Periodic Table |
 |
 |
 |
 |
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
The INTERNET Database of Periodic Tables
There are thousands of periodic tables in web space, but this is the only comprehensive database of periodic tables & periodic system formulations. If you know of an interesting periodic table that is missing, please contact the database curator: Mark R. Leach Ph.D. The database holds information on periodic tables, the discovery of the elements, the elucidation of atomic weights and the discovery of atomic structure (and much, much more).
Periodic Tables from the year 2001 :
| Year: 2001 | PT id = 34, Type = formulation |
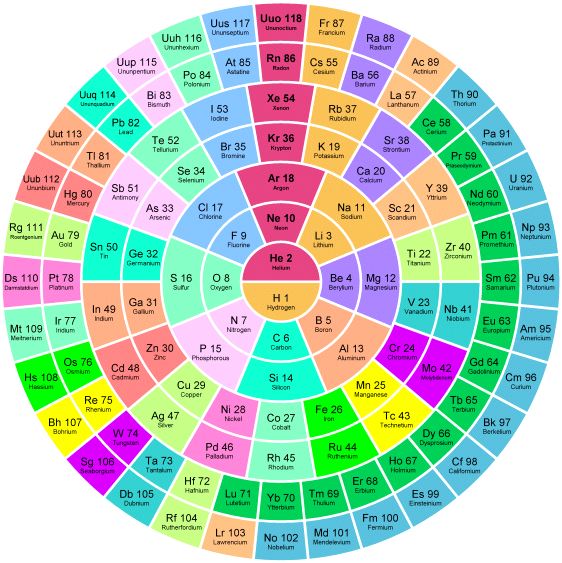
The Mayan Periodic Table
The Mayan Periodic Table of Elements, named for its similarity to the ancient Mesoamerican calendar, is based on electron shells. The shells are shown as concentric circles. Each row in the tabular form is shown as a ring.
Read more and buy the poster and T-Shirts at MayanPeriodic.com.
| Year: 2001 | PT id = 43, Type = formulation |
Vertical Periodic Table
A vertical periodic table from apsidium.com:
| Year: 2001 | PT id = 57, Type = misc |
| Year: 2001 | PT id = 97, Type = non-chem |
Desserts
| Year: 2001 | PT id = 138, Type = data |
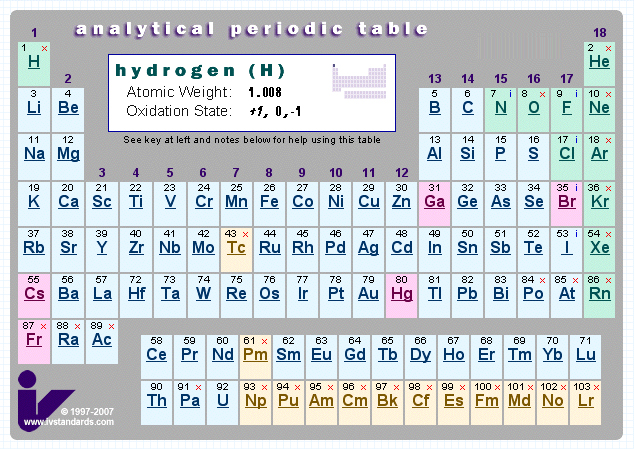
Analytical Chemist's Periodic Table
This PT gives information about storage and analysis of the elements.
| Year: 2001 | PT id = 336, Type = misc non-chem |
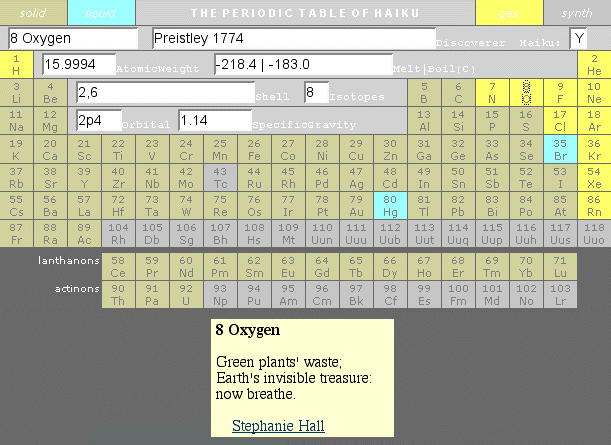
Haiku Periodic Table
The excellent Periodic Table of Haiku has re-emerged from the 'Way Back" web-archive website.
A second 2009/10 Periodic Table of Haiku, from the University of Minnesota is available here.
| Year: 2001 | PT id = 353, Type = formulation |
Muradjan's Universal Periodic System
Muradjan's Universal Periodic System is a variation of the Janet formulation, however, it is worth visiting the web page and scrolling down as there is much interesting material:
| Year: 2001 | PT id = 511, Type = formulation spiral 3D |
ElemenTouch Periodic Table
Yoshiteru MAENO writes:
"I am a Physics Prof. at Kyoto University, Japan. My field of study is experimental superconductivity. I recently found the work by Schaltenbrand in 1920 on your website. One might say that Elementouch is a re-invention of Schaltenbrand's, but by arranging the element names helically on three cylinders, its usefulness has been improved":
| Year: 2001 | PT id = 555, Type = misc |
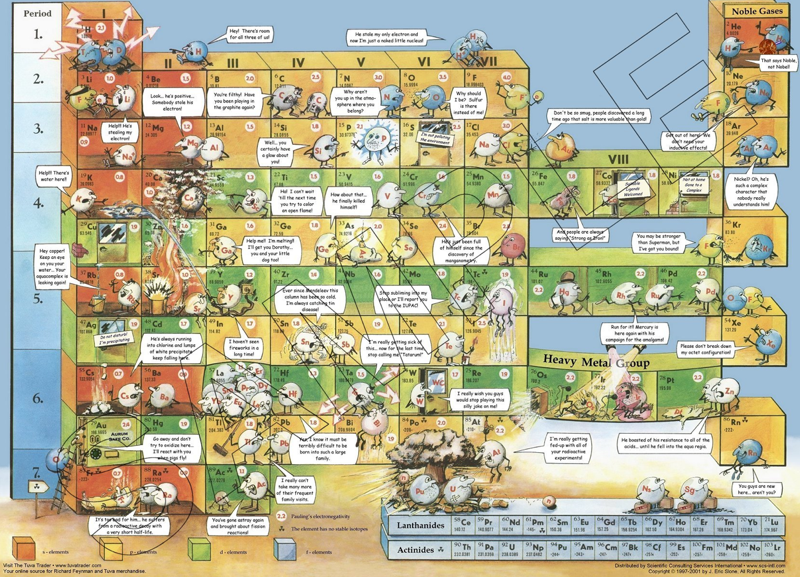
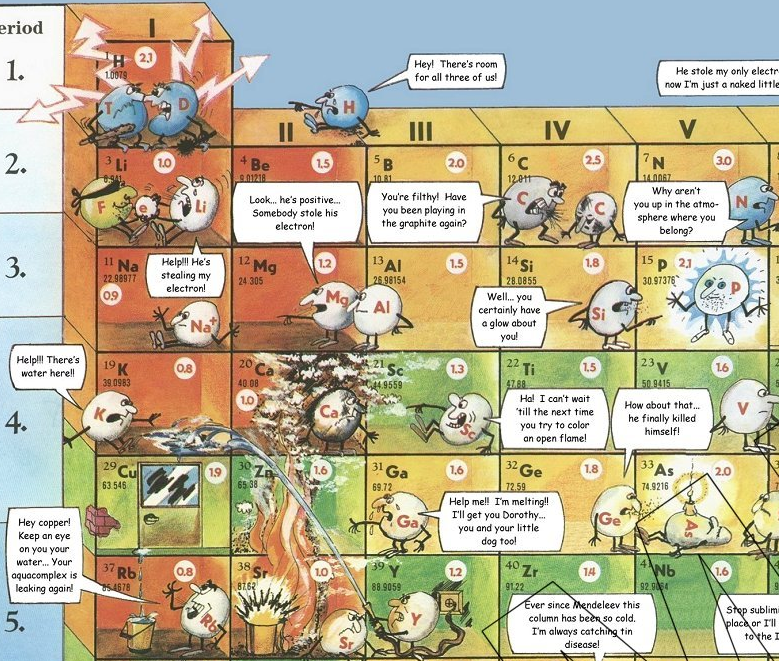
Funny Periodic Table
By Eric J Stone a Funny Periodic Table of chemical reactivity.
"This periodic table is unique -- it is informational, educational, and humorous at the same time. Arranged in the standard Mendeleev layout, this table depicts the elements interacting with each other in many interesting ways. The jokes are designed to impart useful information within the context of humor. Ideal for science buffs of all ages -- this is truly the periodic table for the masses. It can be appreciated by children and professionals alike. Children especially like the table, which draws them in with its funny vignettes. This poster is based on the original art of Slavomir Koys. The poster makes a great promotional item. Use it to promote your schools chemistry club or as science fair prizes":
| Year: 2001 | PT id = 1025, Type = data formulation review |
Wikipedia Periodic Table
The Wikipedia Periodic Table pages are astonishing, giving hyper-linked data about:
- Formulations
- History
- Discovery
- Elements
- Isotopes
- Personalities
- Compounds
- etc.
| Year: 2001 | PT id = 1062, Type = formulation |
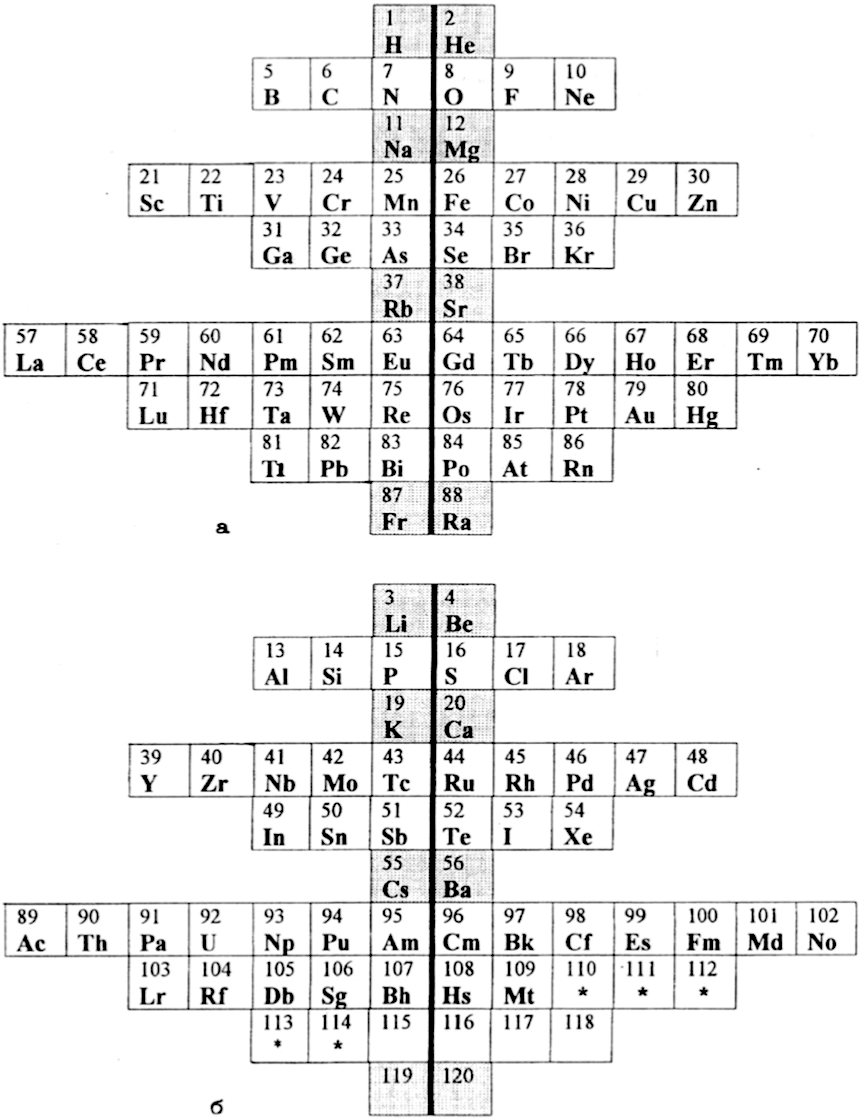
Gorbunov and Filippov's Doubled Periodic Table
Gorbunov, A. I., Filippov, G. G.: Fine Structure of D. I. Mendeleev Periodic Table: secondary periodicity, early and late elements. Khim-ya Tekhnol. 11, 43–45 (2001). (in Russian)
Naum S. Imyanitov (Foundations of Chemistry) writes:
"The two-table design is of particular interest. Atoms with odd n+l are located in the upper table, and the ones with even n+l are placed in the bottom table (Tables 5). The elements are divided by a vertical line of symmetry to the early and late ones both in the upper and bottom tables. The advantage of Tables 5 is a clear demarcation into subsets, with each subset having its own separate place in the table. The drawback is directly related to this advantage: this table does not reflect the similarity between members of different subsets."
| Year: 2001 | PT id = 1322, Type = review misc formulation |
Oliver Sacks, Uncle Tungsten: Memories of Tungsten of a Chemical Beyond
René Vernon writes:
On the paperback cover of Oliver Sack's Uncle Tungsten (below) the periodic table shows a 16–wide set of elements at its base. This is quite unusual since this set is normally shown as being 15— or 14— elements wide. See, for example, the table found on the site of the International Union of Pure & Applied Chemistry which shows a 15–wide set of elements at its base.
It looks like the second pair are La and Ac, but what then are two immediately preceding elements?
I suspect they are probably the alkaline earth metals, Ba and Ra. This may be an homage to Mr Rare Earth^ aka Karl A. Gschneidner Jr (1930–2016), who wrote that:
...since Ba has a 4f06s2 configuration, these three elements are the first (Ba), mid (Eu), and end (Yb) members of the divalent 4f transition series.
The notion of 4f0 is not unprecedented; the IUPAC periodic table, with its 15-wide f-block presumably implies La as 4f0 5d1 6s2.
There is some good chemistry going on here, given the pronounced similarities between Ba and the lanthanides, and the alkaline earth metals generally with about 20 properties involved:
- Most of the physical properties of Eu and Yb, "such as the atomic volumes, metallic radii, melting and boiling points, heats of sublimation, compressibilities, and coefficients of expansion are more like those of the alkaline-earth metals, Ca, Sr, and Ba, than those of the rare-earth metals" (Pauling 1960, p. 418; Gschneidner 1964, p. 286).
- Liquid ammonia dissolves certain alkali, alkaline earth, and Ln metals, and... combines with them to form solid compounds. Those metals whose compound-forming ability has been confirmed are Li, Ca, Sr, Ba, Eu and Yb. (Mammano (1970, p. 367)
- The lanthanides are sometimes regarded as trivalent versions of the alkaline earth metals (Evans 1982).
- The electron configurations of lanthanide cations are similar to those of alkaline earth metal cations, as the inner f- orbitals are largely or completely unavailable for bond formation; (Choppin & Rizkalla 1994)
- The lanthanide trivalent cations are essentially spherical and present an environment very similar to alkali and alkaline earth ions towards complex formation... the standard electrode potentials for the lanthanides have similar values and are comparable with the redox potentials of alkaline earth metals (Sastri et al. 2003)
- Ba-Eu-Yb have cubic crystalline structures whereas the rest of the Ln are hexagonal, or rhombohedral in the case of Sm (Russell & Lee 2005)
- There is a close alloying similarity between the lanthanides and Ca, Sr and Ba (Artini 2007)
- Lanthanides are effective mimics of calcium and can stimulate or inhibit the function of calcium-binding proteins (Brayshaw 2019)
- Lanthanide cations can substitute for Ca2+ and Sr2+ cations in host materials for solid state lasers (Ikesue 2013)
- There is a knight’s move relationship between Ca and La:
- The ionic radius of Ca2+ is 114 pm; that of La3+ is 117 pm
- The similarity in sizes means La3+ will compete with Ca2+ in the human body, and usually win on account of having a higher valence for roughly the same hydrated radius
- The basicity of La2O3 is almost on par with CaO2 Freshly prepared La2O3 added to water reacts with such vigour that it can be quenched like burnt lime (CaO)
- The electronegativity of Ca is 1.0; that of La is 1.1.
Kudos to Oliver.
^Pecharsky 2016
Sources
- Artini C (ed.) 2017, Alloys and Intermetallic Compounds: From Modeling to Engineering, CRC Press, Boca Raton, p. 92
- Brayshaw et al. 2019, Lanthanides compete with calcium for binding to cadherins and inhibit cadherin-mediated cell adhesion, Metallomics, vol. 11, no. 5, 2019, pp. 914–924
- Choppin GR & Rizkalla EN 1994, Solution chemistry of actinides and lanthanides, Handbook on the Physics and Chemistry of Rare Earths, pp. 559–590(560)
- Evans WJ 1982, Recent advances in the low valent approach to f-element organometallic chemistry, in McCarthy GJ, Silber HB and Rhyne JJ (eds), The Rare Earths in Modern Science and Technology, vol. 3, Plenum Press, New York, pp. 61–70(62)
- Gschneidner KA 1965, in Seitz F & Turnbull D (eds), Solid State Physics, vol. 16, Academic Press, New York, p. 286
- Ikesue A, Aung YL, Lupei V 2013, Ceramic Lasers, Cambridge University Press, Cambridge, pp. 26, 28
- Mammano N 1970, Solid metal ammonia compounds, in Metal–Ammonia Solutions, Proceedings of an International Conference on the Nature of Metal–Ammonia Solutions: Colloque Weyl II, pp. 367-393 (367), https://doi.org/10.1016/B978-0-408-70122-8.50030-4
- Pauling L 1960, The Nature of the Chemical Bond, 3rd ed., Cornell University Press, Ithaca, p. 418
- Pecharsky V 2016, Karl A. Gschneidner Jr (1930–2016), Nature Materials, vol. 15, no. 1059, https://doi.org/10.1038/nmat4751
- Russell AM & Lee KL 2005, Structure-property relations in nonferrous metals, John Wiley & Sons, Hoboken, inside cover
- Sastri et al. 2003, Modern Aspects of Rare Earths and their Complexes, Elsevier, Amsterdam, pp. 377, 878

 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
© Mark R. Leach Ph.D. 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com
This free, open access web book is an ongoing project and your input is appreciated.