Periodic Table |
 |
 |
 |
 |
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
The INTERNET Database of Periodic Tables
There are thousands of periodic tables in web space, but this is the only comprehensive database of periodic tables & periodic system formulations. If you know of an interesting periodic table that is missing, please contact the database curator: Mark R. Leach Ph.D. The database holds information on periodic tables, the discovery of the elements, the elucidation of atomic weights and the discovery of atomic structure (and much, much more).
Periodic Tables from the year 1949 :
| Year: 1949 | PT id = 27, Type = formulation |
Pauling's Formulation
Linus Pauling borrowed von Antropoff 1926 design, without acknowledgement, for his 1949 book, General Chemistry (and subsequently in later editions of The Chemical Bond).
The periodic table below is scanned in from Pauling's The Nature of The Chemical Bond, 3rd ed., 1960:
| Year: 1949 | PT id = 276, Type = formulation spiral |
Clark's Periodic Arrangement of The Elements
Origionally developed in 1933, the colour version of Clark's arrangement was used the the May 1949 edition of Life Magazine, part of a 16 page feature on the atom.
This periodic table formulation was the model for Longman's 1951 Festival of Britain mural. Information supplied by Philip Stewart.
| Year: 1949 | PT id = 295, Type = formulation 3D |
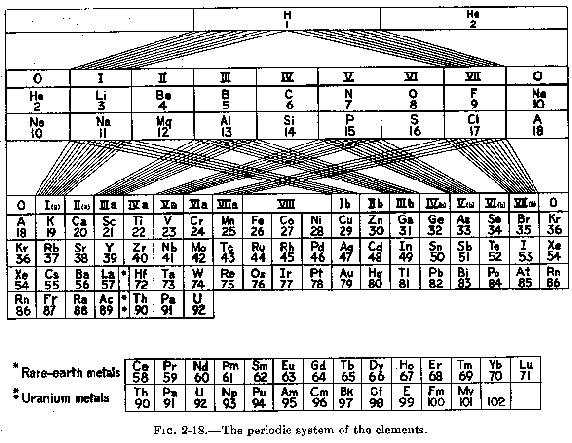
Wrigley's Lamina System
In two papers: A.N. Wrigley, W.C. Mast, and T.P. McCutcheon, "A Laminar Form of the Periodic Table, part 1," Journal of Chemical Education 26 (1949): 216-218 and A.N. Wrigley, W.C. Mast, and T.P. McCutcheon, "A Laminar Form of the Periodic Table, part 2: Theoretical Development, and Modifications," Journal of Chemical Education 26 (1949): 248-250 a Laminar Periodic Table is introduced. (Thanks to Ann E. Robinson for this informaton & references.)

This formulation was discussed and re-drawn by van Spronsen in 1969:
There is a Russian publication "100 Years of Periodic Law of Chemical Elements", Nauka 1969. On page 87 there is a formulation that appears to be a version of the van Spronsen re-drawing. The caption says: "Volumetric Model of 18-period Long System of D.I.Mendeleev." after Riggli (1949). (Thanks to Larry T for this.)
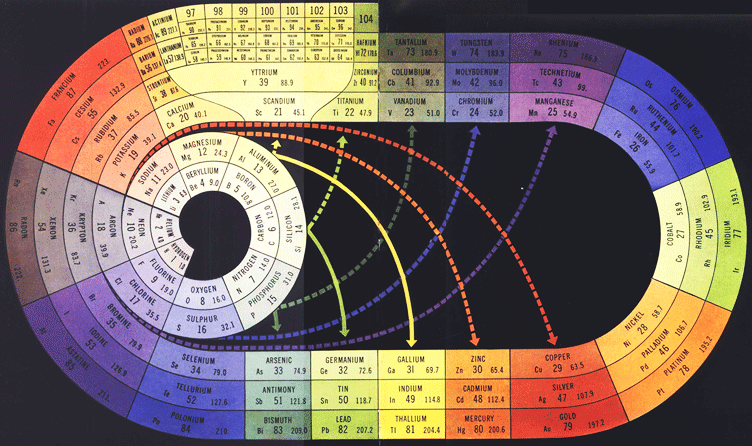
| Year: 1949 | PT id = 698, Type = formulation |
Antropoff's Representation of the Periodic System Revised by Fritz Scheele
Andreas von Antropoff's 1926 representation of the Periodic System, revised by Fritz Scheele in 1949, to include the lanthanides and actinides.
The table was reconstituted, using von Antropoff s colour scheme, by P J Stewart, November 2007:
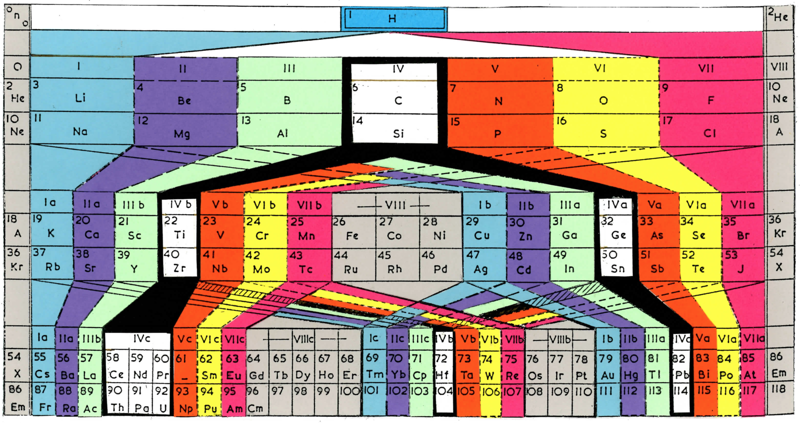
Thanks to Philip Stewart for the tip!
| Year: 1949 | PT id = 877, Type = element |
Discovery of Berkelium
Bk ![]()
Berkelium, atomic number 97, has a mass of 247 au.
Synthetic radioactive element.
Berkelium was first observed in 1949 by G. Thompson, A. Ghiorso and G. T. Seaborg.
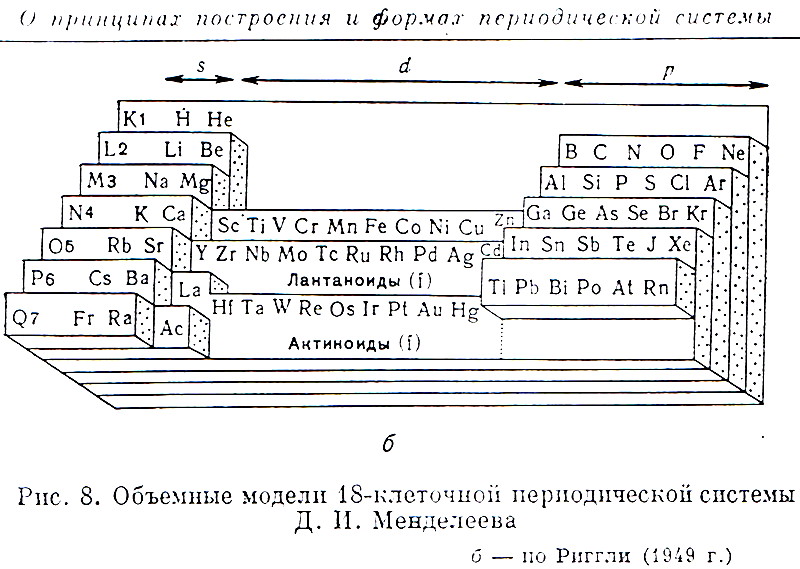
| Year: 1949 | PT id = 921, Type = formulation 3D |
Riggli's Volumetric Model of the Periodic Table
From the Russian Book "100 Years of Periodic Law of Chemical Elements", Nauka 1969, p.87.
The caption says: "Volumetric Model of 18-period Long System of D.I.Mendeleev." after Riggli (1949).

Thanks to Larry T for the tip!
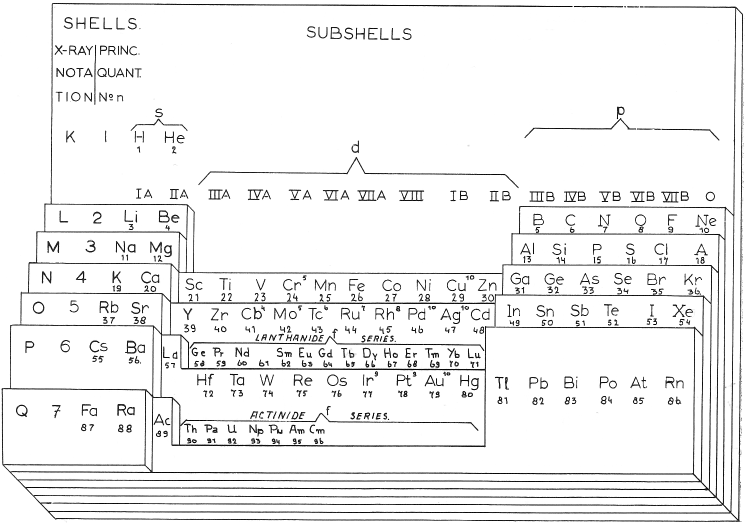
| Year: 1949 | PT id = 1018, Type = formulation 3D |
Scherer's Student Model of Spiral Periodic Chart
George A. Scherer, New Aids for Teaching the Periodic Law, School Science and Mathematics, vol. 49, no. 2 (1949).
René Vernon writes:
"This is a Left-Step periodic table with a split d-block, that can be rearranged into a cylinder. Students were expected to keep a copy of the two halves of the table in their note books, for reassembly as required. It was a clever way of introducing the 32-column form, and the transition from 2D to 3D (that faded into obscurity)":
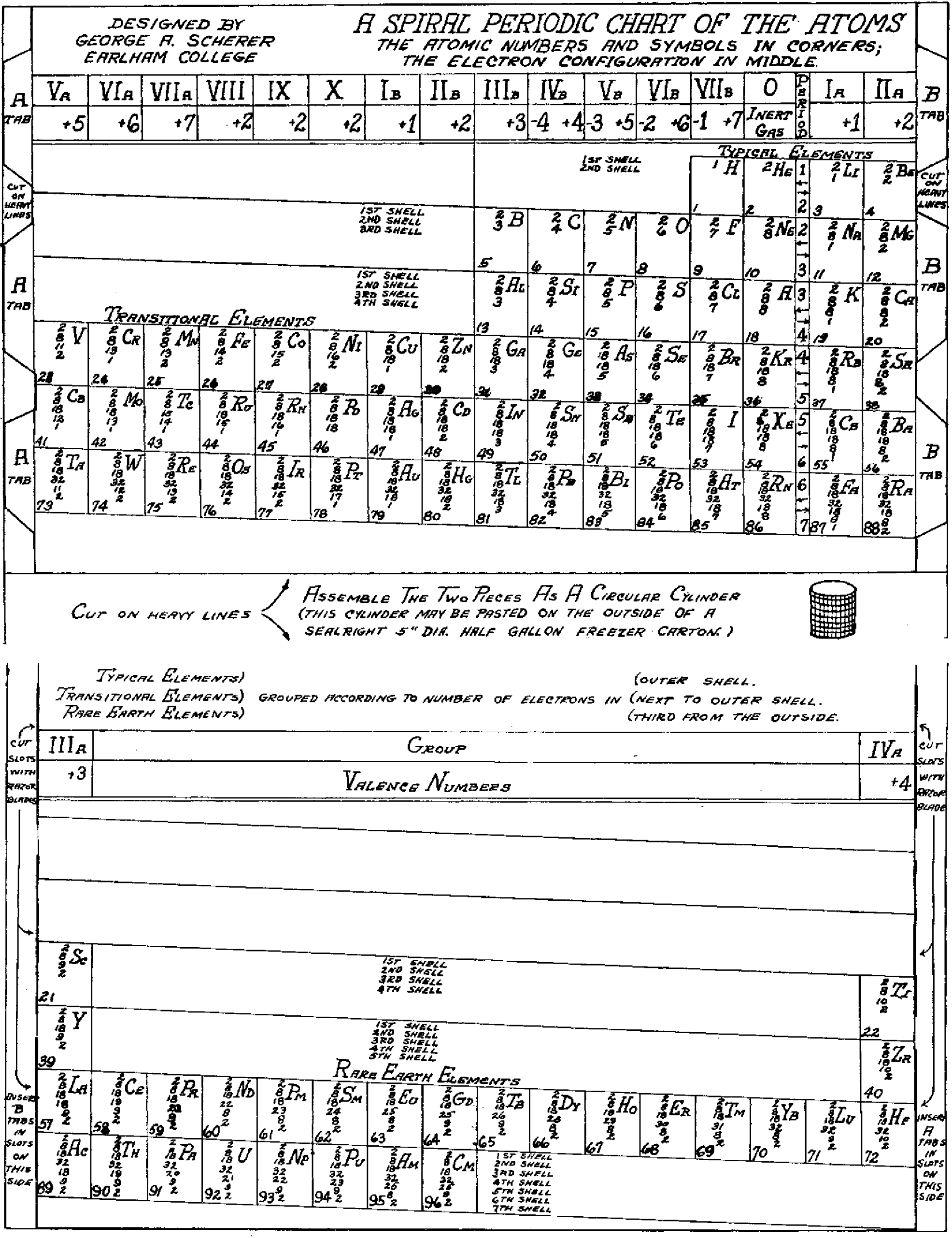

Thanks to René for the tip!
| Year: 1949 | PT id = 1052, Type = formulation |
Catalán's Periodic System/Sistema Periodico Ampliado
Two versions of Catalán's Periodic System/Sistema Periodico Ampliado. The first from Moore, Charlotte E., Atomic Energy Levels, National Bureau of Standards, Circular no. 467 (Washington, D. C.: U. S. Government Printing Office, 1949), vol. 1, Table 25. and the second as referenced here: http://www.miguelcatalan.net/pdfs/bibliografia/biblio09.pdf.
René Vernon, who provided the graphics, writes:
"I feel the footnote along the base of the first table could merit better attention being drawn to it. It says:
This arrangement is by Catalán. The electrons indicated in column two that are connected by braces have approximately the same binding energy. Consequently, for some elements one type of electron is preferred over another in the normal configuration, as for example, Cr, Cb, Pd, La, Ac, Th.
"The connecting braces hone in on the source of much of the controversy concerning notions of an ideal, optimal, better, this or that, or fundamental periodic table. I can't recall seeing a table with such a feature. For the second table, turning it on its side (attached) reminds of the ADOMAH [formulation].
Click on the images to enlarge:
Thanks to René for the tip!
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
© Mark R. Leach Ph.D. 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com
This free, open access web book is an ongoing project and your input is appreciated.

