Periodic Table |
 |
 |
 |
 |
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
The INTERNET Database of Periodic Tables
There are thousands of periodic tables in web space, but this is the only comprehensive database of periodic tables & periodic system formulations. If you know of an interesting periodic table that is missing, please contact the database curator: Mark R. Leach Ph.D. The database holds information on periodic tables, the discovery of the elements, the elucidation of atomic weights and the discovery of atomic structure (and much, much more).
Periodic Tables from the year 1925 :
| Year: 1925 | PT id = 84, Type = formulation 3D |
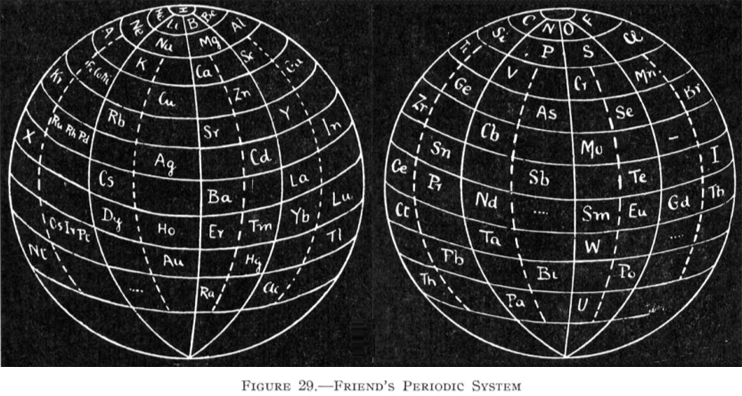
Friend's Periodic Sphere
J. A. N. Friend, "The periodic sphere and the position of the rare earth metals", Chem. News., 130, 196-7 (Mar., 1925).
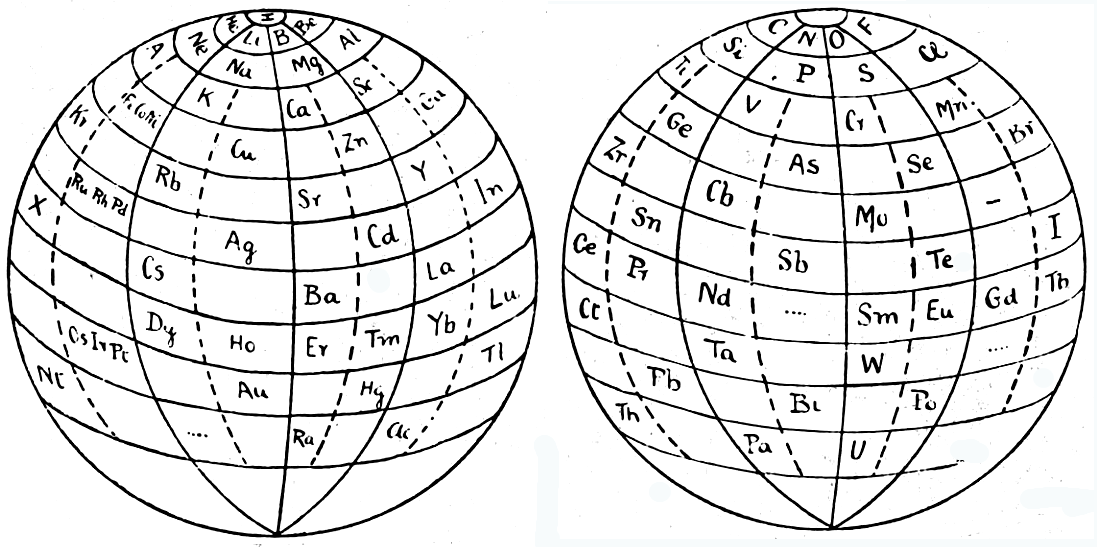
From Quam & Quam's 1934 review paper.pdf
| Year: 1925 | PT id = 308, Type = formulation |
Noddack Periodic Table
Ida Noddack studied the periodic table in the first half of the 20th century and was the co-discoverer of the last non-radioactive element to be isolated, rhenium. Later she worked on nuclear fission. In 1925 presented Noddack her formulation:
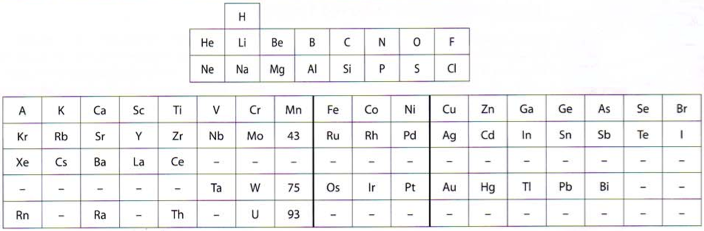
From Ida Noddack and the Missing Elements by Fathi Habashi, Education in Chemistry (March 2009)
| Year: 1925 | PT id = 460, Type = formulation |
Deming's (Updated) Periodic Table
This 1925 table has the Heavy Metals spread out, and the Rare Earth Elements (fifteen, including La and Lu) withdrawn into a box that is divorced from the body of the table. Ce, Gd, Yb form a vertical triad.
Th is assigned to Group IV below Hf.
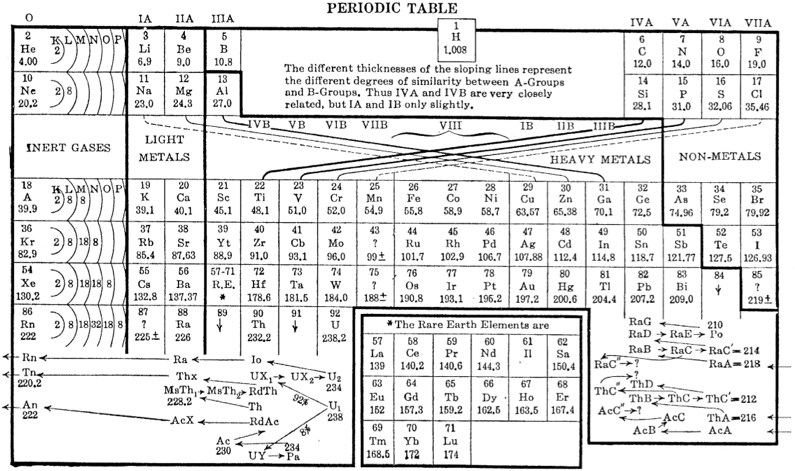
From Michael Laing's paper: A Revised Periodic Table with the Lanthanides Repositioned, Found. Chem. (2005) 7: 203–233
| Year: 1925 | PT id = 735, Type = formulation 3D spiral |
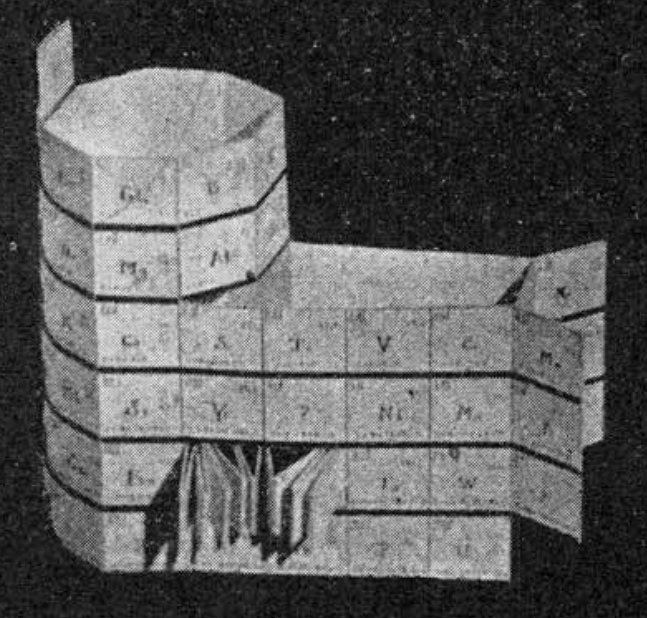
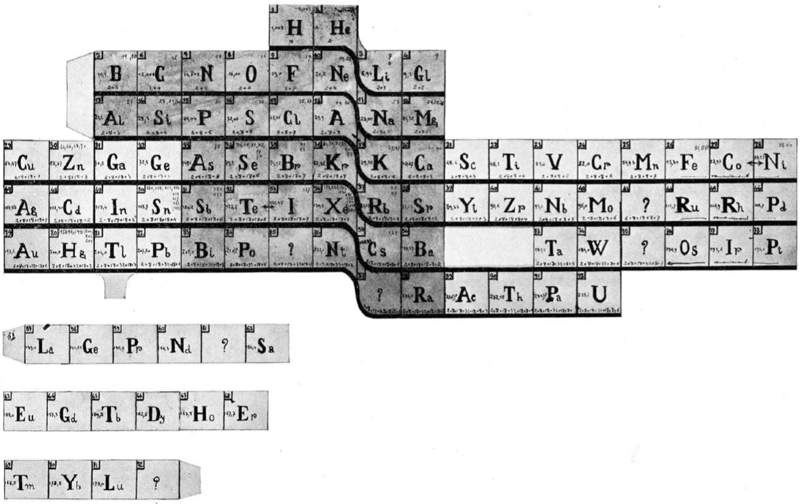
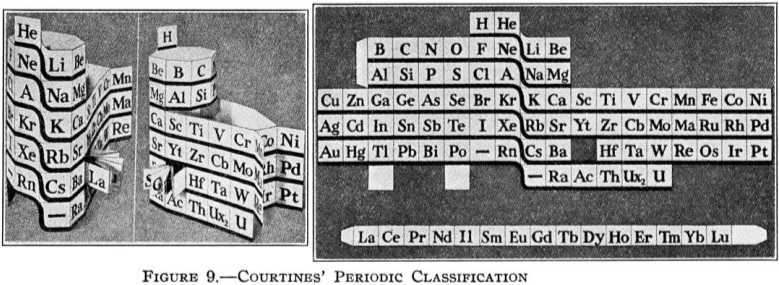
Courtines' Model of the Periodic Table or Periodic Classification
Published in J. Chem. Ed., 2, 2, 107-109 in 1925 by M. Courtines of the Laboratory of Experimental Physics, College of France, Paris.
Q&Q write:
"The unfolded tower arrangement appears much like a modernised Chauvierre chart cut on a line between Ni and Cu, Cu, with the right part fitted to the left in order of increasing atomic numbers. The rare-earth elements, however, are placed on a novel accordion-like folded strip with ends made secure just below Xt and between Ba and Hf. The author describes in detail the method of folding the chart into a tower-like cylindrical model. H is folded back to show its lack of relationship other groups of elements. In the space for each symbol, electron arrangements and isotopes are also enumerated."
And, in what appears to be a 'top down' view of the above 3D formulation, Courtine M 1926, Oùen est la physique, Gauthier-Villars et Cie, Paris:
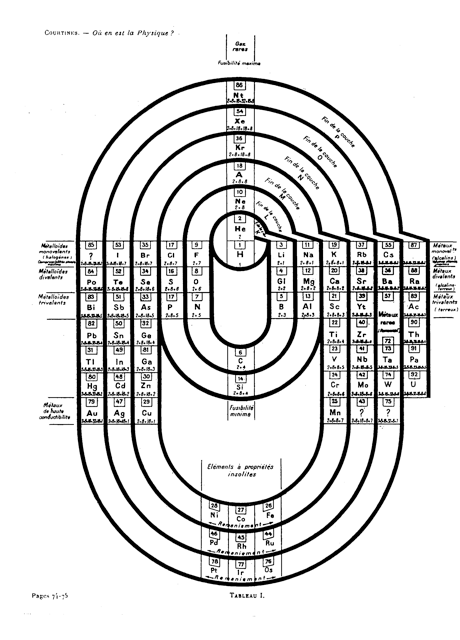
Thanks to Eric Scerri for the tip & René Vernon's additions!
See the website EricScerri.com and Eric's Twitter Feed
| Year: 1925 | PT id = 926, Type = formulation |
Sommerfeld's Electon Filling Diagram
Arnold Sommerfeld diagram appears in an issue of Memoirs and Proceedings of the manchester Literary and Philosophical Society for 1925-26. volume 70, p. 141-151.
Eric Scerri writes:
"The electron groupings are not exactly the same as what is believed to exist today but it amounts to the same order of filling. For example p orbitals were thought to consist of two groups of 2 and 4 electrons, rather than 2, 2, 2 as believed today. Similarly d orbitals were thought to be formed of two groups of 4 and 6 electrons. With that in mind you will see that Sommerfeld was the first to propose an aufbau filling system: The occupation of 4s before 3d or as represented here the 2 electrons in orbit 11 followed by the 4 and 6 from orbits 3,s and 3,3.
"Sommerfeld does indicate sub-shells. They are just not the same groupings as the current ones. For example 2,1 and 2,2 indicates subshells within the 2nd main shell. Similarly the 3rd shell is presented as 3,2 and 3,3. The totals are of course the same, namely 6 for what we now call p orbitals and 10 for what we call d orbitals. All this came before the discovery of the 4th or spin quantum number. This is in keeping with Bohr's original assignment of shells and sub-shells.
"The discovery of sub-structure to electron shells was not an 'all or nothing' development, but a gradual and almost organic evolution."
Eric has a new book out – A Tale of Seven Scientists and a New Philosophy of Science – in which the gradual evolution of electronic structure involving Bohr, Sommerfeld, Bury, Main Smith, Pauli and others is traced out.
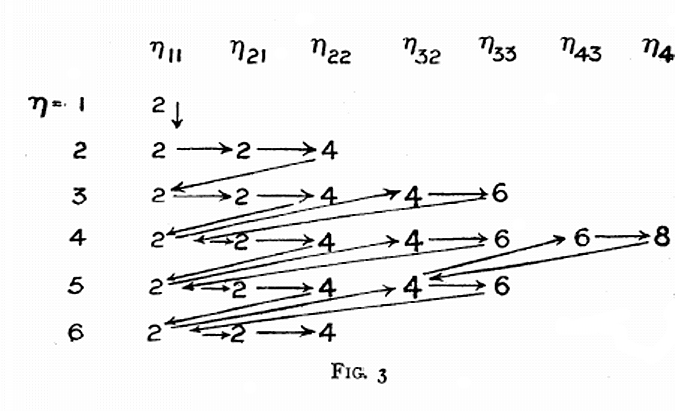
Thanks to Eric Scerri for the tip!
See the website EricScerri.com and Eric's Twitter Feed
| Year: 1925 | PT id = 1035, Type = formulation 3D |
Model of the Periodic System of de Chancourtois
From the Science Museum in the UK collection, a model of the Periodic System of de Chancourtois from 1862:
"Model demonstrating the telluric screw periodic system of Alexander-Emile Beguyer de Chancourtois proposed in a paper published in 1862.
"This model, made by the Science Museum in 1925, provides a rare physical realisation of arguably the earliest periodic system of for the elements. It was devised by the French geologist, Alexander-Emile Beguyer de Chancourtois in 1862, 7 years prior to Dmitri Mendeleev's periodic table.
"De Chancourtois arranged the elements in the order of their atomic weights along a helix which was traced on the surface of a vertical cylinder, with an angle of 45 degrees to its axis. The base of the cylinder was divided into 16 equal parts (the atomic weight of oxygen), and the lengths of the spiral corresponding to the weights of the elements were found by taking the one-sixteenth part of a complete turn as a unit":

| Year: 1925 | PT id = 1373, Type = structure |
Pauli and The Exclusion Principle
Pauli, W. Über den Zusammenhang des Abschlusses der Elektronengruppen im Atom mit der Komplexstruktur der Spektren. Zeitschrift für Physik, 31, 765–783 (1925).
"Wolfgang Pauli was an Austrian–Swiss theoretical physicist and a pioneer of quantum mechanics. In 1945, after having been nominated by Albert Einstein, Pauli received the Nobel Prize in Physics 'for the discovery of the Exclusion Principle, also called the Pauli Principle'. The discovery involved spin theory.
"The Pauli exclusion principle (German: Pauli-Ausschlussprinzip) states that two or more identical particles with half-integer spins (i.e. fermions) cannot simultaneously occupy the same quantum state within a system that obeys the laws of quantum mechanics. This principle was formulated by Austrian physicist Wolfgang Pauli in 1925 for electrons, and later extended to all fermions with his spin–statistics theorem of 1940.
"In the case of electrons in atoms, the exclusion principle can be stated as follows: in a poly-electron atom it is impossible for any two electrons to have the same two values of all four of their quantum numbers, which are: n, the principal quantum number; ℓ, the azimuthal quantum number; mℓ, the magnetic quantum number; and ms, the spin quantum number.
"If two electrons reside in the same orbital, then their values of n, ℓ, and mℓ are equal. In that case, the two values of ms (spin) pair must be different. Since the only two possible values for the spin projection ms are +1/2 and –1/2, it follows that one electron must have ms = +1/2 and one ms = –1/2.
"To preserve the conservation of energy in beta decay, Pauli proposed the existence of a small neutral particle, dubbed the neutrino by Enrico Fermi, in 1930. Neutrinos were first detected in 1956. "
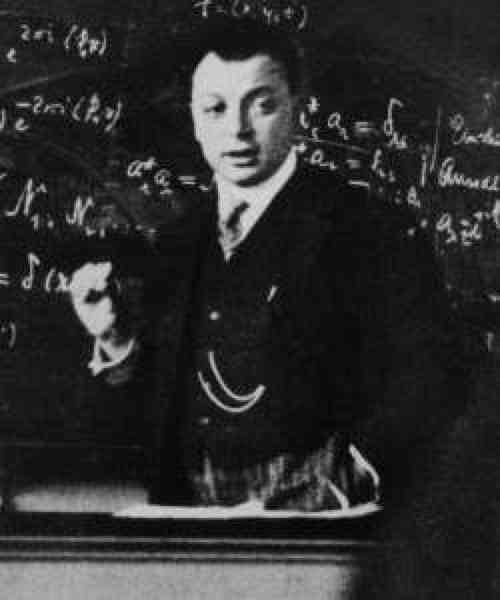
| Year: 1925 | PT id = 1374, Type = structure |
Heisenberg’s Matrix Mechanics and the Uncertainty Principle
Heisenberg, W. Über quantentheoretische Umdeutung kinematischer und mechanischer Beziehungen. (On the quantum-theoretical reinterpretation of kinematic and mechanical relationships.) Zeitschrift für Physik, 33, 879–893 (1925); Heisenberg, W. Über den anschaulichen Inhalt der quantentheoretischen Kinematik und Mechanik. (On the intuitive content of quantum-theoretical kinematics and mechanics. "The Uncertainty Principle") Zeitschrift für Physik, 43, 172–198 (1927).
"Werner Karl Heisenberg was a German theoretical physicist, one of the main pioneers of the theory of quantum mechanics. Heisenberg published his Umdeutung paper in 1925, a major reinterpretation of old quantum theory.
"In the subsequent series of papers with Max Born and Pascual Jordan, during the same year, his matrix formulation of quantum mechanics was substantially elaborated. He is also known for the uncertainty principle, which he published in 1927. He received the Nobel Prize in Physics in 1932 'for the creation of quantum mechanics'."
Below is a largely non-mathematical video that discusses the development of Heisenberg's matrix mechanics and then compares it to the Schrödinger wave equation analysis:

 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
© Mark R. Leach Ph.D. 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com
This free, open access web book is an ongoing project and your input is appreciated.