Periodic Table |
 |
 |
 |
 |
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
The INTERNET Database of Periodic Tables
There are thousands of periodic tables in web space, but this is the only comprehensive database of periodic tables & periodic system formulations. If you know of an interesting periodic table that is missing, please contact the database curator: Mark R. Leach Ph.D. The database holds information on periodic tables, the discovery of the elements, the elucidation of atomic weights and the discovery of atomic structure (and much, much more).
Periodic Tables from the year 2005:
| Year: 2005 | PT id = 28, Type = formulation |
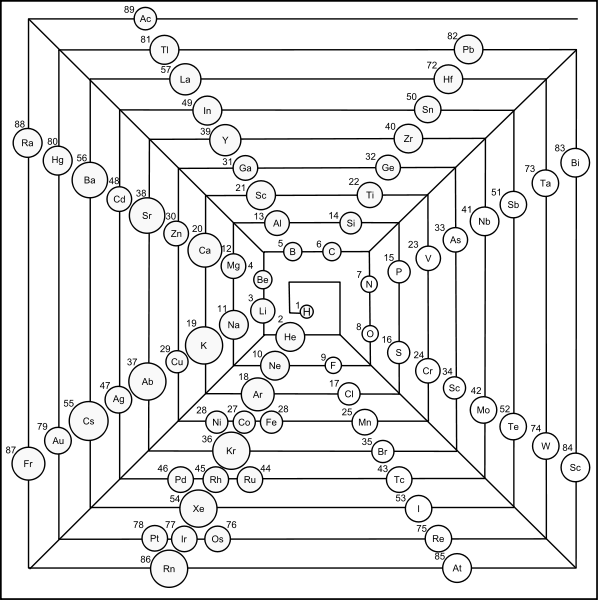
Laing's Revised Periodic Table with the Lanthanides Repositioned
Michael Laing's "Revised Periodic Table with the Lanthanides Repositioned", from Foundations of Chemistry 7:203-233.
- The new system emphasizes oxidation states.
- The elements Eu and Yb fall directly below Ba, indicating the common oxidation state of 2+.
- Elements La, Gd, Lu fall in a column below Y, all having the dominant oxidation state 3+.
- Ce and Tb fall between Zr and Hf in keeping with their 4+ oxidation state. Pm (Z=61) lies immediately below Tc (Z=43), both without stable naturally occurring isotopes.
- The easily attained oxidation states of 2+ for Am and No, and 4+ for Bk are evident from the analogous positioning of the actinides.
Philip Stewart's modification of the Laing formulation:
Philip Stewart says (in a personal communication):
"It seems wrong to suggest an analogy between Pr to Sm and Dy to Tm with the V, Cr, Mn, Fe groups. I have pushed them to the right to suggest that those lanthanides are like the old group VIII (including the coinage metals); like them they cannot use all their outer electrons in bonding (with the exception of Ru viii and Os viii. I have treated the actinides differently to take account of Pa v and U vi. It's ability to lose the juxtaposition of Tc and Pm, but it is physical rather than chemical anyway."
| Year: 2005 | PT id = 37, Type = formulation spiral |
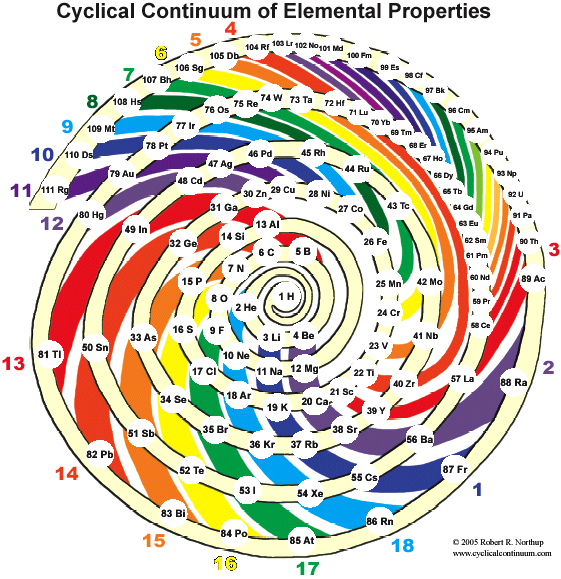
Cyclical Continuum of Elemental Properties by Robert R. Northup
The Cyclical Continuum of Elemental Properties Periodic Table by Robert R. Northup
"The Cyclical Continuum of Elemental Properties is a user-friendly teaching tool that is intended to accompany the Periodic Table of Elements. Hydrogen is shown at the center, atomic numbers and symbols form an unbroken spiral, and element groups 1 through 18 (noble gases, alkali metals, halogens, etc.) are displayed by colored arcs. Beginning chemistry students can visually see the continuity of atomic numbers in the Cyclical Continuum as a way to introduce and orient them to the Periodic Table. Advanced chemistry students can test their understanding of the Periodic Table's organization by applying that knowledge to interpretation of the Cyclical Continuum."
Read more and buy the poster at the Cyclical Continuum web site.
| Year: 2005 | PT id = 41, Type = formulation |
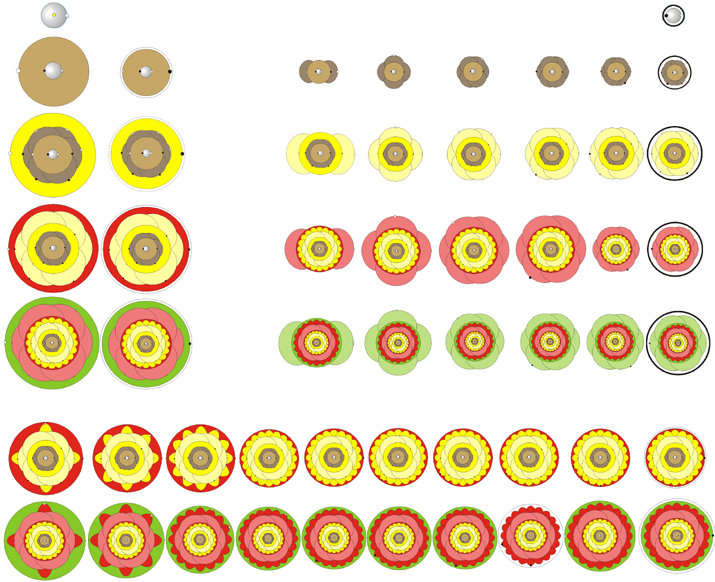
AtomFlowers by Boy Boer
A periodic table that gives a representation of the electron orbitals that look like flowers:

| Year: 2005 | PT id = 98, Type = non-chem |
Nerdiness
A chart in GQ, by Fred Woodward, on nerdiness:
| Year: 2005 | PT id = 100, Type = non-chem |
Cereal Typologies
A chart on cereal typologies published in 2wice:
| Year: 2005 | PT id = 127, Type = data |
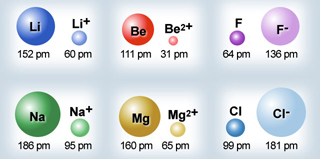
Atomic Radii Periodic Table

By Mark Leach
| Year: 2005 | PT id = 128, Type = data |
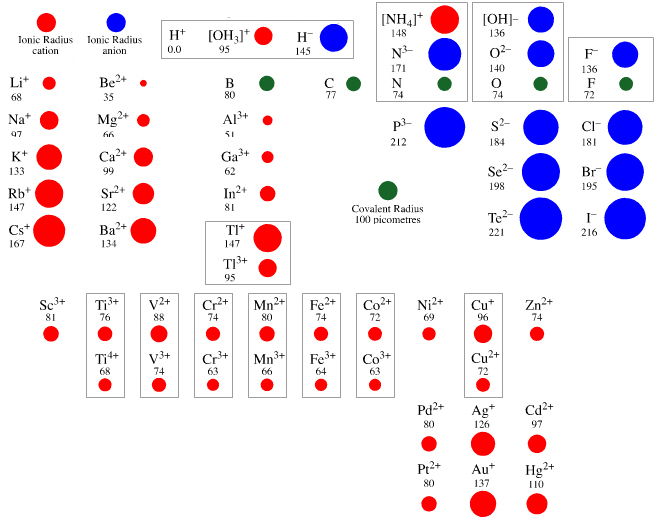
Ionic Radii Periodic Table
| Year: 2005 | PT id = 137, Type = data |
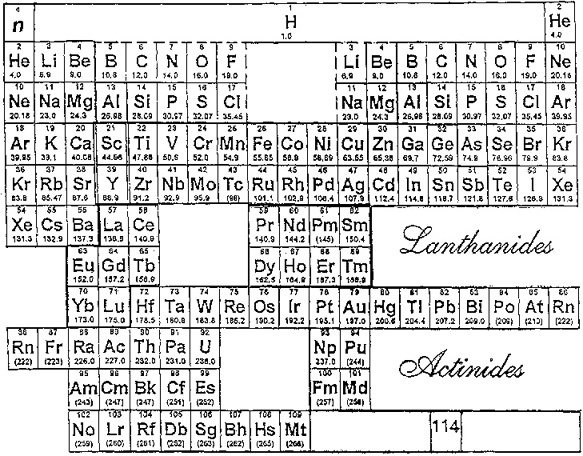
Extraction from Ore to Pure Element
A periodic table showing how pure elements are extracted:
Highly electropositive elements (Na, K) and electronegative elements (Cl2, F2) can only be obtained by electrolysis.
By Mark Leach
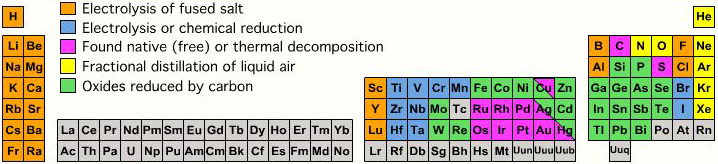
| Year: 2005 | PT id = 141, Type = data |
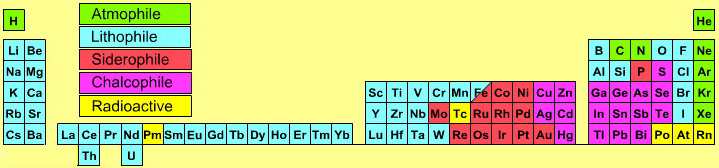
Atmophile Elements - noble gases and covalently bonded gaseous molecules. The atoms and molecules are attracted by weak van der Waals forces and so these elements remain gaseous at room temperature.
Lithophile Elements - Those elements which form ionic bonds generally have filled outer electron shells. They typically bond to oxygen in silicates and oxides.
Siderophile Elements - The metals near iron in the periodic table that exhibit metallic bonding, have a weak affinity for oxygen and sulfur and are readily soluble in molten iron. Examples include iron, nickel, cobalt, platinum, gold, tin, and tantalum. These elements are depleted in the earth crust because they have partitioned into the earth's iron core.
Chalcophile Elements - The elements that bond to S, Se, Te, Sb, and As. These bonds are predominantly covalent in character.
As discussed in more detail here.
By Mark Leach
| Year: 2005 | PT id = 146, Type = non-chem |
A periodic table of criminal elements:
| Year: 2005 | PT id = 151, Type = data |
Merck Periodic Table of The Elements
The Merck periodic table of the elements, here:
| Year: 2005 | PT id = 157, Type = data |
Student's Periodic Table
Students are expected to know that in all equations hydrogen is molecular should [nearly always] be written as H2. Likewise, nitrogen is N2, oxygen O2, fluorine F2, chlorine Cl2, bromine Br2 and iodine I2. But somehow students are expected to know that molecular sulfur, S8, should be written as S and molecular phosphorus, P4, should be written as P.
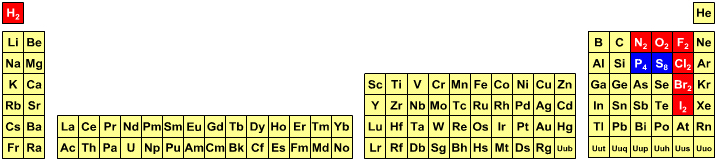
By Mark Leach
| Year: 2005 | PT id = 171, Type = data |
Minerals by Chemical Composition
Lists minerals by percent element. From the excellent webmineral mineralogy database:
| Year: 2005 | PT id = 184, Type = non-chem |
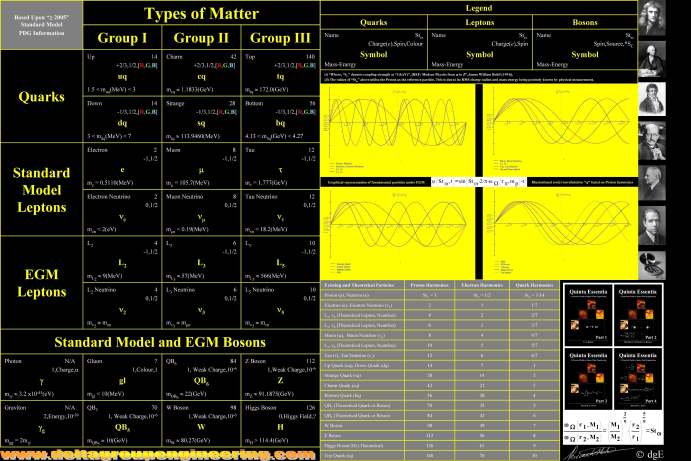
Fundamental Particles
A periodic table of Fundamental Particles. Click on the image to go to the website and access a 7000 x 5000 sized image:
| Year: 2005 | PT id = 191, Type = non-chem |
Artist's Periodic Table
“Periodic Table” found object assemblage and construction. Dimensions variable, by David Redfern:
| Year: 2005 | PT id = 213, Type = non-chem |
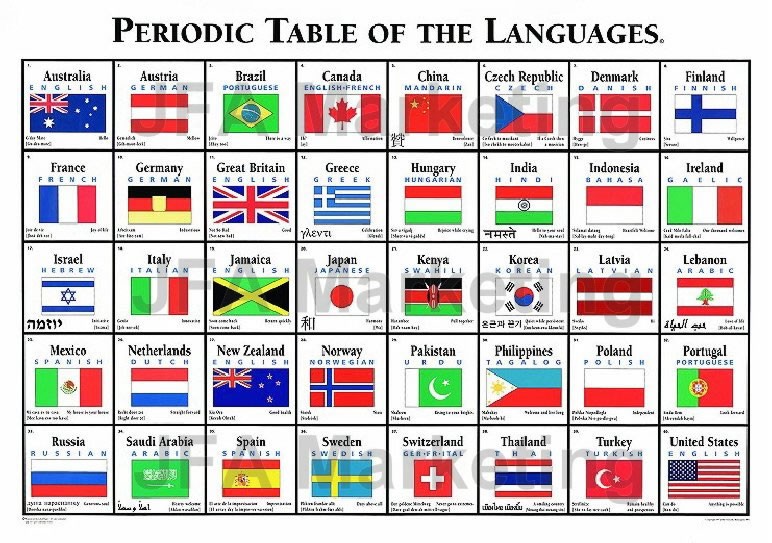
Languages
A periodic table of languages, here:
and for sale here:
| Year: 2005 | PT id = 214, Type = non-chem |
Money
A periodic table of money, here:
| Year: 2005 | PT id = 242, Type = formulation misc non-chem spiral |
Painting of The Elements
From Gabrielle David's website, here, a painting called Elements, inspired by Melinda Green's Periodic Fractal formulation of 1995:
- The tiniest ball in the center is hydrogen, the next helium, lithium, etc.
- Colors indicate the chemical group.
| Year: 2005 | PT id = 312, Type = data misc |
Smart Elements
Smart Elements, at smart-elements.com, is a company selling physical samples of chemical elements for research, education & collection.
- High purity Elements for Science, Laboratory and Education
- High-End element samples for collectors, museums, lectures and exhibitions
- Free picture service for educational purposes
- Professional advisory service
- Purchase of Elements
Smart Elements sell numerous examples of all the naturally occuring elements. For example they sell 26 copper, Cu, products including samples in acrylic blocks, vials and bottles:
| Year: 2005 | PT id = 347, Type = formulation spiral 3D |
Pyramid Format Periodic Table
From Wikipedia, this Pyramid Format Periodic Table is Based on a graphic from Scholten J."Secret Lanthanides", 2005, ISBN 90-74817-16-5;
| Year: 2005 | PT id = 467, Type = misc |
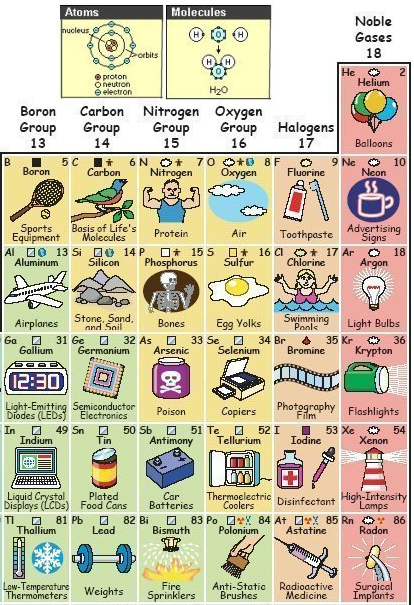
Pictures, Periodic Table of The Elements
By Keith Enevoldsen, a Periodic Table of The Elements in Pictures, and here.
| Year: 2005 | PT id = 634, Type = formulation |
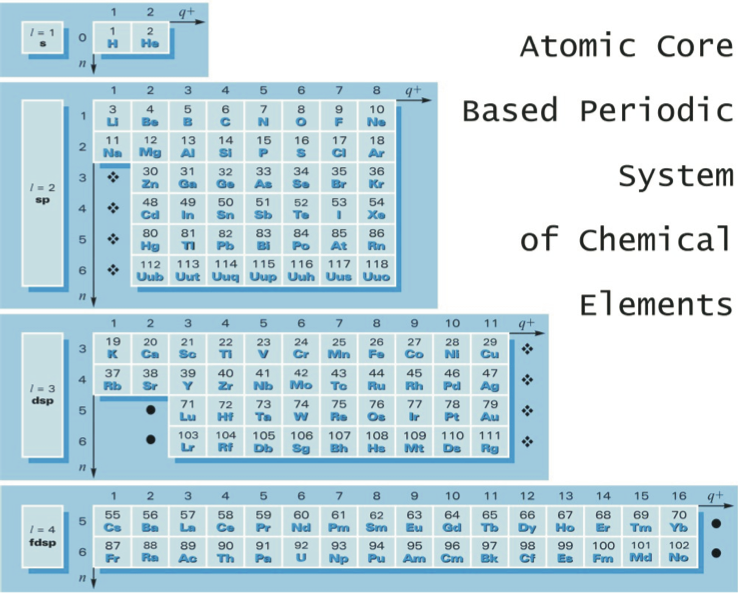
Górski's Atomic Core Based Periodic System
From Go?rski A., Pol. J. Chem., 79, 1435 (2005), an atomic core based periodic system of the chemical elements. In this version of the Periodic Table, He is placed close to H and simultaneously above Be (and not above Ne):
| Year: 2005 | PT id = 657, Type = data |
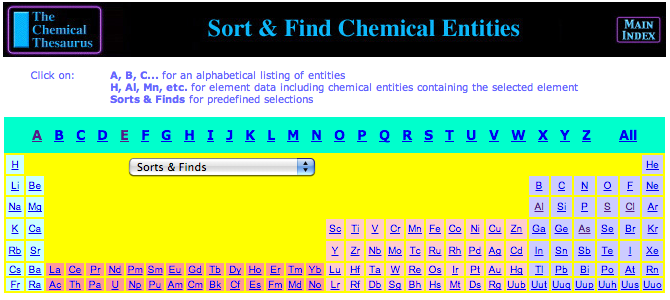
Chemical Thesaurus Reaction Chemistry Database Periodic Table
A periodic table front end to the Chemical Thesaurus Reaction Chemistry Database Periodic Table. Clicking on an element gives access to database searches of chemical species and their interactions.
A quote neatly sums up what the ChemThes reaction chemistry database project is trying to achieve:
"The Chemical Thesaurus is a reaction chemistry information system that extends traditional references by providing hyperlinks between related information. The program goes a long way toward meeting its ambitious goal of creating a nonlinear reference for reaction information. With its built-in connections, organizing themes, and multiple ways to sort and view data, The Chemical Thesaurus is much greater than the sum of the data in its database.
"The program does an excellent job of removing the artificial barriers between different subdisciplinary areas of chemistry by presenting a unified vision of inorganic and organic reaction chemistry."
By Mark Leach
| Year: 2005 | PT id = 1242, Type = formulation |
Rich's Periodic Chart Exposing Diagonal Relationships
Rich, R. L. (2005). Are Some Elements More Equal Than Others? Journal of Chemical Education, 82(12), 1761. doi:10.1021/ed082p1761.
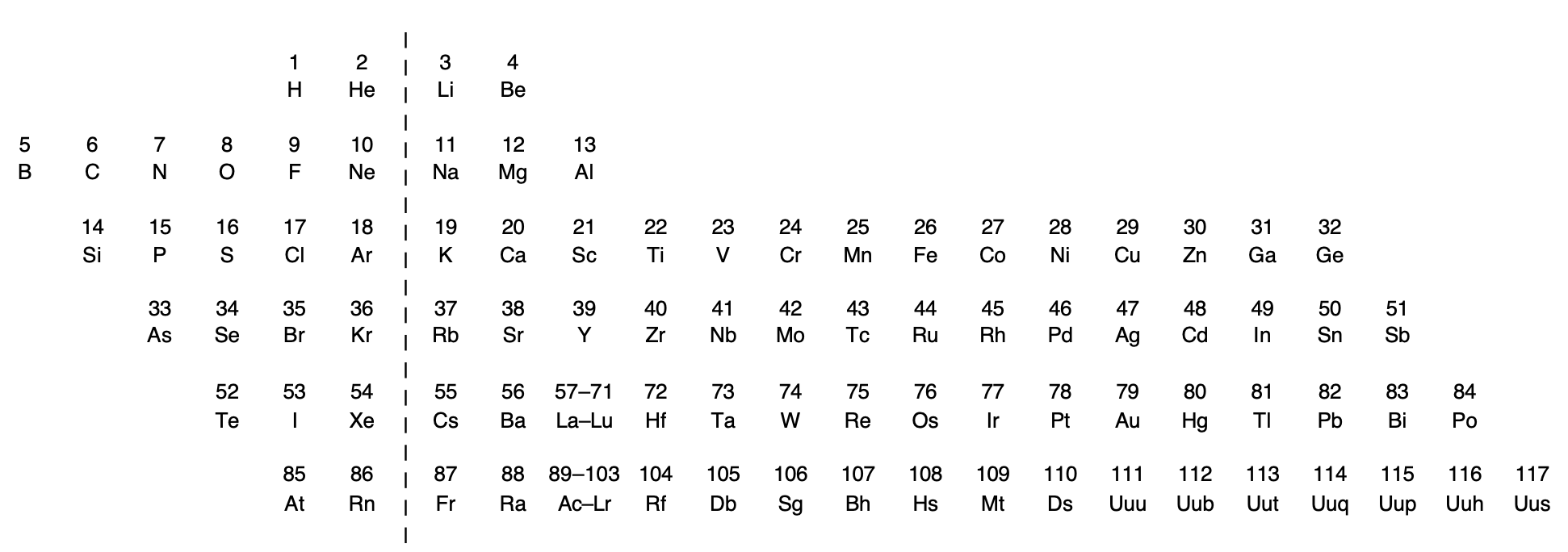
Thanks to René for the tip!
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
© Mark R. Leach Ph.D. 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com
This free, open access web book is an ongoing project and your input is appreciated.