Periodic Table |
 |
 |
 |
 |
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
The INTERNET Database of Periodic Tables
There are thousands of periodic tables in web space, but this is the only comprehensive database of periodic tables & periodic system formulations. If you know of an interesting periodic table that is missing, please contact the database curator: Mark R. Leach Ph.D. The database holds information on periodic tables, the discovery of the elements, the elucidation of atomic weights and the discovery of atomic structure (and much, much more).
Periodic Tables from the year 1951 :
| Year: 1951 | PT id = 24, Type = formulation spiral |
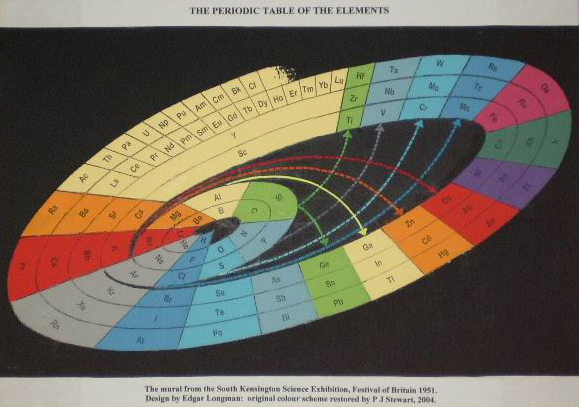
Longman's Mural from Festival of Britain
Edgar Longman's mural from the 1951 Festival of Britain Science Exhibition, restored by Philip Stewart:

| Year: 1951 | PT id = 461, Type = formulation |
Mellor's Periodic Series of the Elements
Mellor's periodic series of the elements lists the rare earths as a vertical column below Y in Group III. Element Z=61, is identified as Il, illinium.
The peculiarities of Ce, Eu, Tb and Yb are not evident. U is positioned below W emphasizing its 6+ oxidation state.
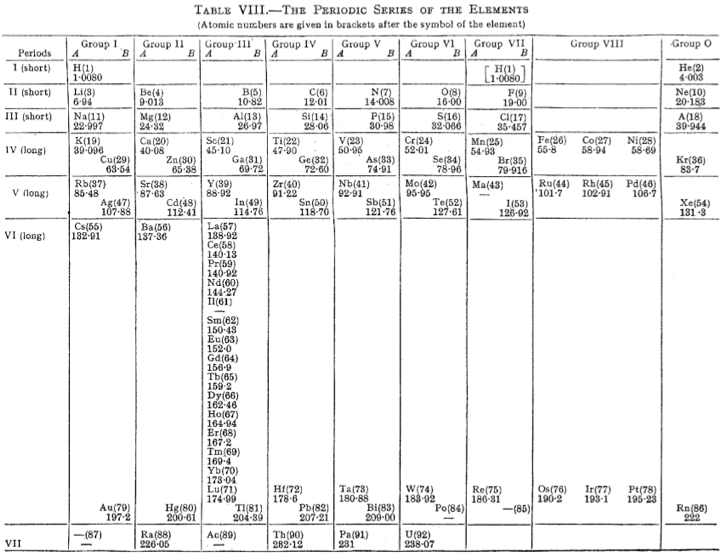
From Michael Laing's paper: A Revised Periodic Table with the Lanthanides Repositioned, Found. Chem. (2005) 7: 203-233
| Year: 1951 | PT id = 677, Type = formulation |
Tomkeieff's Periodic Table Formulation Formula
A short letter to Nature in which Tomkeieff gives a formula to generate the periodic table:

Thanks to Eric Scerri for the tip!
See the website EricScerri.com and Eric's Twitter Feed
| Year: 1951 | PT id = 1245, Type = formulation |
Friend's Updated Periodic Table
René Vernon writes:
"This 1951 table succeeds Friend’s table of 1926. Notice how Pu, Am, and Cm have been assigned to group VIII. The splitting of the Ln across two periods is bizarre."
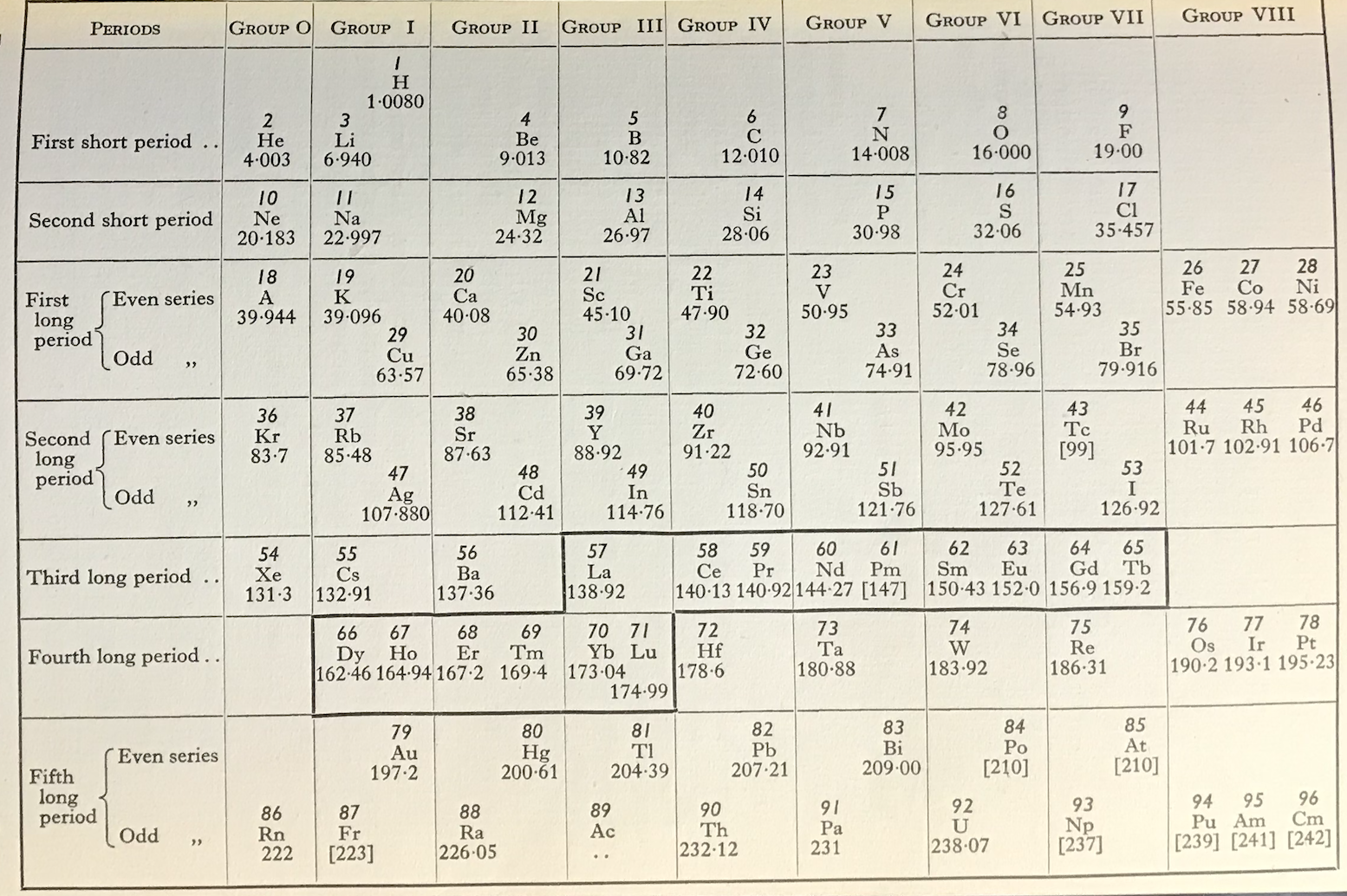
| Year: 1951 | PT id = 1272, Type = formulation |
Spedding's Rare Earths Periodic Table
Ref: Spedding FH 1951 The Rare Earths, Scientific American, vol. 185, no. 5, pp. 26–31
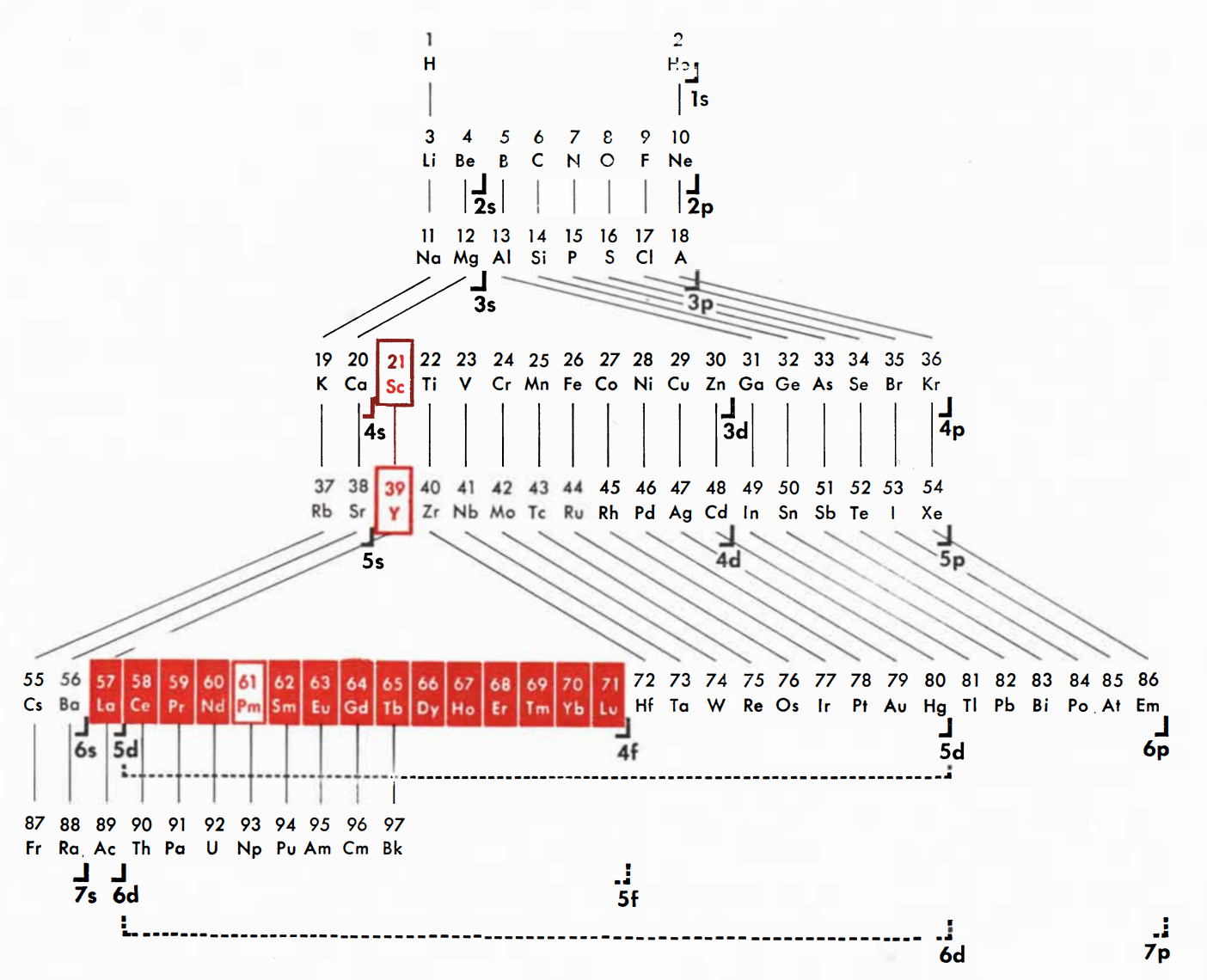
Thanks to René for the tip!
| Year: 1951 | PT id = 1324, Type = formulation |
Kapustinsky's Structure of The System of Elements
René Vernon writes:
Kapustinsky AF 1951, Structure of the periodic table of chemical elements (in Russian), Proceedings of the USSR Academy of Sciences, vol. 81, no. 1, pp. 47–50
Below the title Kapustinsky gives two equations that he says determine the structure of the system:
- The equation of periods Š = 4 x n2;
- The equation of cycles/dyads S = 2(n21 + n22).
The legend at the bottom left is:
- Period
- Cycle
- Number of elements
Kapustinsky refers to the periodic system of elements in terms of its emergence (proto-elements), formation (typical elements), and disintegration (synthetic elements). Kapustinsky refers to e, n, H, He as "proto-elements".
The electron and the neutron are not chemical elements but are elements in the sense of each being a rudiment, which means a beginning; an initial or imperfect form or stage. As Kapustinsky says, the properties of ordinary elements are not yet associated with them.
H and He can be considered "proto-elements" in the sense that they were the first building blocks from which heavier elements were later formed through nucleosynthesis in stars. Kapustinsky says that the system is thus:
- a harmonious whole, with a beginning and end
- regular and symmetrical
- without exception, it embraces all of its constituent members with simple mathematical expressions

 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
© Mark R. Leach Ph.D. 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com
This free, open access web book is an ongoing project and your input is appreciated.