Periodic Table |
 |
 |
 |
 |
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
The INTERNET Database of Periodic Tables
There are thousands of periodic tables in web space, but this is the only comprehensive database of periodic tables & periodic system formulations. If you know of an interesting periodic table that is missing, please contact the database curator: Mark R. Leach Ph.D. The database holds information on periodic tables, the discovery of the elements, the elucidation of atomic weights and the discovery of atomic structure (and much, much more).
Periodic Tables from the year 1994 :
| 1994 | Where Should Aluminium Go? |
| 1994 | Darmstadtium, Discovery of |
| 1994 | Roentgenium, Discovery of |
| 1994 | f-Block Elements 3D Periodic Table |
| 1994 | Treplow's Periodic Table of The Atoms |
| Year: 1994 | PT id = 18, Type = formulation |
Fathi Habashi aruges in Chemistry in Education (1994) that aluminium, Al, should be placed above scandium and next to magnesium. There is more information about this formulation here:
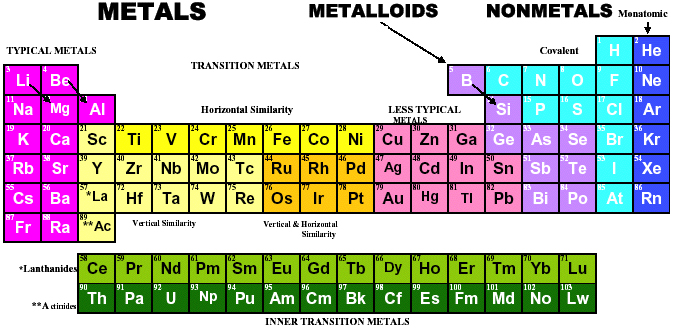
| Year: 1994 | PT id = 890, Type = element |
Discovery of Darmstadtium
Ds ![]()
Darmstadtium, atomic number 110, has a mass of 281 au.
Synthetic radioactive element.
Darmstadtium was first observed in 1994 by S. Hofmann et al.
| Year: 1994 | PT id = 891, Type = element |
Discovery of Roentgenium
Rg ![]()
Roentgentium, atomic number 111, has a mass of 280 au.
Synthetic radioactive element.
Roentgenium was first observed in 1994 by S. Hofmann et al.
| Year: 1994 | PT id = 1016, Type = formulation 3D |
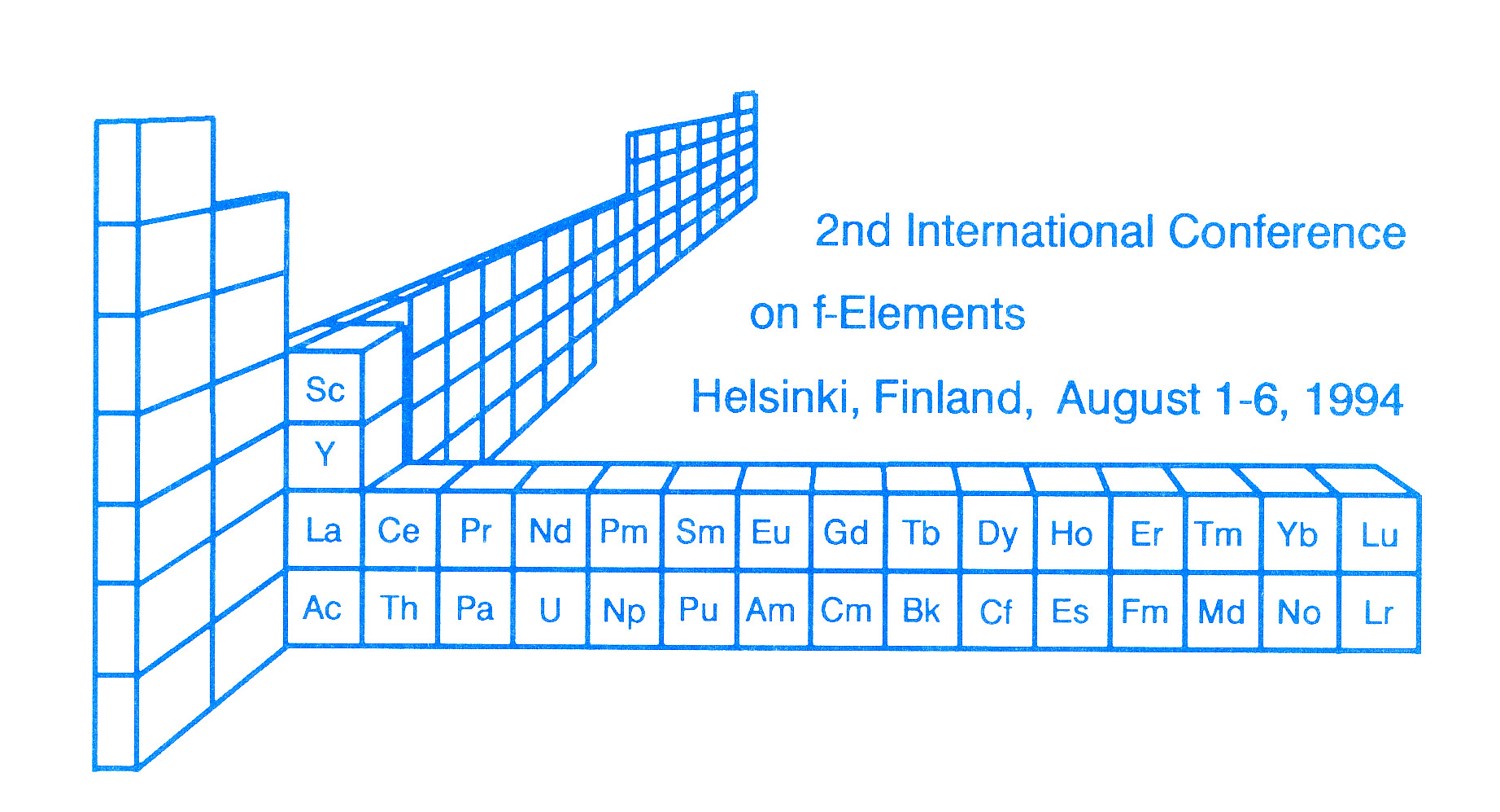
f-Block Elements 3D Periodic Table
From conference in Helsinki on the f-Block Elements to commemorate the bicentennial of Johan Gadolin's 1794 analysis of Yittria.
Pekka Pykkö writes to say:
"We used [this formulation] in Helsinki in 1994 on the cover of ICFE-2 conference proceedings. Who invented it or where it was copied from, I do not know. Anyway, all the hundreds of participants received it from us":
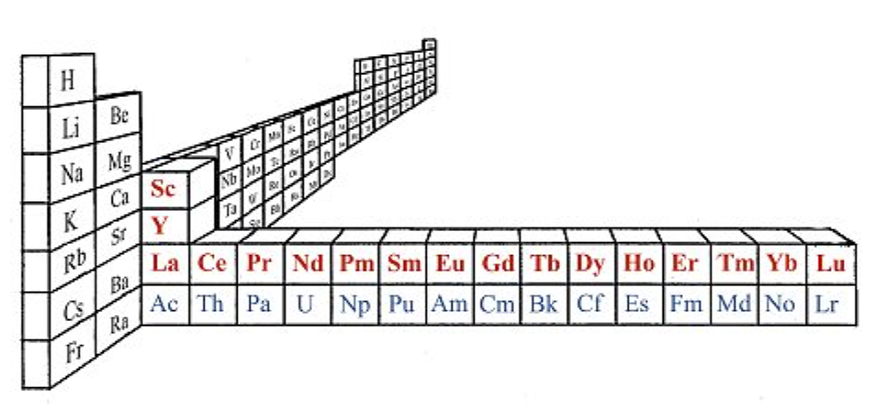
Claude Piguet's paper, Chimia 73 (2019) 165–172, also uses this 3D version of the standard periodic table. The text says: "Periodic table highlighting the location of Rare Earths (red elements). The elements shown in blue correspond to the actinide series":
Thanks to Eric Scerri for the tip!
See the website EricScerri.com and Eric's Twitter Feed.
| Year: 1994 | PT id = 1159, Type = formulation |
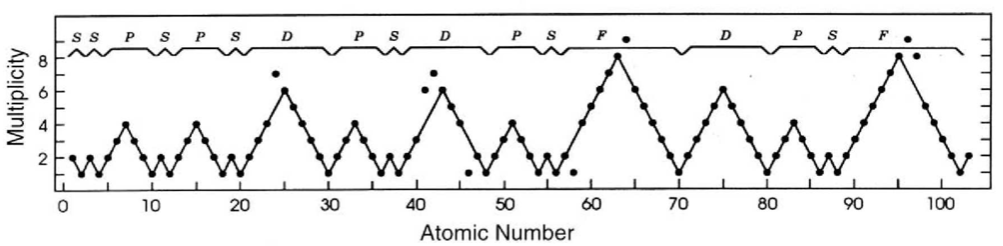
Treplow's Periodic Table of The Atoms
R.S. Treplow, J. Chem. Educ. 1994, 71, 12, 1007: The Periodic Table of Atoms: Arranging the Elements by a Different Set of Rules.
"Although periodic tables differ greatly in their appearance, examination shows they are all designed according to a common set of conventions. This paper reviews those conventions and asks how the table would look under a different set of rules."
Ground-state multiplicity vs. atomic number for elements 1 to 103. Subblocks are labeled S, P, D & F. Lines connecting the dots show the "ideal" pattern. Atoms not on the lines are "nonideal" (where ideal refers to Madelung's rule):
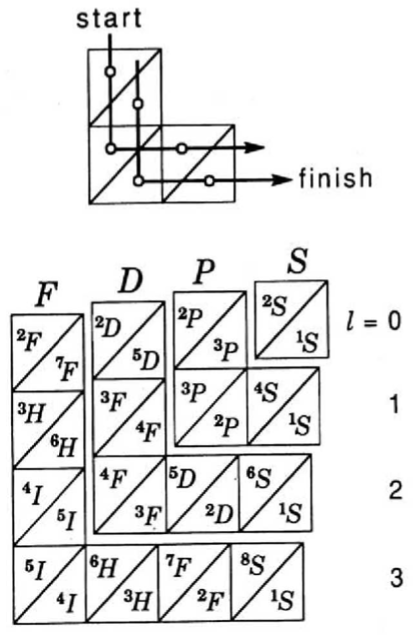
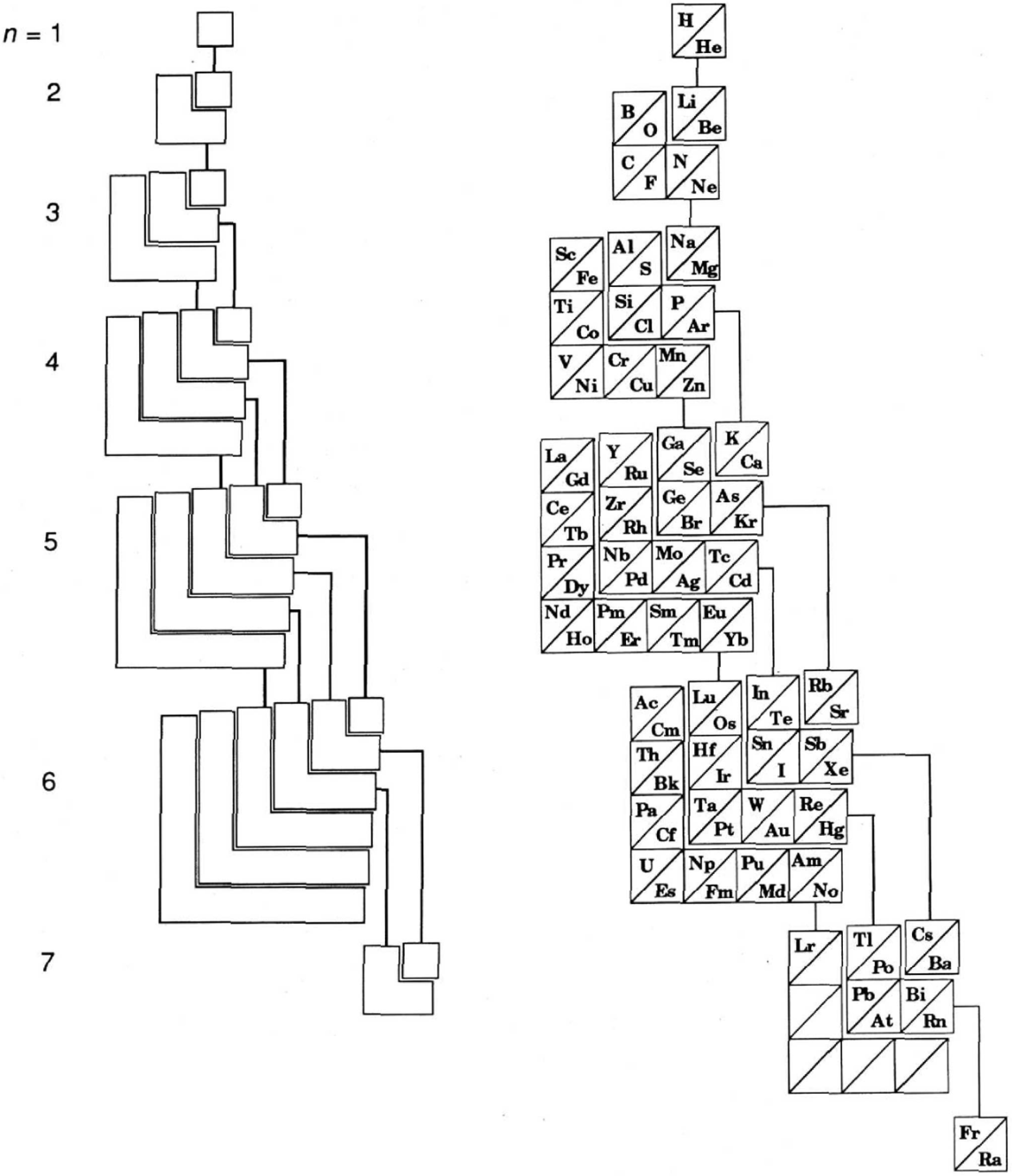
Thanks to René Vernon for his help.
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
© Mark R. Leach Ph.D. 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com
This free, open access web book is an ongoing project and your input is appreciated.