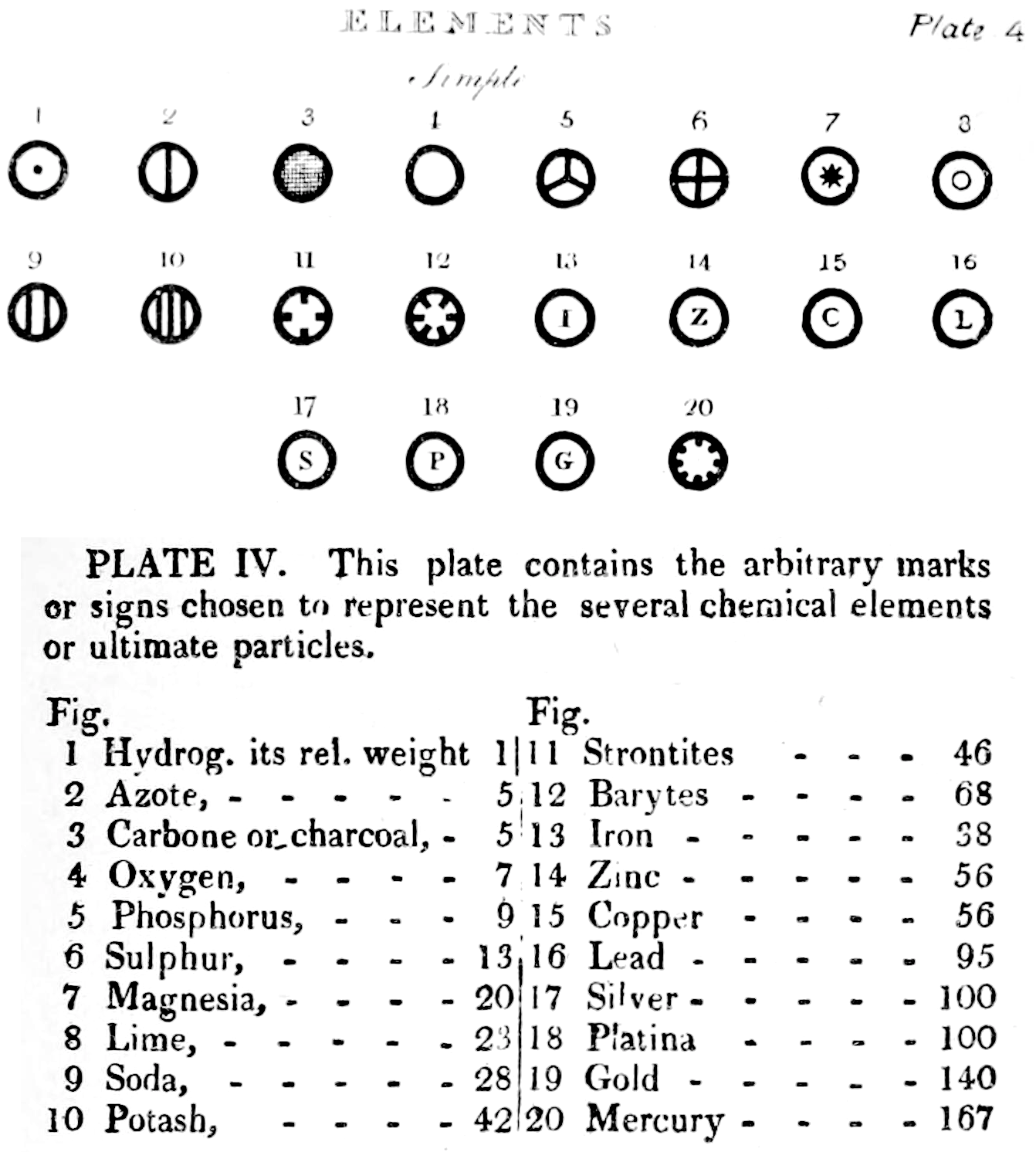
| Name | Modern Symbol | Dalton's Data | Modern Values | % error |
| Hydrog. | H | 1 | 1 | 0% |
| Azote | N | 5 | 14 | -180% |
| Carbone | C | 5 | 12 | -140% |
| Oxygen | O | 7 | 16 | -129% |
| Phosphorus | P | 9 | 31 | -244% |
| Sulphur | S | 13 | 32.1 | -147% |
| Magnesia | Mg | 20 | 24.3 | -22% |
| Lime | Ca | 24 | 40.1 | -67% |
| Soda | Na | 28 | 23 | 18% |
| Potash | K | 42 | 39.1 | 7% |
| Strontites | Sr | 46 | 87.6 | -90% |
| Barytes | Ba | 68 | 137.3 | -102% |
| Iron | Fe | 50 | 55.8 | -12% |
| Zinc | Zn | 56 | 65.4 | -17% |
| Copper | Cu | 56 | 63.5 | -13% |
| Lead | Pb | 90 | 200.6 | -123% |
| Silver | Ag | 190 | 107.9 | 43% |
| Gold | Au | 190 | 197 | -4% |
| Platina | Pt | 190 | 195.1 | -3% |
| Mercury | Hg | 167 | 200.6 | -20% |
- Dalton states that he is considering "chemical elements or ultimate particles"
- Dalton assigns hydrogen as having a relative weight of 1.
- Note the seemingly huge % errors in the atomic weights, compared with modern values.
- These errors occurred because while Dalton had deduced that atoms combine in fixed (stoichiometric) ratios in compounds, he not always know what the ratios were. Thus there were two unknowns: the atomic weights (masses) and the stoichiometric ratios.

By Mark Leach
| Year: 1808 | PT id = 785, Type = element |
Discovery of Boron
B
Boron, atomic number 5, has a mass of 10.814 au.
Boron has properties that are borderline between metal and non-metal (semimetallic).
Boron was first observed or predicted in 1808 by L. Gay-Lussac and L.J. Thénard and first isolated in 1808 by H. Davy.
| Year: 1808 | PT id = 791, Type = element |
Discovery of Magnesium
Mg
Magnesium, atomic number 12, has a mass of 24.306 au.
Magnesium is a Group 2 element, and these are called: "alkaline earth metals".
Magnesium was first observed or predicted in 1755 by J. Black and first isolated in 1808 by H. Davy.
| Year: 1808 | PT id = 800, Type = element |
Discovery of Calcium
Ca
Calcium, atomic number 20, has a mass of 40.078 au.
Calcium is a Group 2 element, and these are called: "alkaline earth metals".
Calcium was first isolated in 1808 by H. Davy.
| Year: 1808 | PT id = 818, Type = element |
Discovery of Strontium
Sr
Strontium, atomic number 38, has a mass of 87.62 au.
Strontium is a Group 2 element, and these are called: "alkaline earth metals".
Strontium was first observed or predicted in 1787 by W. Cruikshank and first isolated in 1808 by H. Davy.
| Year: 1808 | PT id = 836, Type = element |
Discovery of Barium
Ba
Barium, atomic number 56, has a mass of 137.327 au.
Barium is a Group 2 element, and these are called: "alkaline earth metals".
Barium was first observed or predicted in 1772 by W. Scheele and first isolated in 1808 by H. Davy.
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
© Mark R. Leach Ph.D. 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com
This free, open access web book is an ongoing project and your input is appreciated.