Periodic Table |
 |
 |
 |
 |
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
The INTERNET Database of Periodic Tables
There are thousands of periodic tables in web space, but this is the only comprehensive database of periodic tables & periodic system formulations. If you know of an interesting periodic table that is missing, please contact the database curator: Mark R. Leach Ph.D. The database holds information on periodic tables, the discovery of the elements, the elucidation of atomic weights and the discovery of atomic structure (and much, much more).
Periodic Tables from the year 1919 :
| Year: 1919 | PT id = 434, Type = formulation |
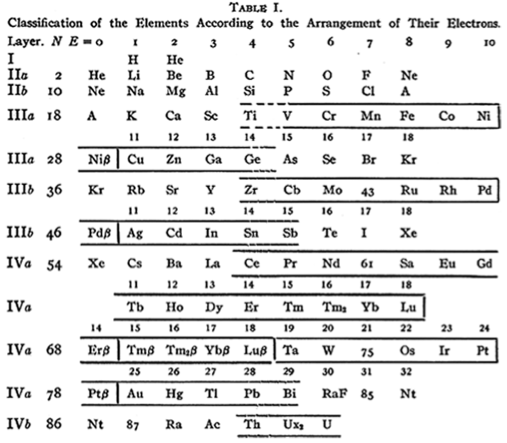
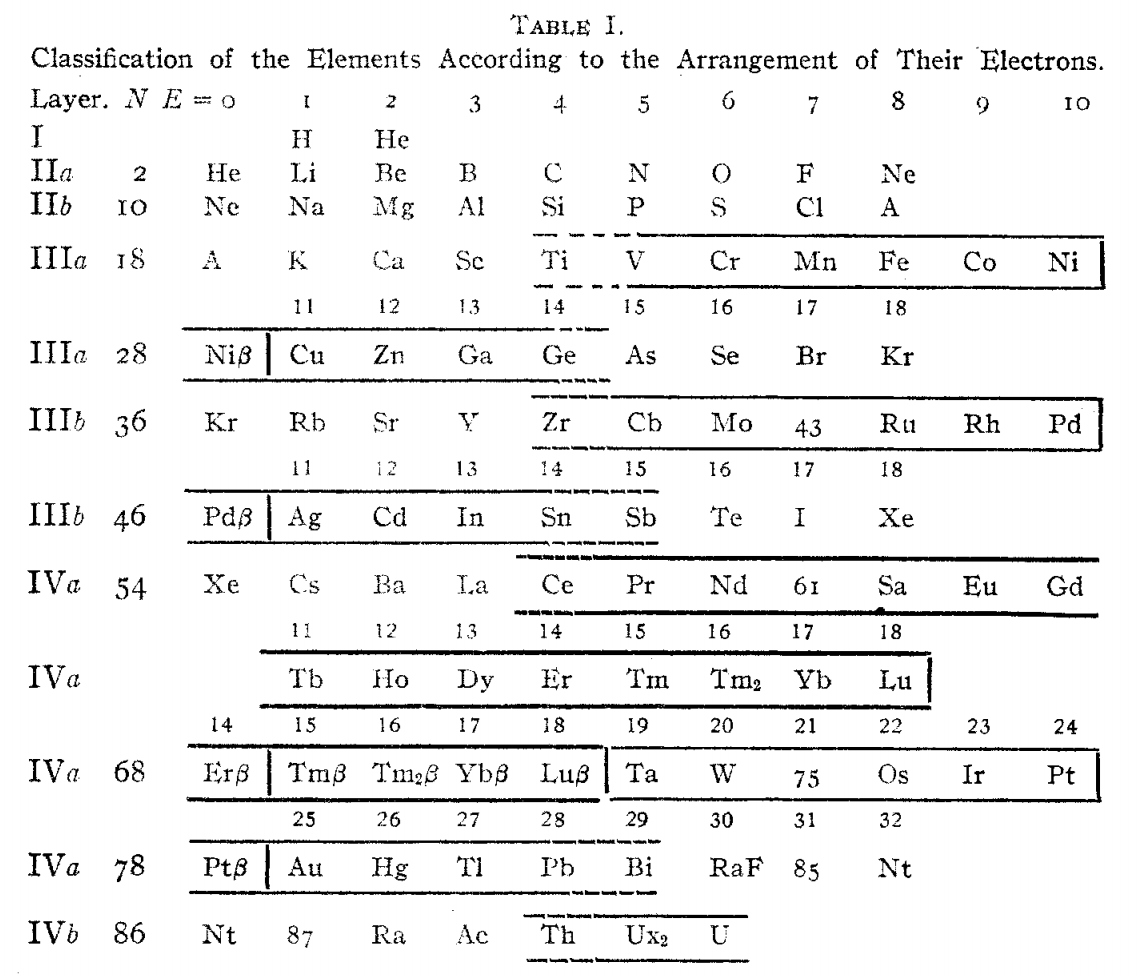
Langmuir's Periodic Table
From Irving Langmuir's theory of the Arrangement of Electrons in Atoms, J.Am.Chem.Soc., 41, 868 (1919), Langmuir's 1919 periodic table formulation.
This formulation seems to be the basis of Seaborg's formulations of 1939, 1942 & 1945:
| Year: 1919 | PT id = 547, Type = formulation |
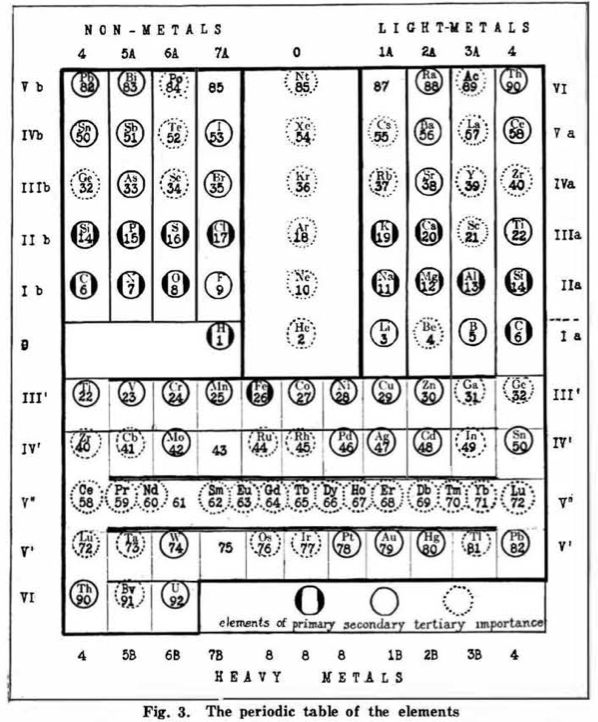
Hackh's Classification of the Elements, Updated
From a Scientific American in March 1919, an article by Ingo W. D. Hackh discussing the classification of the elements.
Shown is a periodic table slightly updated from a version from two years before, and referenced by Quam & Quam:
Thanks to Eric Scerri for the tip!
See the website EricScerri.com and Eric's Twitter Feed.
| Year: 1919 | PT id = 548, Type = formulation spiral |
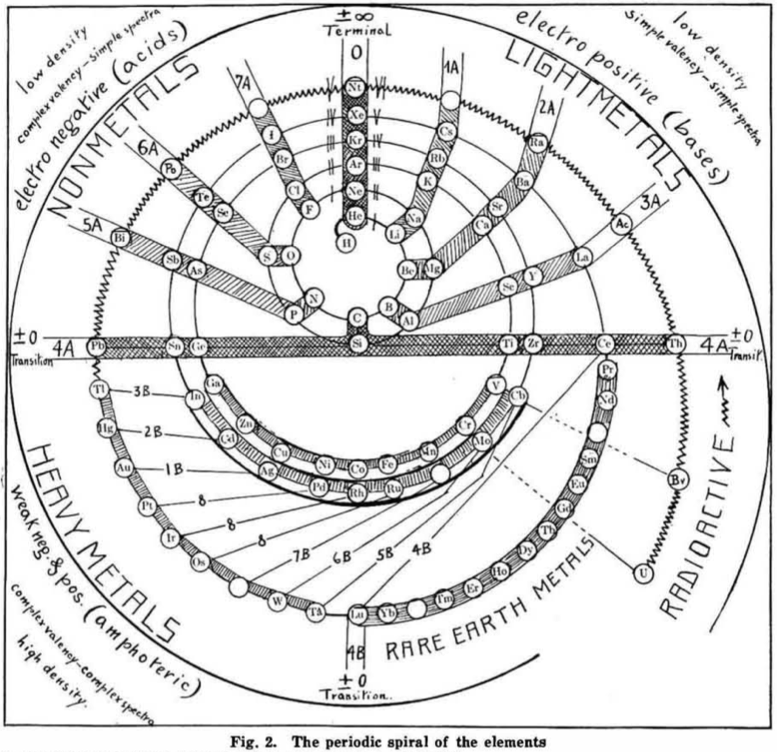
Hackh's Periodic Spiral
From a Scientific American in March 1919, an article by Ingo W. D. Hackh discussing the classification of the elements.
Included is a periodic spiral, developed from Hackh's 1914 version:
Thanks to Eric Scerri for the tip!
See the website EricScerri.com and Eric's Twitter Feed.
| Year: 1919 | PT id = 549, Type = formulation data |
Hackh's Periodic Chain
From a Scientific American in March 1919, an article by Ingo W. D. Hackh discussing the classification of the elements.
Included is a periodic chain showing the redox states of the elements:
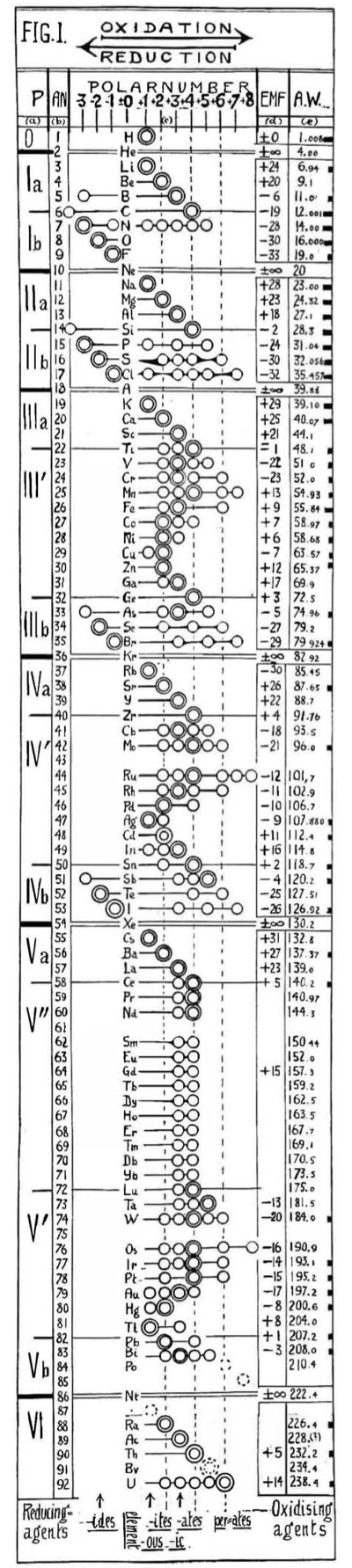
Thanks to Eric Scerri for the tip!
See the website EricScerri.com and Eric's Twitter Feed.
| Year: 1919 | PT id = 855, Type = element |
Discovery of Rhenium
Re
Rhenium, atomic number 75, has a mass of 186.207 au.
Rhenium was first observed or predicted in 1908 by M. Ogawa and first isolated in 1919 by M. Ogawa.
| Year: 1919 | PT id = 1293, Type = formulation |
Snyder's Fundamental Periodic Table of The Elements
Snyder MB 1919, The Fundamental Periodic Table of the Chemical Elements, filed in Congressional Library, Washington.
René Vernon writes:
"Notable for:
- Its attempted integration of the Ln and An into the short form of the periodic table
- Placement of H over He, Li and F
- Elements 108 = Pleon; 126 = Akron; 143 = Ultine"
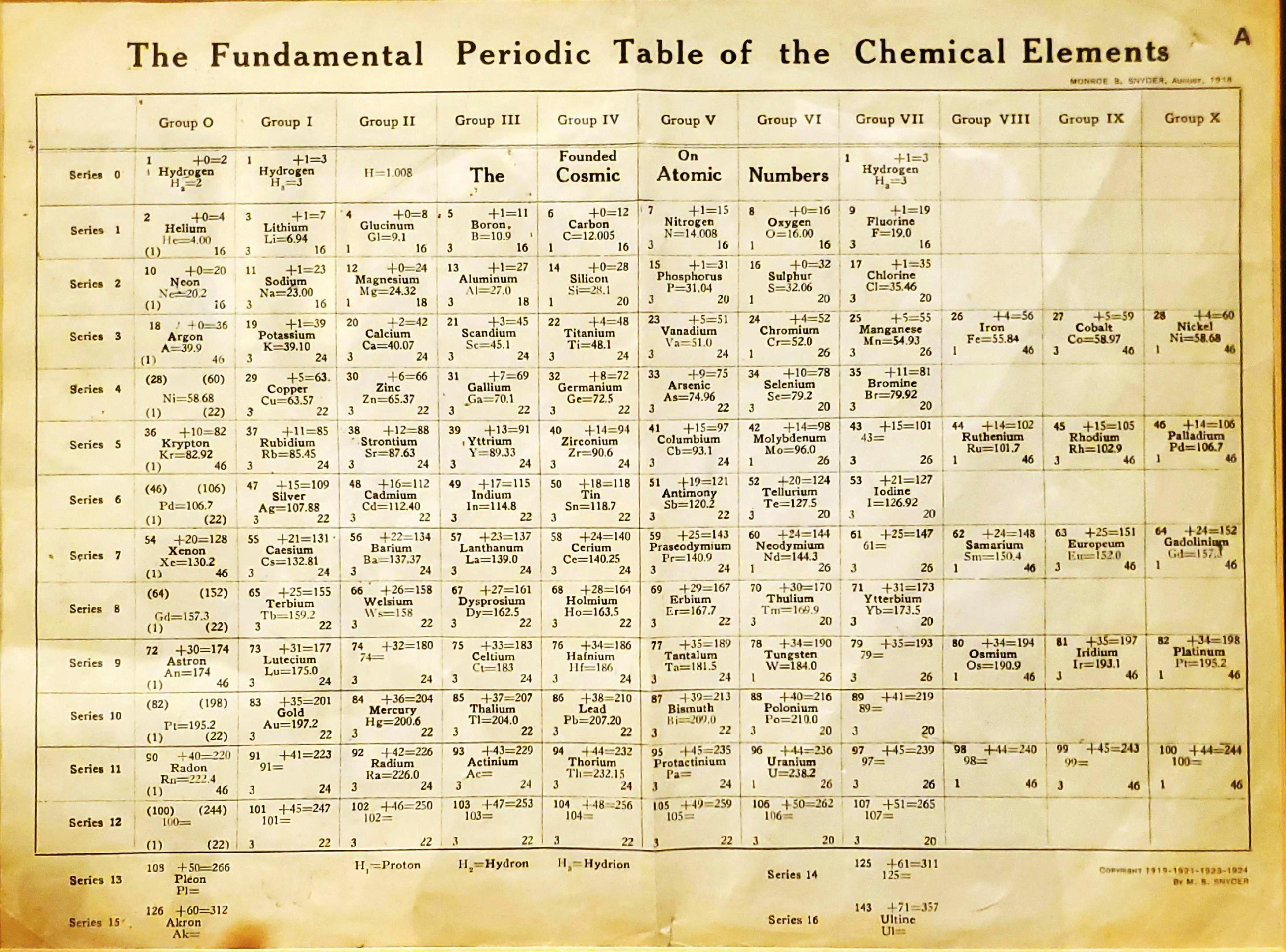
| Year: 1919 | PT id = 1386, Type = formulation |
Hackh's Classification of The Chemical Elements
Hackh, I. W. D. (1919). The classification of the chemical elements: The fundament of chemistry, Scientific American, 87 (supp. no. 2253), pp. 146–149 (148). https://zenodo.org/records/2454321
René Vernon writes:
Note that Group 4 (including Lu) appears twice, on the left and right.
Hackh does not get it quite right when he refers to a vertical similarity prevailing in the upper half of the table and a horizontal similarity in the lower half. A horizontal similarity prevails along the first row of the transition metals; vertical similarities tend to prevail among the second and third row dyads of the transition metals. That said, a horizontal similarity does prevail among the lanthanides.
On the noble gases, Hackh (p. 146) wrote: "...they combined the two extreme ends of a period, they formed the bridge from a non-metallic halogen (electro-negative element) to a metallic alkali (electro-positive element). For this reason we may speak of these elements, the rare or inert gases, as the terminals of the periods, which are either positive nor negative... The first three elements following an inert gas are always strong positive, while the last three before an inert gas are always strong negative and thus a kind of a transition is formed by the fourth element, or the elements of the carbon group."
For chemical properties he wrote: "The chemical characteristics of the elements can equally well be studied, for there are the acid- and base-forming elements on the chart, whose zones gradually infiltrate from strong basic to weak basic to atmospheric to weak acid to strong acid or vice versa."
Read more in the paper.
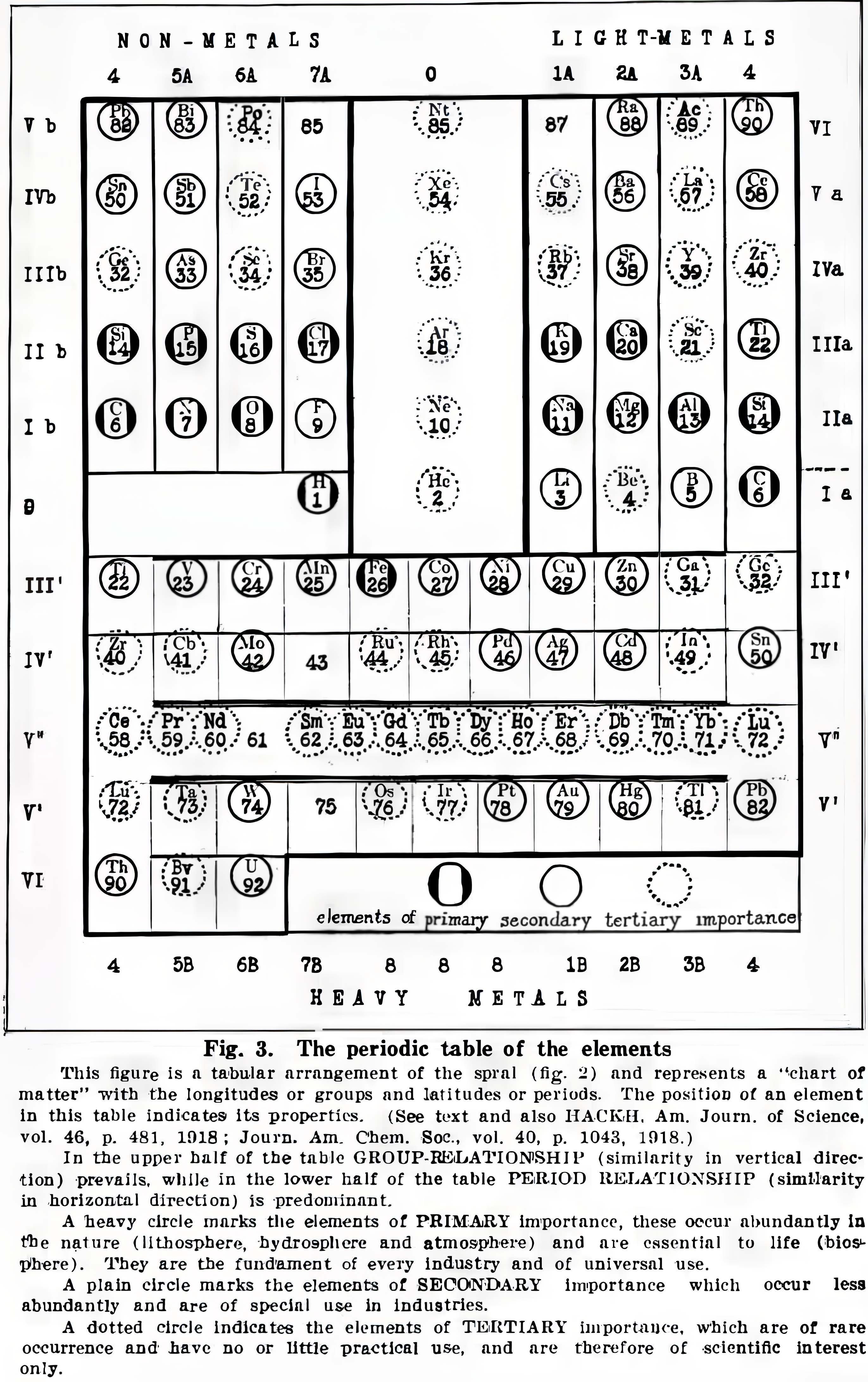
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
© Mark R. Leach Ph.D. 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com
This free, open access web book is an ongoing project and your input is appreciated.