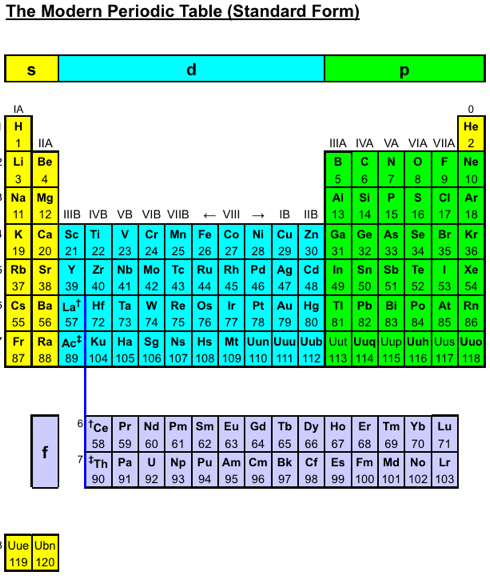
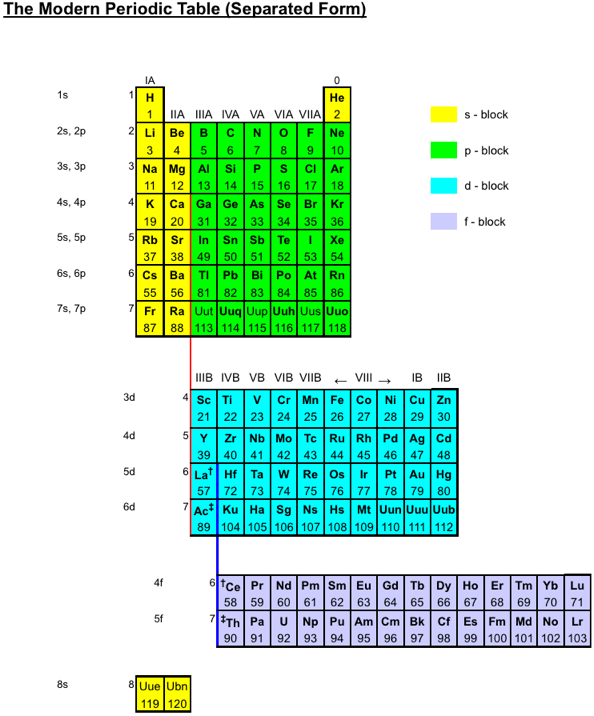
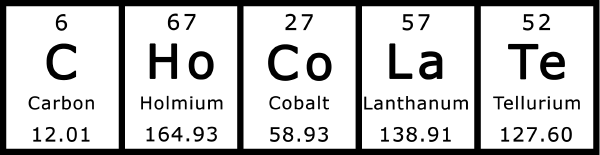Periodic Table |
 |
 |
 |
 |
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
The INTERNET Database of Periodic Tables
There are thousands of periodic tables in web space, but this is the only comprehensive database of periodic tables & periodic system formulations. If you know of an interesting periodic table that is missing, please contact the database curator: Mark R. Leach Ph.D. The database holds information on periodic tables, the discovery of the elements, the elucidation of atomic weights and the discovery of atomic structure (and much, much more).
Periodic Tables from the year 2012:
| Year: 2012 | PT id = 133, Type = data |
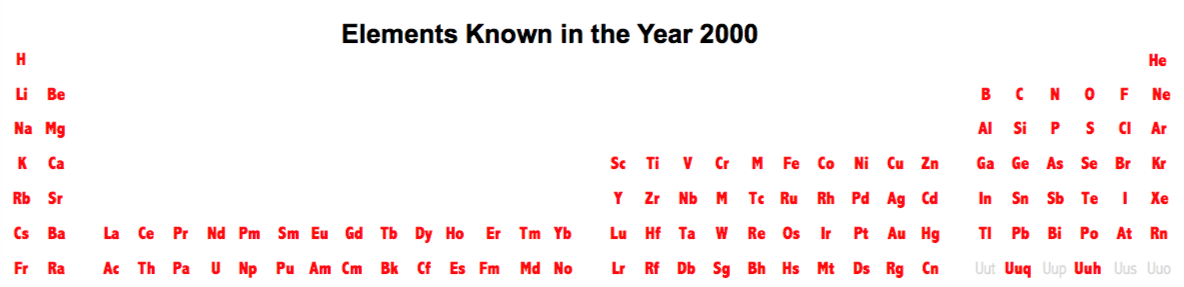
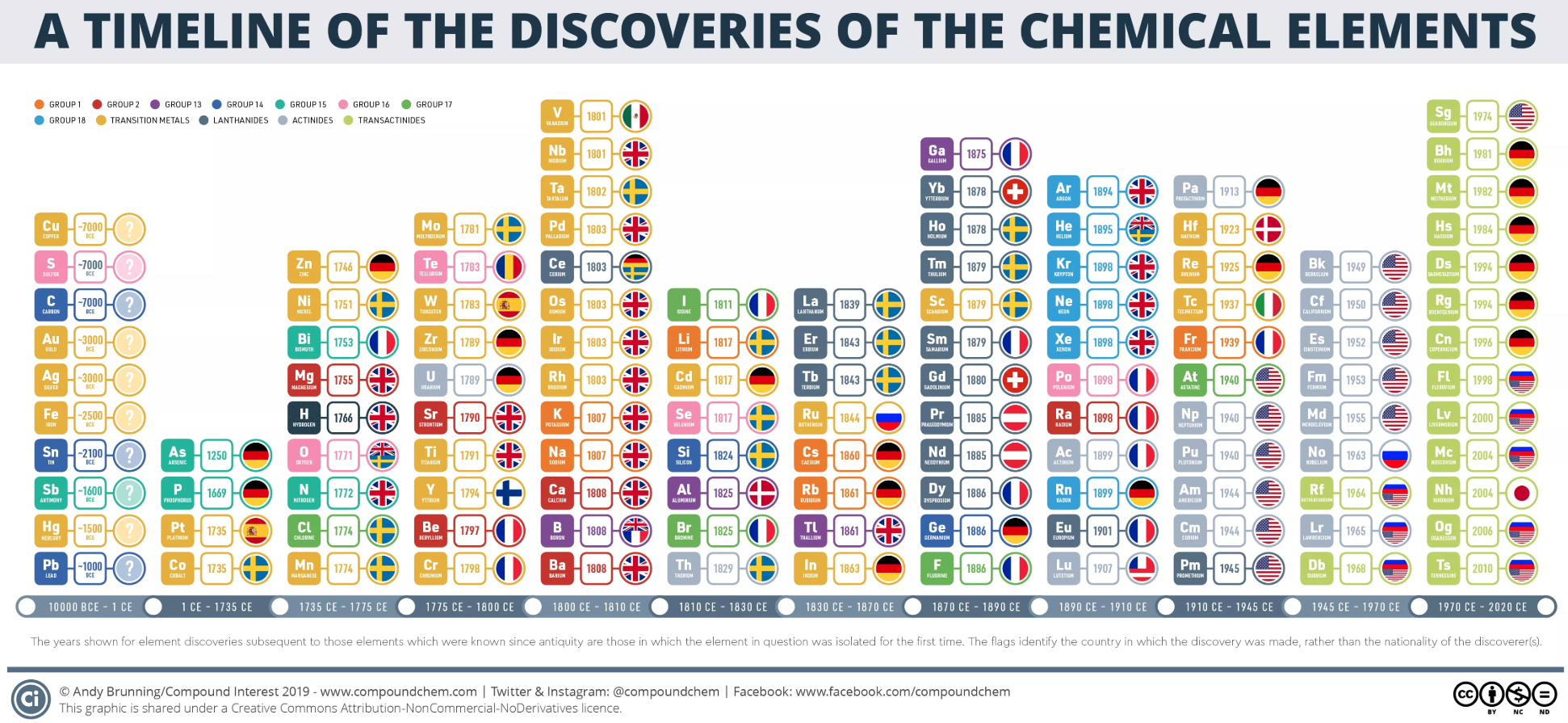
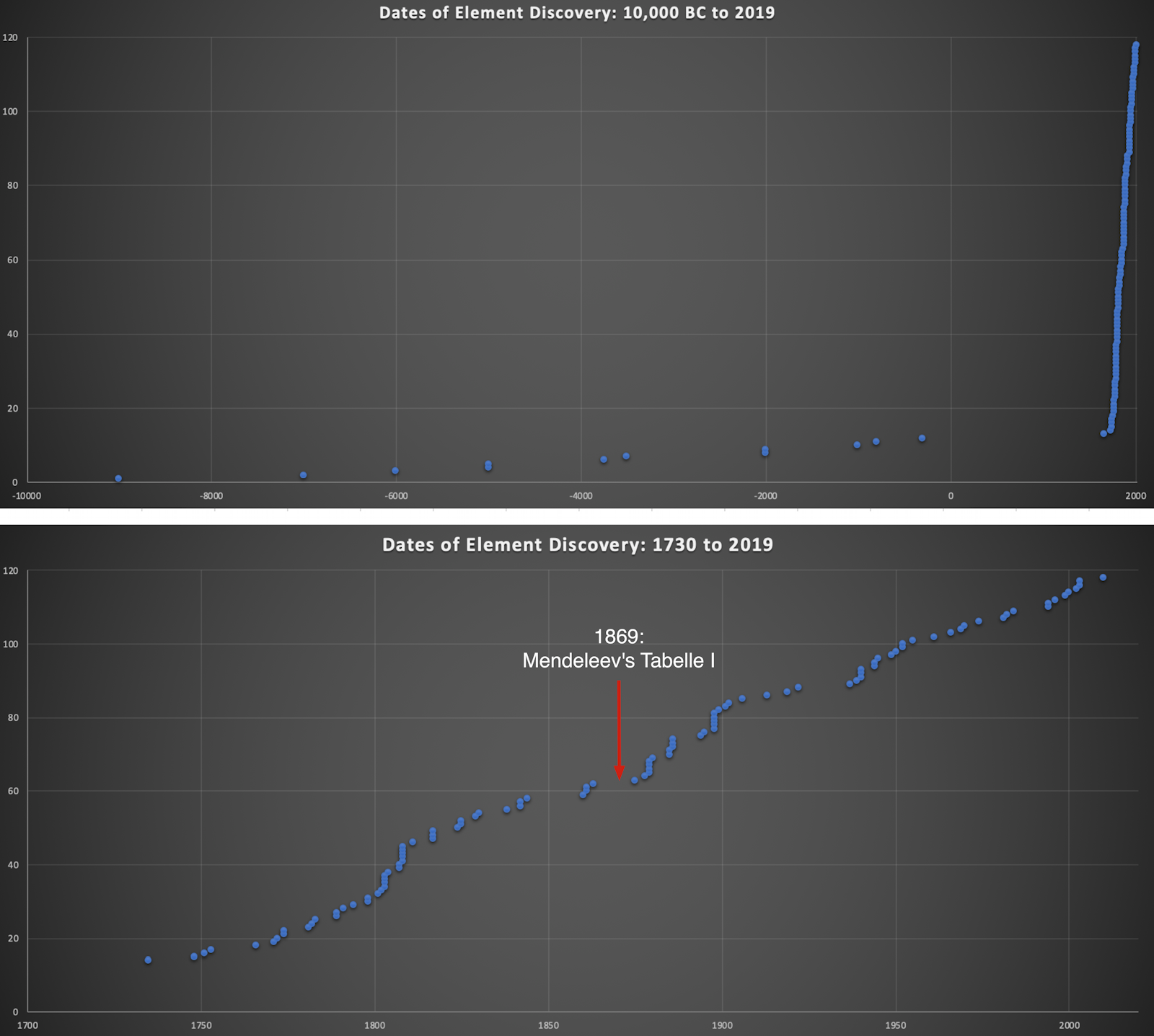
Dates of Discovery of the Elements
The Elements and their dates of discovery, taken from this Wikipedia page:
Two charts showing the dates of discovery of the elements, one from the 'time of the ancients' (10,000 BC) to the present day, and the second from 1700 to the present day.
These show that there were two distinct phases for the discovery of the 118 known elements:
- The first from about 10,000 BC to 1000 AD when 12 elements were discovered/used; one every 900 years or so.
- From 1669 until the present day when the other 106 have been rather steadily (and formally) discovered; one every couple of years.
- The last element to be made/discovered was in 2010.
Data from: this Wikipedia page.
| Discovery of Copper | -9000 |
| Discovery of Lead | -7000 |
| Discovery of Gold | -6000 |
| Discovery of Iron | -5000 |
| Discovery of Silver | -5000 |
| Discovery of Carbon | -3750 |
| Discovery of Tin | -3500 |
| Discovery of Sulfur (Sulphur) | -2000 |
| Discovery of Mercury | -2000 |
| Discovery of Zinc | -1000 |
| Discovery of Antimony | -800 |
| Discovery of Arsenic | -300 |
| Discovery of Phosphorus | 1669 |
| Discovery of Cobalt | 1735 |
| Discovery of Platinum | 1748 |
| Discovery of Nickel | 1751 |
| Discovery of Bismuth | 1753 |
| Discovery of Hydrogen | 1766 |
| Discovery of Oxygen | 1771 |
| Discovery of Nitrogen | 1772 |
| Discovery of Chlorine | 1774 |
| Discovery of Manganese | 1774 |
| Discovery of Molybdenum | 1781 |
| Discovery of Tellurium | 1782 |
| Discovery of Tungsten | 1783 |
| Discovery of Zirconium | 1789 |
| Discovery of Uranium | 1789 |
| Discovery of Titanium | 1791 |
| Discovery of Yttrium | 1794 |
| Discovery of Beryllium | 1798 |
| Discovery of Chromium | 1798 |
| Discovery of Niobium | 1801 |
| Discovery of Tantalum | 1802 |
| Discovery of Palladium | 1803 |
| Discovery of Cerium | 1803 |
| Discovery of Osmium | 1803 |
| Discovery of Iridium | 1803 |
| Discovery of Rhodium | 1804 |
| Discovery of Sodium | 1807 |
| Discovery of Potassium | 1807 |
| Discovery of Boron | 1808 |
| Discovery of Magnesium | 1808 |
| Discovery of Calcium | 1808 |
| Discovery of Strontium | 1808 |
| Discovery of Barium | 1808 |
| Discovery of Iodine | 1811 |
| Discovery of Lithium | 1817 |
| Discovery of Selenium | 1817 |
| Discovery of Cadmium | 1817 |
| Discovery of Silicon | 1824 |
| Discovery of Aluminium (Aluminum) | 1825 |
| Discovery of Bromine | 1825 |
| Discovery of Thorium | 1829 |
| Discovery of Vanadium | 1830 |
| Discovery of Lanthanum | 1838 |
| Discovery of Terbium | 1842 |
| Discovery of Erbium | 1842 |
| Discovery of Ruthenium | 1844 |
| Discovery of Cesium | 1860 |
| Discovery of Rubidium | 1861 |
| Discovery of Thallium | 1861 |
| Discovery of Indium | 1863 |
| Discovery of Gallium | 1875 |
| Discovery of Ytterbium | 1878 |
| Discovery of Scandium | 1879 |
| Discovery of Samarium | 1879 |
| Discovery of Holmium | 1879 |
| Discovery of Thulium | 1879 |
| Discovery of Gadolinium | 1880 |
| Discovery of Praseodymium | 1885 |
| Discovery of Neodymium | 1885 |
| Discovery of Fluorine | 1886 |
| Discovery of Germanium | 1886 |
| Discovery of Dysprosium | 1886 |
| Discovery of Argon | 1894 |
| Discovery of Helium | 1895 |
| Discovery of Neon | 1898 |
| Discovery of Krypton | 1898 |
| Discovery of Xenon | 1898 |
| Discovery of Polonium | 1898 |
| Discovery of Radium | 1898 |
| Discovery of Radon | 1899 |
| Discovery of Europium | 1901 |
| Discovery of Actinium | 1902 |
| Discovery of Lutetium | 1906 |
| Discovery of Protactinium | 1913 |
| Discovery of Rhenium | 1919 |
| Discovery of Hafnium | 1922 |
| Discovery of Technetium | 1937 |
| Discovery of Francium | 1939 |
| Discovery of Astatine | 1940 |
| Discovery of Neptunium | 1940 |
| Discovery of Plutonium | 1940 |
| Discovery of Americium | 1944 |
| Discovery of Curium | 1944 |
| Discovery of Promethium | 1945 |
| Discovery of Berkelium | 1949 |
| Discovery of Californium | 1950 |
| Discovery of Einsteinium | 1952 |
| Discovery of Fermium | 1952 |
| Discovery of Mendelevium | 1955 |
| Discovery of Lawrencium | 1961 |
| Discovery of Nobelium | 1966 |
| Discovery of Rutherfordium | 1969 |
| Discovery of Dubnium | 1970 |
| Discovery of Seaborgium | 1974 |
| Discovery of Bohrium | 1981 |
| Discovery of Meitnerium | 1982 |
| Discovery of Hassium | 1984 |
| Discovery of Darmstadtium | 1994 |
| Discovery of Roentgenium | 1994 |
| Discovery of Copernicium | 1996 |
| Discovery of Flerovium | 1999 |
| Discovery of Livermorium | 2000 |
| Discovery of Oganesson | 2002 |
| Discovery of Nihonium | 2003 |
| Discovery of Moscovium | 2003 |
| Discovery of Tennessine | 2010 |
By Mark Leach
A nice graphic from Compound Interest: (click image to enlarge)
| Year: 2012 | PT id = 458, Type = misc |
Coat of Arms Periodic Table
Amy Gramour has created a version of the Periodic Table that presents a coat of arms for each element. The attributes of the coats of arms symbolize the electron configuration and other selected features of each element.


| Year: 2012 | PT id = 479, Type = misc formulation |
Mathematical Expression of Mendeleev's Periodic Law
Valery Tsimmerman, of the ADOMAH Tetrahedron periodic table formulation and the Perfect Periodic Table website, presents a Mathematical Expression of Mendeleev's Periodic Law:

| Year: 2012 | PT id = 480, Type = data misc |
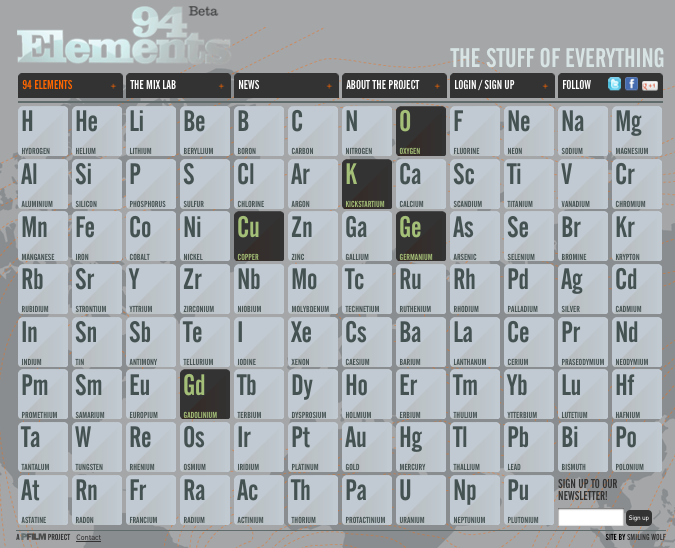
94 Elements: The Stuff of Everything
There are 94 naturally occuring elements, from hydrogen to plutonium. Together they make up everything in the world.
94 Elements is a global filmmaking project, exploring our lives through the lens of the elements. Everything that surrounds us is made from these 94 building blocks, each with its own properties and personality. Our own bodies are mostly made from just 6 of them.
The stories of the elements are the stories of our own lives. They reveal the patterns of our economies and the state of our relationships with our natural resources. The project is in part a celebration of the art of documentary film and some of the best filmmakers working today are making new films for the project. There'll also be opportunities for talented new and emerging filmmakers and animators to pitch their own films, with the winners chosen by you - the project community.
| Year: 2012 | PT id = 481, Type = misc |
Cupcakes, Periodic Table
From Kayla N. Green, Assistant Professor of Chemistry (Texas Christian University) comes a periodic table constructed from cupcakes baked for Chemistry Week 2012:
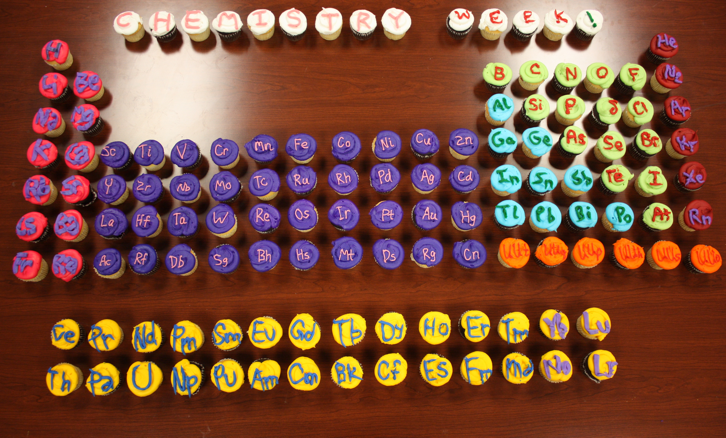
| Year: 2012 | PT id = 482, Type = misc |
3 Year-Old Sings Tom Lehrer's Elements Song
Rose turned 3 in November. It's been a little over a year since her initial elements video.
She's still interested in elements, but not so much by playing the cards anymore, mostly via this song:
| Year: 2012 | PT id = 483, Type = formulation |
Zigzag Periodic Table
In this periodic table we can see that the elements are arranged in a different way. Hydrogen is placed in between (and above) fluorine and lithium. This is because there is an issue on the placement of hydrogen as it has the properties of both alkali metals and halogens.
How to read the Zigzag periodic table
For periods (1), (2B), (3B) etc. read from right to left.
For periods (2A), (3A), (4A) etc. read from left to rightThe arrows will guide you through the periodic table:
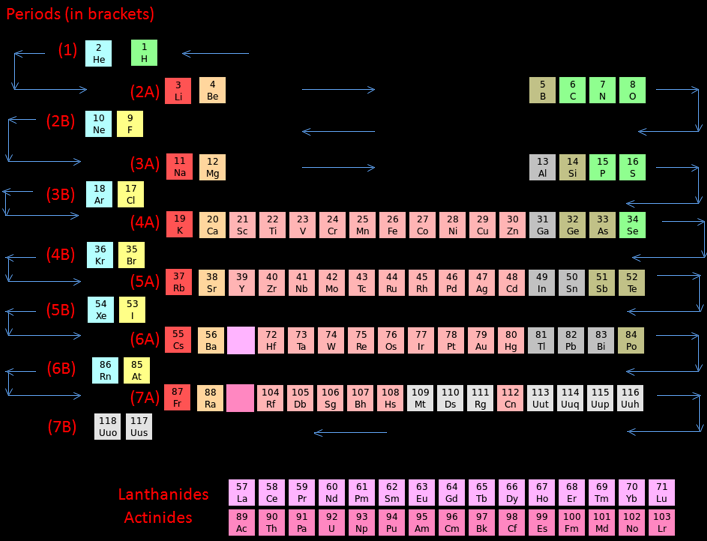
- Periodic Trends Atomic Radius- When moving opposite to the zigzag line in a particular period, the atomic radius of the elements increase.
- Metallic Character- Metallic Character decreases when moving along the zigzag line in a particular period.
- Ionization Energy- When moving along the zigzag line in a particular period, the ionization energy increases. Electron Affinity- Electron Affinity increases when moving along the zigzag line in a particular period.
By Akash Srivaths, High School Student, Chennai, India
| Year: 2012 | PT id = 484, Type = review misc |
Eric Scerri.com
ericscerri.com is the personal internet domain and website of Eric Scerri: chemist and leading philosopher of science specializing in the history and philosophy of the periodic table. He is founder and editor-in-chief of the international journal Foundations of Chemistry, which publishes academic papers concerned with the PT, and is the author of the respected book: The Periodic Table and Its Significance (Oxford University Press, 2007).
The website has links to all of Eric's extensive publications, including online video lectures and interviews and external links.
| Year: 2012 | PT id = 485, Type = review |
A Tale of 7 Elements
A new book by Eric Scerri: A Tale of 7 Elements about seven 'missing' elements: protactinium, hafnium, rhenium, technetium, francium, astatine, promethium:
| Year: 2012 | PT id = 486, Type = formulation |
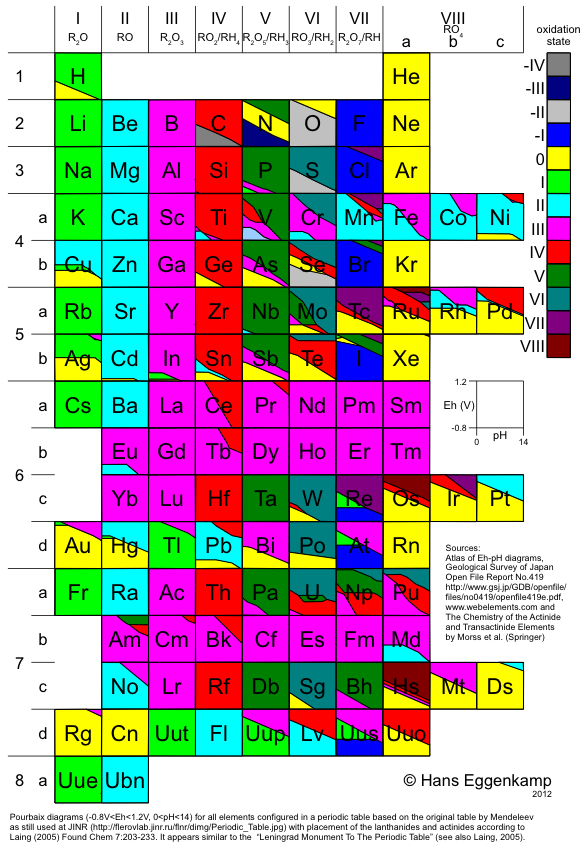
Eggenkamp's Periodic Table
Hans EggenkampI presents a periodic table based upon the table by Mendeleev, in combination with the lanthanides and actinides as suggested by Laing. A simplified Pourbaix (Eh-pH) diagram is shown for each element, colored according to the oxidation stage showing the systematics in the Periodic Table:
| Year: 2012 | PT id = 489, Type = non-chem |
Elemental Foundation of All Rap Songs
From the blog, The Internet Sucks Today, a mini-PT arrangement which shows "the elemental foundation of all rap songs":
| Year: 2012 | PT id = 490, Type = misc |
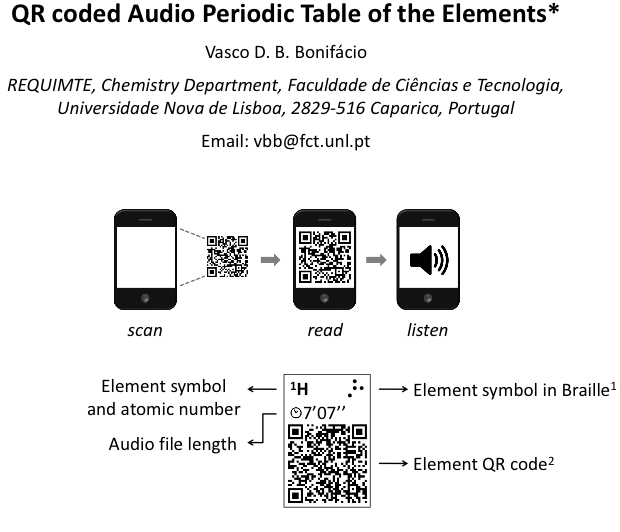
QR Coded Audio Periodic Table of the Elements
The QR coded Audio Periodic Table of the Elements by Vasco D. B. Bonifa?cio, REQUIMTE, Chemistry Department, Faculdade de Cie?ncias e Tecnologia, Universidade Nova de Lisboa, 2829-516 Caparica, Portugal. Email: vbb@fct.unl.pt.
From the paper in The Journal of Chemical Education "A quick response coded audio periodic table of the elements (QR-APTE) was developed using free online resources. The potential of QR-APTE was tested using a smart phone and is envisaged to become a truly powerful tool to teach chemistry to blind and visually impaired students under a mobile-learning environment":
| Year: 2012 | PT id = 492, Type = data |
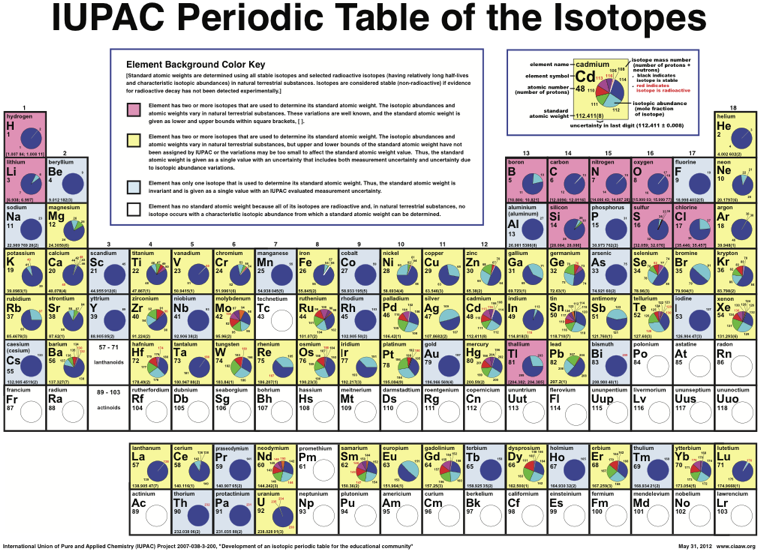
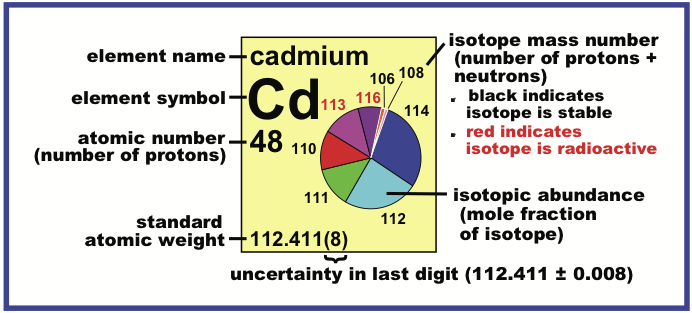
IUPAC Periodic Table of the Isotopes
The Periodic Table of the Isotopes, published by International Union of Pure and Applied Chemistry (IUPAC), is now available from the Commission on Isotopic Abundances and Atomic Weights, which is a commission under the Inorganic Division (Division II) of IUPAC.
The text identifies four types of atom, with respect to isotopes:
- Element has two or more isotopes that are used to determine its standard atomic weight. The isotopic abundances and atomic weights vary in natural terrestrial substances. These variations are well known, and the standard atomic weight is given as lower and upper bounds within square brackets, [ ].
- Element has two or more isotopes that are used to determine its standard atomic weight. The isotopic abundances and atomic weights vary in natural terrestrial substances, but upper and lower bounds of the standard atomic weight have not been assigned by IUPAC or the variations may be too small to affect the standard atomic weight value. Thus, the standard atomic weight is given as a single value with an uncertainty that includes both measurement uncertainty and uncertainty due to variations in isotopic abundances.
- Element has only one isotope that is used to determine its standard atomic weight. Thus, the standard atomic weight is
invariant and is given as a single value with an IUPAC evaluated measurement uncertainty.
- Element has no standard atomic weight because all of its isotopes are radioactive and, in natural terrestrial substances, no isotope occurs with a characteristic isotopic abundance from which a standard atomic weight can be determined.
| Year: 2012 | PT id = 493, Type = formulation data misc |
JR's Chemistry Set
For the iPhone and iPad, JR's Chemistry Set makes chemistry interesting and fun to learn. Based upon the innovative Rota Period, it is a handy and powerful reference tool for chemistry enthusiasts and practitioners at all ages and all levels.
| Year: 2012 | PT id = 495, Type = misc |
Mug Periodic Table
From www.msmugs.com, a coffee mug with the periodic table of the elements with the elements Lu and Lr correctly positioned... and a gift from Chris H.:


| Year: 2012 | PT id = 497, Type = non-chem |
Tour de France – 'le tour des Merveilles'
By onasixpence.com, 'le tour des Merveilles' is a visual tribute to a wonderful sporting event, using the iconic periodic table as the framework.
Information is included on each year's Tour de France winning rider. The race was first staged in 1903. The race was the idea of Géo Lefèvre, a journalist with L'Auto magazine. He first discussed the race with his editor at the Taverne Zimmer, a restaurant on the Boulevard de Monmartre. His editor, Henri Desgrange, famously responded, "If I understand you, petit Géo, you're proposing a Tour de France?":
| Year: 2012 | PT id = 498, Type = non-chem |
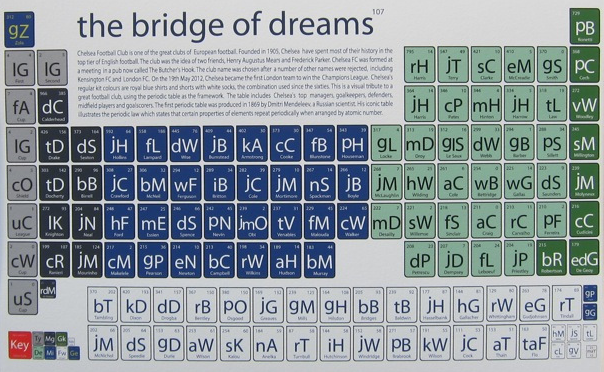
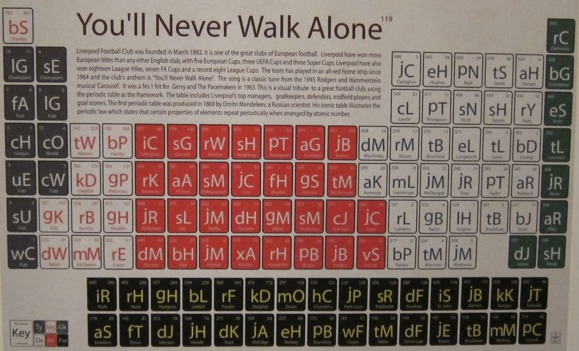
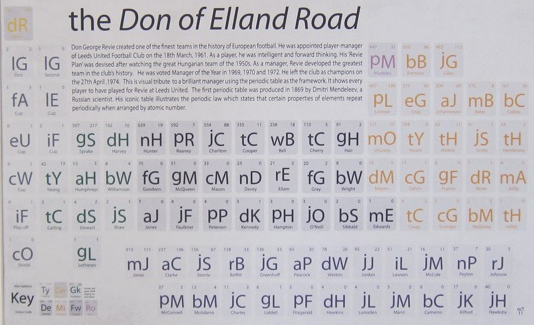
Football Club Periodic Tables by On A Six Pence
By onasixpence.com, offer a series of visuals of some British Premier League football clubs:
- Bridge of Dreams (Chelsea)
- Professor of Science (Arsenal)
- Science of Dreams (Manchester United)
- You'll Never Walk Alone (Liverpool FC)
- The Don of Elland Road (Leeds FC)
- Audere est Facere (Tottenham Hotspur FC)
- School of Science (Everton FC)
- Arsene Wenger (Manager):
| Year: 2012 | PT id = 499, Type = misc |
Mnemonic Periodic Table Song
By Ballroom Jam, a mnemonic song to help memorise the chemical elements:
Thanks to Eric Scerri for the tip!
See the website EricScerri.com and Eric's Twitter Feed.
| Year: 2012 | PT id = 500, Type = formulation misc |
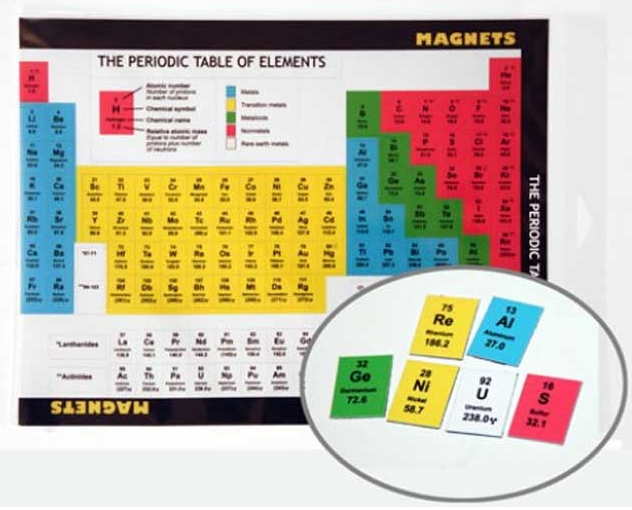
Magnetic Periodic Table
By Particle Zoo, sellers of Higgs Boson and Anticharm Quark soft toys, comes a magnetic periodic table which you can arrange into any formulation you like!
| Year: 2012 | PT id = 501, Type = review |
Books on the Chemical Elements and the Periodic Table/System
From Eric Scerri's forthcoming book A Tale of Seven Elements (Oxford University Press, 2013) and used by permission of the author, is the most complete and up-to-date list of Books on the Chemical Elements and the Periodic Table/System, including some titles in foreign languages.
Additional books in other languages can be found listed in Mazurs, 1974
- H. Alderesey-Williams, Periodic Tales, Viking Press, 2011
- N.P. Agafoshin, Ley Periódica y Sistema Periódico de los Elementos de Mendeleiev, Ed. Reverté S.A., Barcelona, 1977
- I. Asimov, The Building Blocks of the Universe, Lancer Books, New York, 1966
- P.W. Atkins, The Periodic Kingdom, Basic Books, New York, NY, 1995
- O. Baca Mendoza, Leyes Geneticas de los Elementos Quimicos. Nuevo Sistema Periodico, Universidad Nacional de Cuzco, Cuzco, Peru, 1953
- P. Ball, A Guided Tour of the Ingredients, Oxford University Press, Oxford, 2002
- P. Ball, A Very Short Introduction to the Elements, Oxford University Press, 2004
- I. Barber, Sorting The Elements: The Periodic Table at Work, Rourke Publishing, Vero Beach, Florida, US, 2008
- R. Baum (ed), Celebrating the Periodic Table, Chemical & Engineering News, A Special Collector's Issue, September 8, 2003
- H.A. Bent, New Ideas in Chemistry from Fresh Energy for the Periodic Law, Author House, Bloomington IN, 2006
- J. Bernstein, Plutonium, Joseph Henry, Washington DC, 2007
- J. C.A. Boeyens, D.C. Lavendis, Number Theory and the Periodicity of Matter, Springer, Berlin, 2008
- N. Bohr, Collected Works Vol 4. The Periodic System (1920-1923), Nielsen J Rud (Editor), North Holland Publishing Company, 1977
- T. Bondora, The Periodic Table of Elements Coloring Book, Bondora Educational Media Publications, 2010
- D.G. Cooper, The Periodic Table, 3rd edition. Butterworths, London, 1964
- P.A. Cox, The Elements, Oxford University Press, Oxford, 1989
- P. Depovere, La Classification périodique des éléments, De Boeck, Bruxelles, 2002
- H. Dingle and G.R. Martin, Chemistry and Beyond: Collected Essays of F.A. Paneth, Interscience, New York, NY, 1964
- S. Dockx, Theorie Fondamentale du Systeme Periodique des Elements, Office Internationale de Librairie, Bruxelles, 1950
- A. Ducrocq, Les éléments au pouvoir, Julliard, Paris, 1976
- A. Ede, The Chemical Elements, Greenwood Press, Westport, CT, 2006
- J. Emsley, The Elements, 3rd edition. Clarendon, Oxford University Press, 1998
- J. Emsley, Nature's Building Blocks, An A-Z Guide to the Elements, Oxford University Press, Oxford, 2001
- P. Enghag, Encyclopedia of the Elements, Wiley-VCH, Weinheim, 2004
- D.E. Fisher, Much Ado About (Practically) Nothing, The History of the Noble Gases, Oxford University Press, New York, 2010
- I. Freund, The Study of Chemical Composition: An Account of its Method and Historical Development, Dover Publications, Inc., New York, NY, 1968
- J. García-Sancho & F. Ortega-Chicote, Periodicidad Química, Trillas, México, 1984
- A. E. Garrett, The Periodic Law, D. Appleton & Co., New York, 1909
- L. Garzon Ruiperez, De Mendeleiev a Los Superelementos, Universidad de Oviedo, Oviedo, 1988
- L. Gonik, C. Criddle, The Cartoon Guide to Chemistry, Harper Resource, New York, 2005
- M. Gordin, A Well-Ordered Thing, Dimitrii Mendeleev and the Shadow of the Periodic Table, Basic Books, New York, 2004
- T. Gray, The Elements: A Visual Exploration of Every Known Atom in the Universe, Black Dog & Leventhal, 2009
- D. Green, The Elements, The Building Blocks of the Universe, Scholastic Inc. New York, 2012
- R. Hefferlin, Periodic Systems and their Relation to the Systematic Analysis of Molecular Data, Edwin Mellen Press, Lewiston, NY, 1989
- D.L. Heiserman, Exploring the Chemical Elements and their Compounds, McGraw-Hill New York, 1991
- S. Hofmann, Beyond Uranium, Taylor & Francis, London, 2002
- F. Hund, Linienspektren und Periodisches System der Elemente, Verlag von Julius Springer, Berlin, 1927
- W.B. Jensen, Mendeleev on the Periodic Law: Selected Writings, 1869-1905, Dover, Mineola, NY, 2005
- S. Kean, The Disappearing Spoon, Little, Brown & Co., New York, 2010
- D.M. Knight, Classical Scientific Papers, Chemistry Second Series, American, Elsevier, New York, NY
- P.K. Kuroda, The Origin of the Chemical Elements, and the Oklo Phenomenon, Springer-Verlag, Berlin, 1982
- H.M. Leicester and H.S. Klickstein, A Source Book in Chemistry 1400-1900, 1st Edition, McGraw-Hill Book Company Inc., London, 1952
- M.F. L'Annunziata, Radioactivity, Introduction and History, Elsevier, 2007
- S.E.V. Lemus, Clasificación periódica de Mendelejew, Guatemalan Ministry of Public Education, Guatemala, 1959
- P. Levi, The Periodic Table, 1st American Edition. Schocken Books, New York, NY, 1984
- R. Luft, Dictionnaire des Corps Simples de la Chimie, Association Cultures et Techniques, Nantes, 1997
- J. Marshall, Discovery of the Elements, Pearson Custom Publishing, 1998
- E. Mazurs, Graphic Representation of the Periodic System During One Hundred Years, Alabama University Press, Tuscaloosa, AL, 1974
- D. Mendeleeff, An Attempt Towards A Chemical Conception of the Ether, translated by G. Kamensky. Longmans, Green, and Co., London, 1904
- D. Mendeleeff, The Principles of Chemistry, translated by G. Kamensky, 5th Edition, vol. 2. Longmans, Green, and Co., London, 1891
- L. Meyer, Modern Theories of Chemistry, 5th Edition, translated by P.P. Bedson, Longmans, Green, and Co., London, 1888
- L. Meyer, Outlines of Theoretical Chemistry, 2nd Edition, translated by P.P. Bedson and W.C. William. Longmans, Green, and Co., London, 1899
- F. Mohr, (E), Gold Chemistry, Wiley-VCH, 2009
- D. Morris, The Last Sorcerers, The Path from Alchemy to the Periodic Table, Joseph Henry Press, New York, 2003
- I. Nechaev, G.W. Jenkins, The Chemical Elements, Tarquin Publications, Norfolk, UK, 1997
- R.D. Osorio Giraldo, M.V. Alzate Cano, La Tabla Periodica, Bogota, Colombia, 2010
- M.J. Pentz, (General Editor), The Periodic Table and Chemical Bonding, Open University Press, Bletchley, Buckinghamshire, UK, 1971
- I.V. Peryanov, D.N. Trifonov, Elementary Order: Mendeleev's Periodic System, translated from the Russian by Nicholas Weinstein, Mir Publishers, Moscow, 1984
- J.S.F. Pode, The Periodic Table, John Wiley, New York, NY, 1971
- R.J. Puddephatt, The Periodic Table of the Elements, Oxford University Press, Oxford, 1972
- R.J. Puddephatt and P.K. Monaghan, The Periodic Table of the Elements, 2nd edition. Oxford University Press, Oxford, 1986
- H.-J. Quadbeck-Seeger, World of the Elements, Wiley-VCH, Weinheim, 2007
- E. Rabinowitsch, E. Thilo, Periodisches System, Geschichte und Theorie, Stuttgart, 1930
- R. Rich, Periodic Correlations, Benjamin, New York, 1965
- J. Ridgen, Hydrogen, The Essential Element, Harvard University Press, Cambridge, MA, 2002
- H. Rossotti, Diverse Atoms, Oxford University Press, Oxford, 1998
- D.H. Rouvray, R.B. King, The Periodic Table Into the 21st Century, Research Studies Press, Baldock, UK, 2004
- D.H. Rouvray, R.B. King, The Mathematics of the Periodic Table, Nova Scientific Publishers, New York, 2006
- G. Rudorf, The Periodic Classification and the Problem of Chemical Evolution, Whittaker & Co., London, New York, 1900
- G. Rudorf, Das periodische System, seine Geschichte und Bedeutung für die chemische Sysytematik, Hamburg-Leipzig, 1904
- O. Sacks, Uncle Tungsten, Vintage Books, New York, 2001
- R.T. Sanderson, Periodic Table of the Chemical Elements, School Technical Publishers, Ann Arbor, MI, 1971
- S. E. Santos, La Historia del Sistema Periodico, Universidad Nacional de Educación a Distancia, Madrid, 2009
- E.R. Scerri, The Periodic Table, Its Story and Its Significance, Oxford University Press, New York, 2007
- E.R. Scerri, Selected Papers on the Periodic Table, Imperial College Press, London and Singapore, 2009
- E.R. Scerri, A Very Short Introduction to the Periodic Table, Oxford University Press, Oxford, 2011; Also translated into Spanish and Arabic.
- E.R. Scerri, Le Tableau Périodique, Son Histoire et sa Signification, EDP Sciences, 2011, (translated by R. Luft); Japanese Translation by Hisao Mabuchi et. al.
- C. Schmidt, Das periodische System der chemischen Elementen, Leipzig, 1917.
- G.T. Seaborg, W.D. Loveland, The Elements Beyond Uranium, Wiley, New York, 1990
- M.S. Sethi, M. Satake, Periodic Tables and Periodic Properties, Discovery Publishing House, Delhi, India, 1992
- H.H. Sisler, Electronic Structure, Properties, and the Periodic Law, Reinhold, New York, 1963
- P. Strathern, Mendeleyev's Dream, Hamish-Hamilton, London, 1999
- R.S. Timmreck, The Power of the Periodic Table, Royal Palm Publishing, 1991
- M. Tweed, Essential Elements, Walker and Company, New York, 2003
- F.P. Venable, The Development of the Periodic Law, Chemical Publishing Co., Easton, PA, 1896
- M.E. Weeks, Discovery of the Elements, Journal of Chemical Education, Easton PA, 1960
- B.D. Wilker, The Mystery of the Periodic Table, Bethlehem Books, New York, 2003
- J. Van Spronsen, The Periodic System of the Chemical Elements, A History of the First Hundred Years, Elsevier, Amsterdam, 1969
- T. Zoellner, Uranium, Penguin Books, London, 2009
- A. Zwertska, The Elements, Oxford University Press, Oxford, 1998
Works by D. I. Mendeleev
- Nauchnyi arkhiv. Periodicheskii zakon, t. I, ed. B. M. Kedrov. Moscow: Izd. AN SSSR, 1953
- Periodicheskii zakon. Dopolnitel'nye materialy. Klassiki nauki, ed. B. M. Kedrov. Moscow: Izd. AN SSSR, 1960
- Periodicheskii zakon. Klassiki nauki, ed. B. M. Kedrov. Moscow: Izd. AN SSSR, 1958
Thanks to Eric Scerri for the tip!
See the website EricScerri.com and Eric's Twitter Feed.
| Year: 2012 | PT id = 502, Type = formulation data |
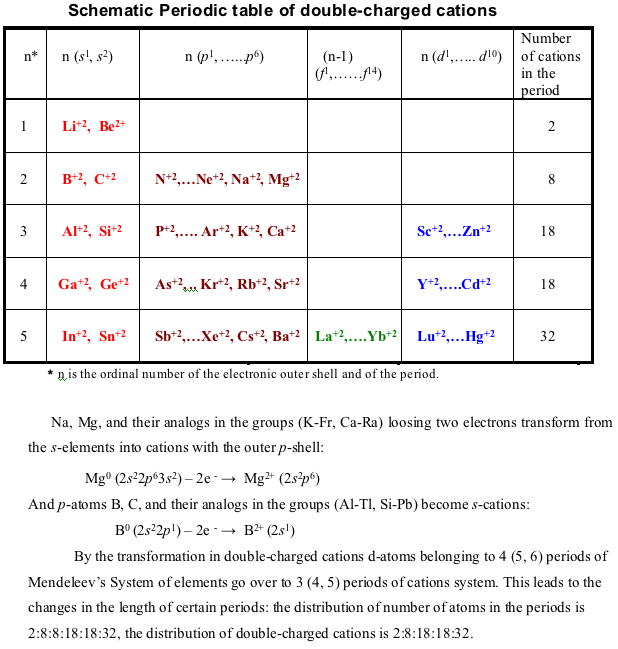
Schematic Periodic Table of Double-Charged Cations
N. S. Imyanitov / The Periodic Law. Formulations, Equations, Graphic Representations, Russian Journal of Inorganic Chemistry, Vol. 56 (14), 2183 - 2200, 2011 (In English), DOI: 10.1134/S0036023611140038
| Year: 2012 | PT id = 503, Type = formulation |
Extended Periodic Table - Alternative
From Rasko Jovanovic's World of Mathematics, an Extended Periodic Table - Alternative:

| Year: 2012 | PT id = 504, Type = formulation |
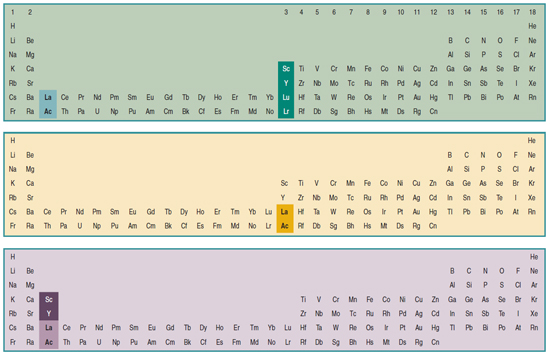
Three Different Long-Form, or 32-Column, Periodic Tables
From an article by Eric Scerri in the IUPAC magazine, Chemistry International, in which three different long-form, or 32-column, periodic tables with differences highlighted.
- Top: Version with group 3 consisting of Sc, Y, Lu, and Lr.
- Middle: Version with group 3 consisting of Sc, Y, La, Ac. The sequence of increasing atomic number is anomalous with this assignment of elements to group 3, e.g., Lu (71), La (57), Hf (72).
- Bottom: Third option for incorporating the f-block elements into a long-form table. This version adheres to increasing order of atomic number from left to right in all periods, while grouping together Sc, Y, La and Ac but at the expense of breaking-up the d-block into two highly uneven portions :
| Year: 2012 | PT id = 506, Type = formulation |
Rihani's 120 Element Periodic Table Formulations
Jeries Rihani writes: "Assuming the periodic table may reach an end at atomic number 120, I wish to draw your attention to the following three variations for the periodic table that I have on my web site Symmetry Of The Periodic Table which I think might be of interest, here, here & here":
| Year: 2012 | PT id = 509, Type = non-chem |
Airline Customer Review Periodic Table
A Periodic Table ranking Airlines by eDreams Customer reviews. Find your favorite way to fly:
Thanks to Eric Scerri for the tip!
See the website EricScerri.com and Eric's Twitter Feed.
| Year: 2012 | PT id = 510, Type = non-chem |
Standard Table of Influence
From Playmaker Systems comes the first Periodic Table of Influence: "For communicators, social media experts, marketers, sales people, politicos, developers and information warfighters. The first complete taxonomy of the 24 most basic stratagems in business, government and society.
"At the front line of The Playmaker Standard Decision System is the first taxonomy of the most basic stratagems observed in communication, social media, marketing, sales, politics and the military. Inspired by the periodic table of chemical elements, The Standard Table of Influence identifies, organizes the irreducibly unique strategies of influence - what we call plays.
"How is it organized? Each of the 24 named plays is assigned to one of three overarching classes (shown along The Table's top row as Assess, Condition and Engage) and then to one of the seven underlying subclasses (shown in The Table's second row as Test, Divert, Frame, Freeze, Press, Preempt and Provoke":
| Year: 2012 | PT id = 512, Type = non-chem |
Four of Diamonds: A Pirate Story
From the Creative Output blog, "Jim was a respectable middle-aged man who suddenly became a pirate. He didn't just start downloading art in ways contrary to the artists' wishes. He actually became a pirate. One minute he was looking at cat pictures on the internet at work, the next he was standing on an enemy ship, with a cutlass in one hand and a hook on the other...":
Thanks to Eric Scerri for the tip!
See the website EricScerri.com and Eric's Twitter Feed.
| Year: 2012 | PT id = 516, Type = review |
Eric Scerri's Lecture on The Periodic Table
A lecture by Eric Scerri at the Oscar Peterson auditorium of Concordia University, in Montreal.
The topic is the history and iconic nature of the Periodic Table, in high quality video, about one hour:
| Year: 2012 | PT id = 517, Type = misc |
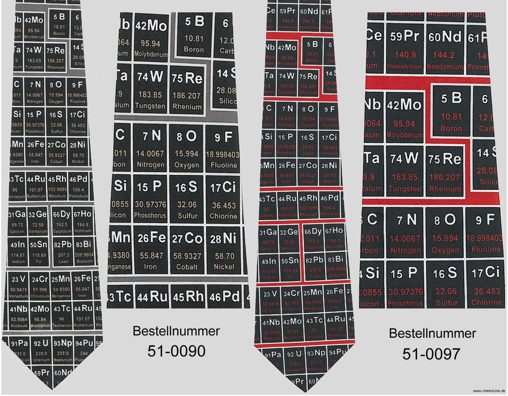
Chemoline Shop
Chemoline is an on-line shop, based in Germany with a multilingual site (click the flag).
The shop sells various science artifacts. Several products feature the periodic table, including:
| Year: 2012 | PT id = 518, Type = misc |
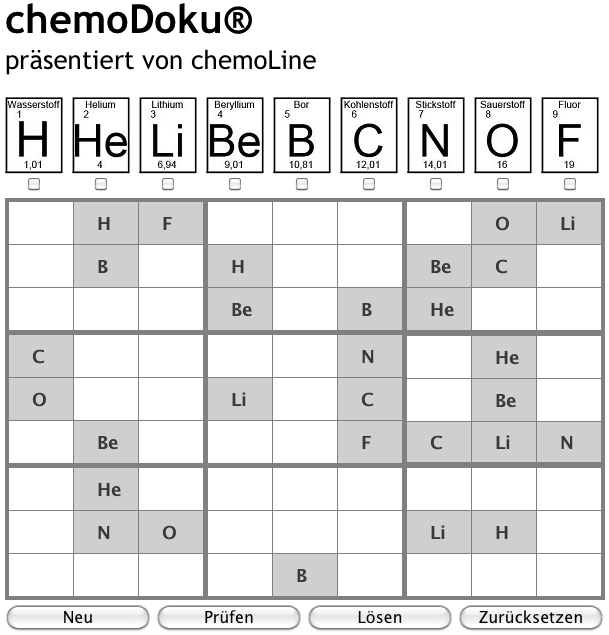
chemoDoku
Chemoline an on-line shop, based in Germany with a multilingual site (click the flag), sells various science artifacts that feature the periodic table.
The site also has an on-line chemical element version of Sudoku called chemoDoku, click to play:
| Year: 2012 | PT id = 519, Type = non-chem |
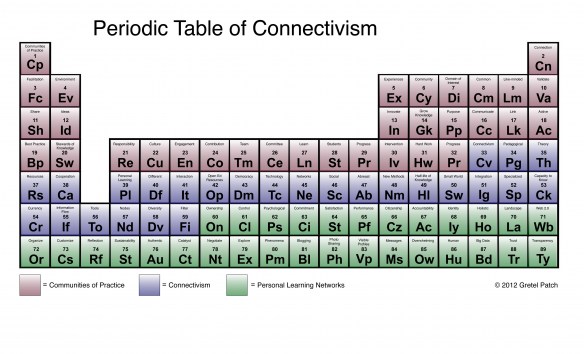
Connectivism, Periodic Table of
Gretal Patch writes in her blog, Edtech Learning Log:
"My EdTech543 assignment this week certainly stretched my creativity, but since I'm a wanna-be chemist at heart, I had to try it. The assignment was to non-linguistically represent the dense concepts of connectivism, personal learning networks, and communities of practice. Here is my attempt":
| Year: 2012 | PT id = 524, Type = formulation |
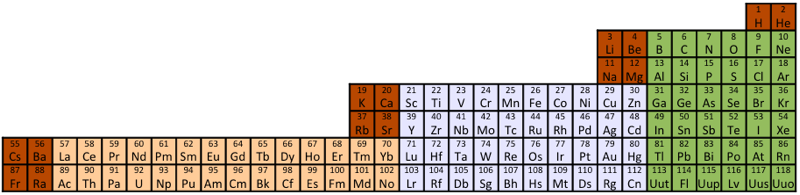
Compact Mendeleev-Moseley-Seaborg Periodic Table (CMMSPT)
A Compact Mendeleev-Moseley-Seaborg Periodic Table (CMMSPT).
This table can be found by two different ways:
- Via MMSPT - All terms of the MMSPT are shifted to the right side without spaces.
- Via Janet Periodic Table - The first row of the Janet PT is deleted. - We remove 2 from all others 118 terms.
These 2 transformations lead to the same table, with 7 rows and 32 columns. Blocks p (green), d (light grey), and f (light orange) are preserved.
The 14 terms of the s block (dark orange/red) are splited in "cascads".
This table can be seen in the A173592 sequence in the On-line Encyclopedia of Integer Sequences (OEIS). Row differences are 8, 8, 18, 18, 32, 32.
| Year: 2012 | PT id = 529, Type = formulation |
Srivaths–Labarca Periodic Table
This is an improved version of the Zigzag Periodic Table (2012). In this new arrangement the main criteria proposed to settle the placement of the elements hydrogen and helium has been taken into account: electronic configurations, the number of electrons needed to fill the outer-shell, chemical behavior, and triads of atomic number.
This is a new categorial criterion recently proposed by Eric Scerri, according to which hydrogen and helium form part of the triads H(1), F(9), Cl(17) and He(2), Ne(10), Ar(18), respectively. Thus, hydrogen preserves its place between alkali metals and halogen while helium is now in between noble gases and alkaline earth elements.
This periodic table allows visualizing easily the relationships of hydrogen and of helium with the different criteria, avoiding drawing lines to see them in contrast to other similar periodic systems.
Akash Srivaths, Chennai, India
Martín Labarca, CONICET & National University of Quilmes, Argentina

| Year: 2012 | PT id = 532, Type = formulation spiral 3D |
Alexander Arrangement of Elements, 3D Illustrated
The design of the 2012 Alexander Arrangement of Elements (AAE) follows the principles of a three-dimensional model developed by Roy Alexander in 1965: a printed representation of element information based on strict adherence to the Periodic Law, with every element data box physically and visually contiguous and continuous within the sequence of atomic numbers in generally accepted element property related columns - "...the periodic table the way it's supposed to be".
This is made possible by wrapping, folding, and joining the printed material and employing the patented p-block downslant of the element data boxes to allow the end element of a period to be adjacent to the first element of the next period.

Several unique features separate it from the previous four versions of the AAE
- The visual effect mirrors the look of Theodore Gray's series of posters, books, element cards and periodictable.com website and apps for the Apple iPod and iPad.
- Each element box is dominated by a Theodore Gray element photograph, with the element name, letter symbol, and atomic number relatively large, often overlapping the photo.
- The period numbers (below, right) are printed at the interface of the end/beginning of the periods, folded 90 degrees on the model, and the blocks and columns (old & new numbers), are identified below the data boxes - and in the case of the Actinoids, above.

- The element blocks connect at a central nexus (below, center), with the d- and f-blocks leaving, looping, and returning there, thus allowing the shorter period gaps above to be closed. For best visibility of the element data, these loops pinch together near the intersection. The p-block bends in a half-circle to join the s-block at the corner described above, with a patented 'downslant' where the element boxes gracefully sweep down a full box height (above) within this block to allow elimination of the "carriage return" effect: each period ending on the row above the next.

- The extended Hydrogen data box, a characteristic of all Alexander Arrangements, is more extended in this model, reaching for the multiple positions of the H box that are still under discussion among experts. The extra-extended Hydrogen box, illustrated by a composite image of a hydrogen cloud in space, (above, right) loops over the s- and p-blocks. Starting up from behind the corner of Helium & Lithium, inside the half-helical tube to loop over Helium, attach above Lithium, Beryllium and then Carbon as the loop descends (joining the ascending portion) over the data boxes of the s- & p-blocks, terminating in contact with Fluorine, Neon, and corner-on to Neon.
- The model size is the same as the previous Display Version of the AAE, but has fewer element data boxes, due to there being no photos of the lab created elements and for simplification of the educational application - introduction to property periodicity and organization of element data - the elements with atomic numbers over 94 are not included (see addendum).
- Where the f-block begins and ends, between Barium and Lutetium, the f-block is held perpendicular to the only flat segment of the element display by a pair of triangular braces, which also create the flat area, aligning the s-block with the 'pinch' of the d-block. This is particularly apparent from the bottom, when the model is supported from above. (see below)

Designed by Roy Alexander, a science museum exhibit and teaching aid designer, the Adobe Illustrator art for the model was started by Ann Grafelman, and continued by Roy from mid 2011 through November of 2012.
Photos were provided by Theodore Gray, and Element Collection funded the printing and die cutting performed by Strine Printing in York, Pennsylvania. The model kit was first offered at Theo's PeriodicTable.com, then at Roy's AllPeriodicTables.com and the new 3dPeriodicTable.com, which site is dedicated to the 3D Forever Periodic Table only, with add-ons, application suggestions, and descriptions and commentary of all sorts.
Assembly instructions and step photos, as well as a number or completed model color photographs are included with the kit. These were developed with prototype models, and while functional, have been upgraded and accompanied by an assembly video at AlexanderArrangementOfElements.com/3D
Addendum:
Text relating to the abbreviation of the ever increasing number of elements is explained at two places on the 3D AAE illustrated periodic table model kit. One will remain with the model and one is removed at the time of assembly.
That which remains runs under the Actinoids and the d-block elements, where the lab created elements might ordinarily be expected to be found, says:
The lab created elements ordinarily found in this part of a periodic table are not to be found in nature, there can be no photographs of them, so nothing needs to be added to this element photo periodic table - ever - so it will never be obsolete, a Forever Periodic Table.
That which is removed says:
Naturally-occurring elements have been numbered variously, generally between 80 and 96, all for cogent scientific reasons.
For easier teaching and learning, we have included on this periodic table only the 92 elements actually currently existing on Earth and in the remainder of the Universe, and adding Technetium and Promethium, which, although they may have no stable forms, serve to fill what would otherwise be gaps in the sequence.
Not added for practical and educational reasons are 'elements' consisting only of pages and pages of computer data from smashing atoms in particle accelerators. Another reason is that there can be no photographs of them to show, and as a result, your arrangement is complete and never be obsolete - your Forever Periodic Table.
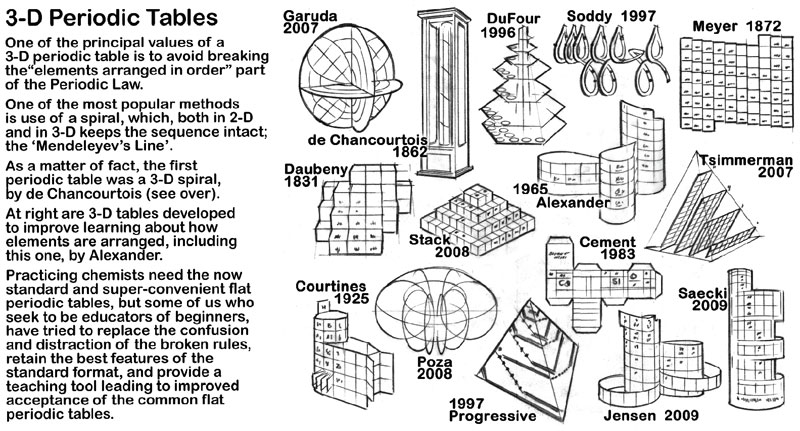
Included with the art of the periodic table on the die cut substrate that makes up the model is some background information about the the history of three dimensional periodic tables.
The first of these is about the discoverer of the concept of arranging the elements in periods suggested by the properties of the elements, de Chancourtois.

The second 3D periodic table information piece (on the rear of the de Chancourtois removable card) are sketches of a number of the 3D periodic tables found on the Chemogenesis website.
| Year: 2012 | PT id = 533, Type = formulation spiral |
Vortic Periodic Table in Marquetry
From Dr David Robson:
"My vortic periodic table created in marquetry may be of interest. I have always thought of vortic energies and with retirement time, I used my Marquetry Hobby to so create. Despite the inevitable Black Hole centre I have included the Higgs Boson there as a tribute to its discovery and potential as a window to elsewhere."
| Year: 2012 | PT id = 534, Type = non-chem |
Chocolate Periodic Table
Two way proof that the periodic table is made of chocolate, from Fo'Drizzle and Robeastro:
| Year: 2012 | PT id = 536, Type = formulation |
Makeyev's Verticle Form Periodic Table
A new version of the periodic table of elements on the vertical table form. Alexander K. Makeyev, a member of the Moscow Society of Naturalists, section of planetonautics; freelance interdisciplinary researcher and inventor, knowall@list.ru.
1. Makeyev A.K. Normal and pathological anatomy and physiology of the human person and society. Fundamental knowledge about the qualities of the human person, human society and the software company, produces and acts of people, based on the universal algorithm of holographic structure and function at all levels and forms of matter. / / Scientific and Technical Library. July 25, 2012. 364 p, here
2. Makeyev A.K. Particles of electrostatic and magnetic fields in the system of matter photons move faster than a photon moves himself. / / The scientific debate: Proceedings IV International Correspondence scientific conference. Part I. (20 August 2012) - Moscow:. "International Centre for Science and Education", 2012. 142., S. 47-65. ISBN 978-5-905945-37-3 UDC 08. BBK 94. H 34, here:
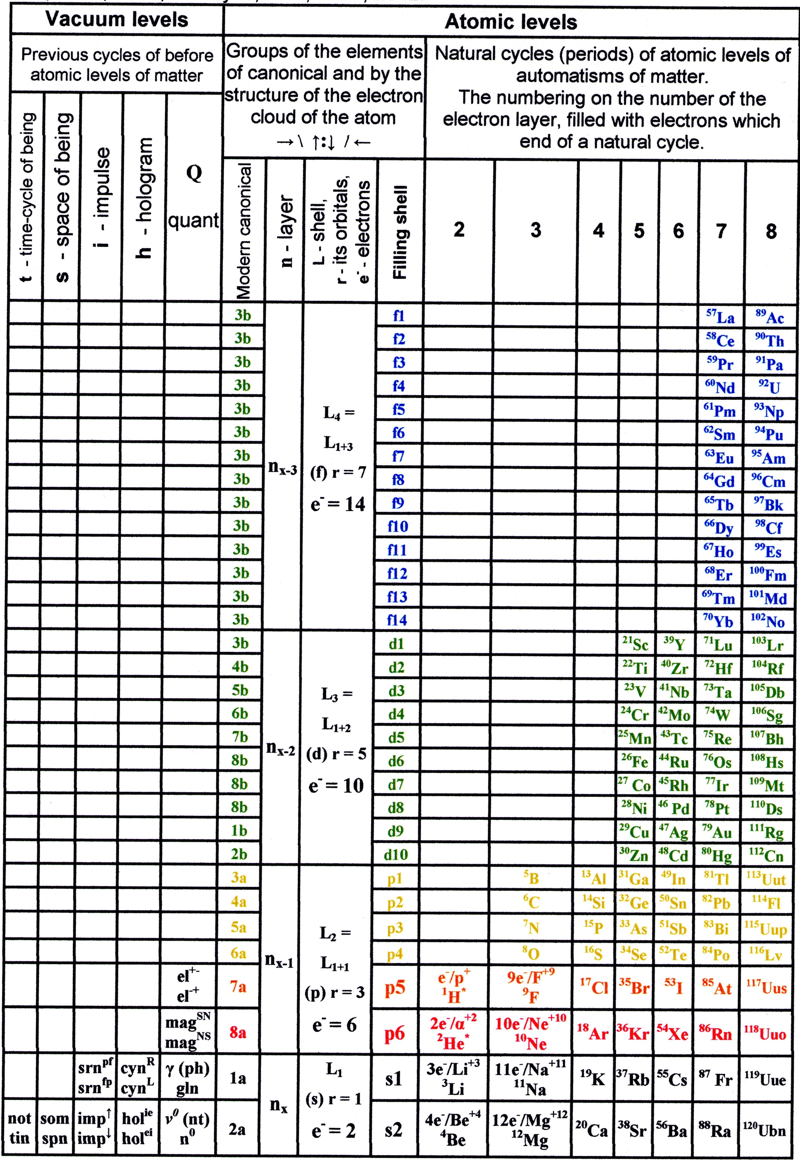
| Year: 2012 | PT id = 537, Type = non-chem |
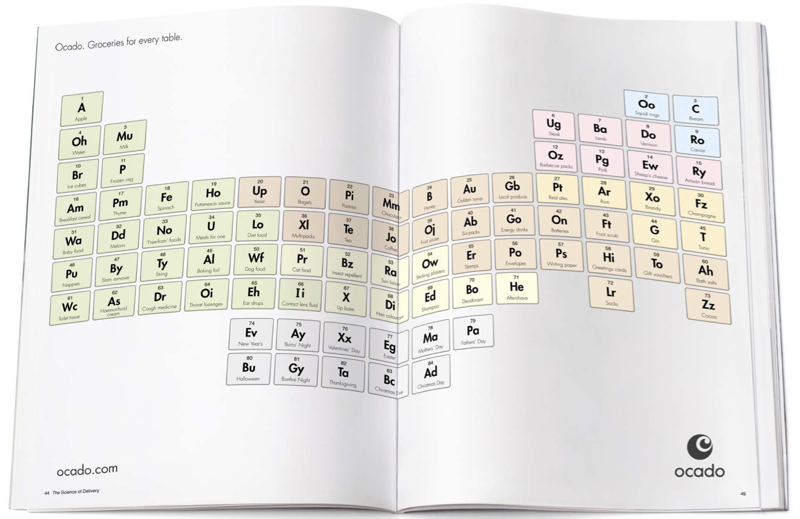
Ocado Groceries for Every Table
From a possible advertising campaign, the Ocado Groceries for Every Table:
| Year: 2012 | PT id = 538, Type = formulation spiral |
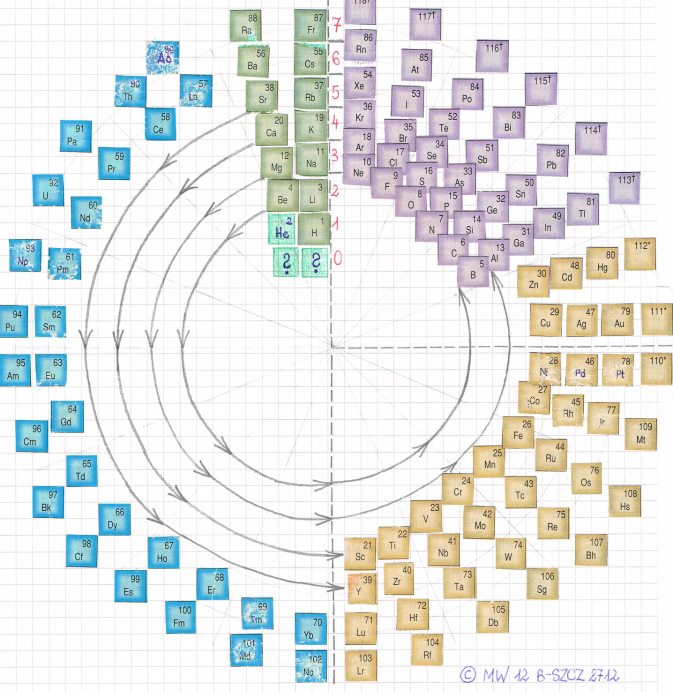
Wheelshaped Table of Elements
From Facebook, a Wheelshaped table of elements.
Please note the symmetry of this representation.
As a result, it is possible that element 118 is the very last one in the periodic table. We have the sequence:
2 x 14 (blue)
4 x 10 (brown)
6 x 6 (violet)
8 x 2 (green)
and, logically, neither first nor last factor can be 0 or -2 (they differ in two columns above respectively by 2 and 4).
On the other hand, the coherence of the structure requires the existence of two additional elements at the beginning!
| Year: 2012 | PT id = 542, Type = formulation |
Bettermann Periodic Table
In the course of my enquiries regarding the peridoc table of the elements your comprehensive and interesting collection of the varying configuration of the elements caught my eye. Responding to a growing interest, I worked through all models but couldn't find any configuration which agrees with mine.
Find my configuration for the elements in the figure below. Please open the attachment in which you find an explanatory statement for the illustrated principle, pdf file here. It bases upon the Moseleysches diagrams and the work of Eugenie Lisitzin from the thirties of the last century.
Heiner Bettermann
| Year: 2012 | PT id = 543, Type = misc review |
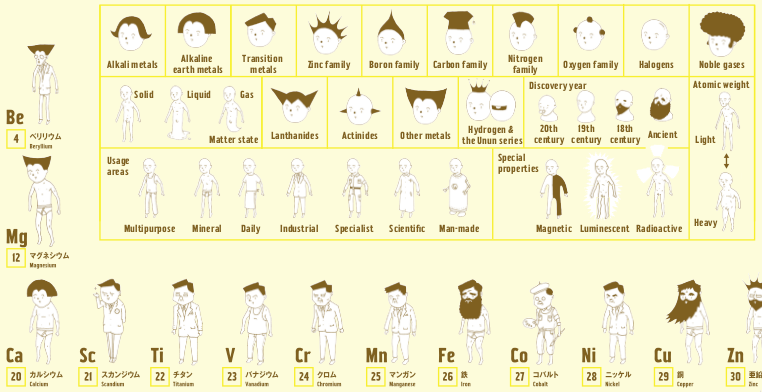
Wonderful Life with the Elements
From the Japanese artist Bunpei Yorifuji comes Wonderful Life with the Elements, an illustrated guide to the periodic table that gives chemistry a friendly face, available from Amazon.
In this super periodic table, every element is a unique character whose properties are represented visually: heavy elements are fat, man-made elements are robots, and noble gases sport impressive afros. Every detail is significant, from the length of an element's beard to the clothes on its back. You'll also learn about each element's discovery, its common uses, and other vital stats like whether it floats - or explodes - in water.
There is also a full review with more images from Wired.
| Year: 2012 | PT id = 544, Type = non-chem |
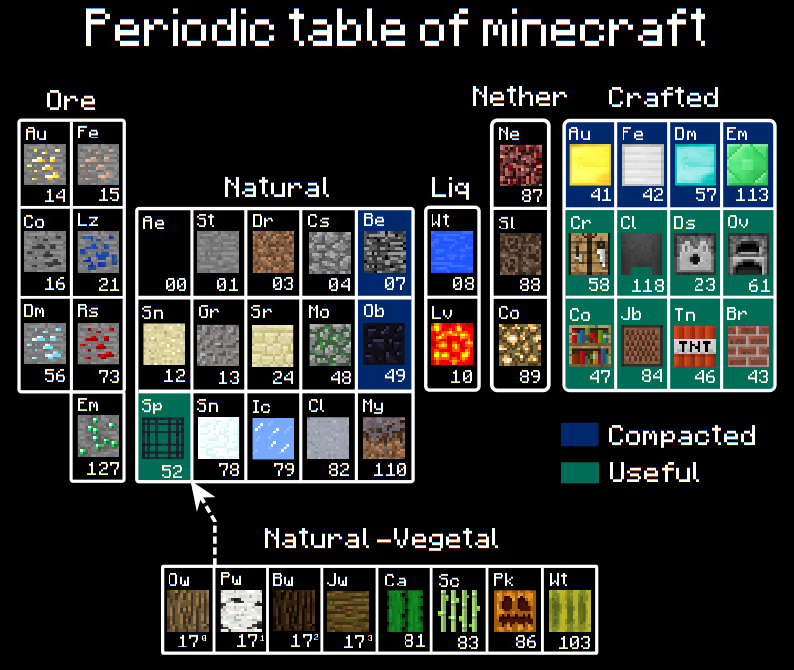
Minecraft, Periodic Table of
A Periodic Table of Minecraft materials by egeres:
| Year: 2012 | PT id = 551, Type = formulation |
Piazzalunga's Pyramidal Periodic Table Formulations
Three Pyramidal Periodic Table Formulations, and a Spiral, from Marco Piazzalunga:
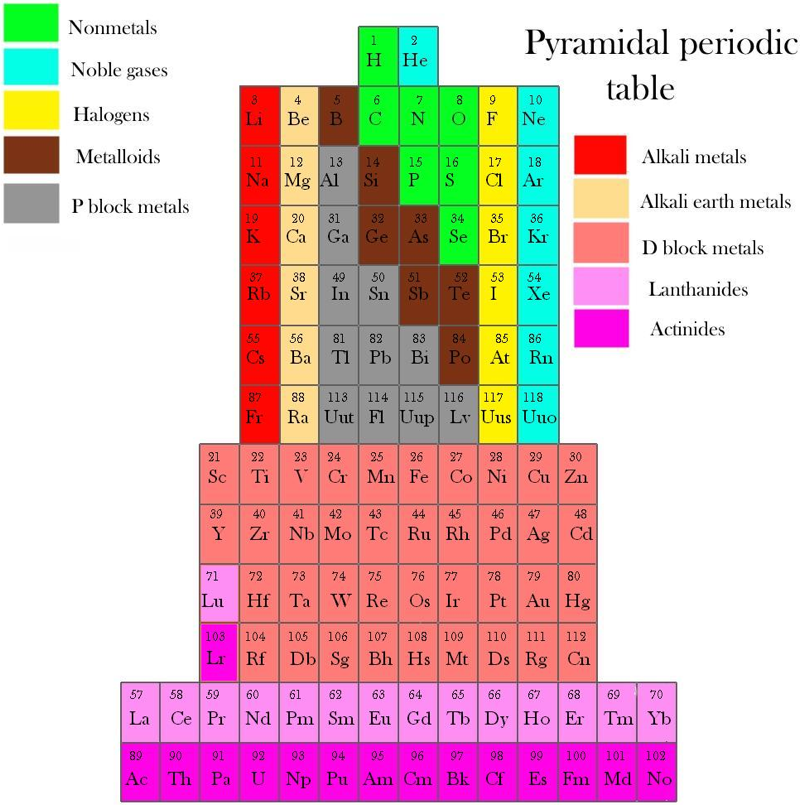
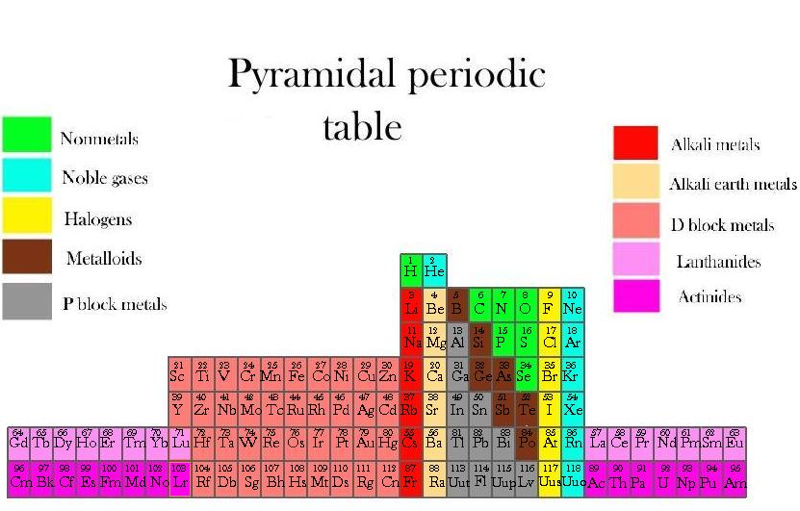
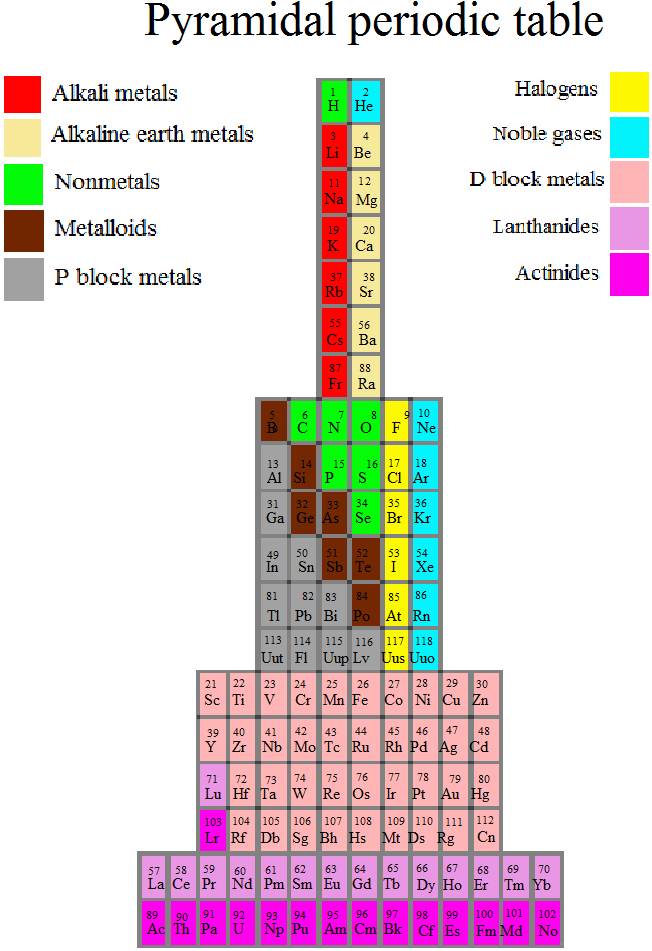
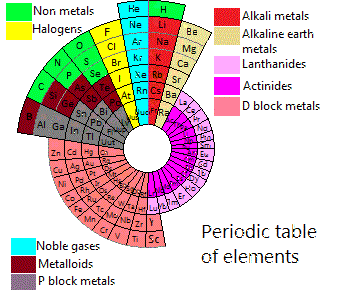
| Year: 2012 | PT id = 553, Type = misc |
Building Block Elements
From Think Geek, element building blocks... so you can build your own PT Formulation:
| Year: 2012 | PT id = 554, Type = non-chem |
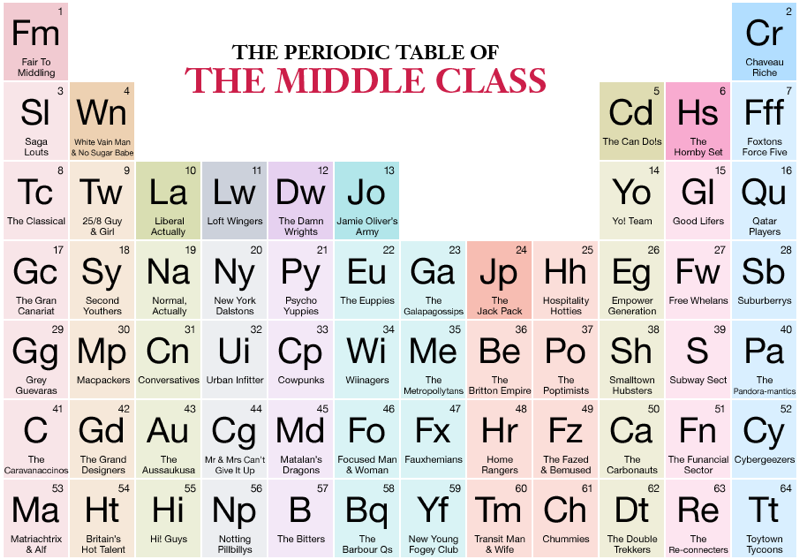
Middle Class, Periodic Table of
From the Middle Class Handbook, The Periodic Table of Middle Class (An illustrated guide to what the British middle classes say, do & buy):
| Year: 2012 | PT id = 558, Type = review |
Scientific American: The Quest for the Periodic Table
From Scientific American, a series of original articles (scanned) dealing with the development of the periodic table dating from 1861 to 1998.
Edited by Eric Scerri.
| Year: 2012 | PT id = 559, Type = data misc |
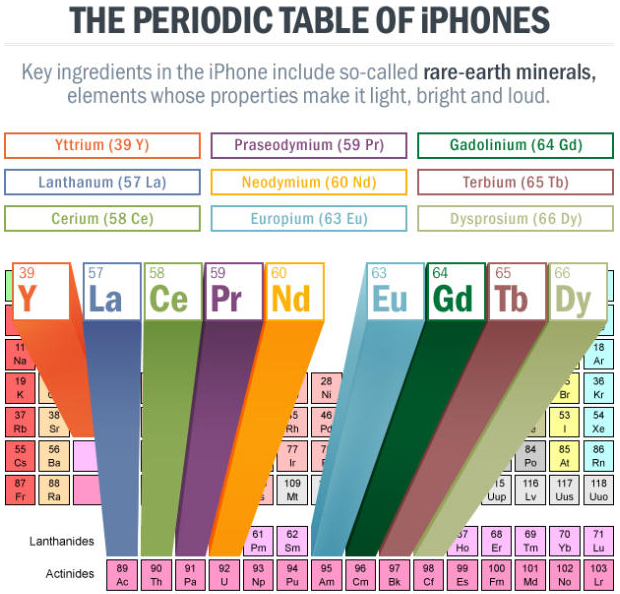
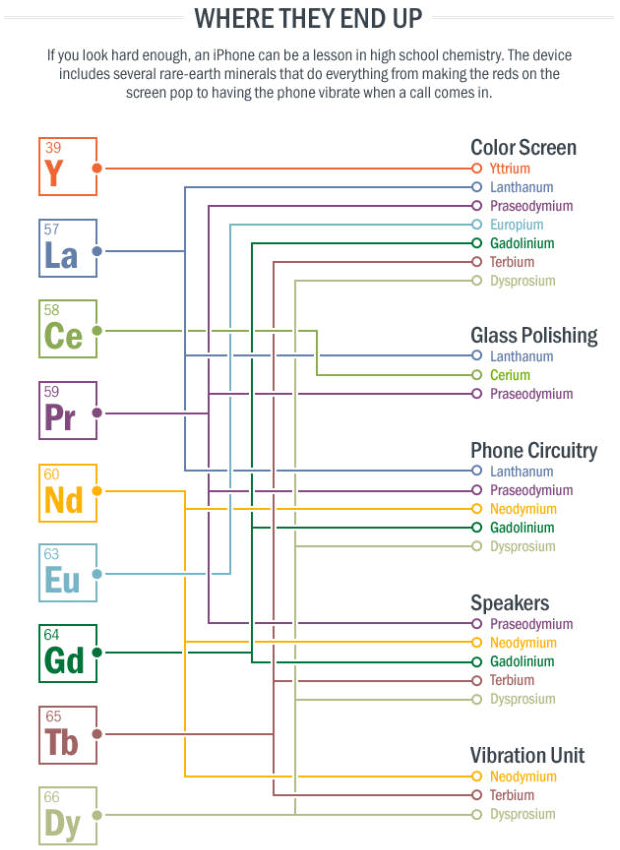
iPhone, Periodic Table of
An article in Scientific American Digging for Rare Earths: The Mines Where iPhones Are Born.
"About 60 miles southwest of Las Vegas, in a mine some 500 feet deep, the beginnings of an iPhone come to life. But the sleek, shiny iPhone is far, far removed from the rocks pulled out of this giant hole, which looks like a deep crater on the moon. Inside the rocks from this mine are rare-earth minerals, crucial ingredients for iPhones, as well as wind turbines, hybrid cars, and night-vision goggles. Minerals such as neodymium are used in magnets that make speakers vibrate to create sound. Europium is a phosphor that creates a bright red on an iPhone screen. Cerium gets put into a solvent that workers use to polish devices as they move along the assembly line, etc.":
Thanks to Eric Scerri for the tip!
See the website EricScerri.com and Eric's Twitter Feed.
| Year: 2012 | PT id = 765, Type = formulation spiral |
Ato Circular Periodic Table by Ramanpreet Singh Jandu
Ramanpreet Singh Jandu writes:
"The present invention relates to a device for the understanding of the periodic table and the structure of the atom together in a better way.
The device consists of seven concentric circular disks rotatable about their centre, wherein the size of the disks increases from the centre to the end like that in the structure of the atom.
Each disk is marked so as to form the sub-blocks and each disk in itself represents the periods of the periodic table.
The disks are divided into sub-blocks and labeled with elements at back side as well.
Thus the Ato Circular Periodic Table, as the name suggests, is the combination of atomic structure of the atom and that of the periodic classification of the elements."
Read more in the pdf file which describes the new formulation in detail.
| Year: 2012 | PT id = 925, Type = misc data |
Atoms, Orbitals & The Periodic Table
One of several animations and explanations/realisations of quantum physics from Data-Burger, scientific advisor: J. Bobroff, with the support of: Univ. Paris Sud, SFP, Triangle de la Physique, PALM, Sciences à l'Ecole, ICAM-I2CAM.
Mark Leach writes:
"What I particularly like about this video is that it shows the quantum fuzziness of the atoms. This explains/shows how and why induced-dipole/induced-dipole (London force) interactions occur, an important class of van der Waals interaction. At any moment, the electron distribution is not perfectly spherical, which means that there is an instantaneous dipole on the atom. This instantaneous dipole is able to induce a dipole on an adjacent atom, with the effect that the two atoms are attracted when they touch. It is as if atoms are 'sticky' like Velcro.
"This effect explains why the Group 18 noble gas elements are able to form liquids and solids [not He] at low temperatures, and why non-polar molecules, such as P4, S8 and hydrocarbons are able to condense."
| Year: 2012 | PT id = 1102, Type = review formulation |
Eric Scerri Lecture, Dedicated to Fernando Dufour
Dr. Eric Scerri from the Chemistry Department at UCLA giving a distinguished invited lecture at the Oscar Peterson auditorium of Concordia University, in Montreal. The topic is the history and iconic nature of the Periodic Table.
Thanks to Eric Scerri – who appears – for the tip!
See the website EricScerri.com and Eric's Twitter Feed.
| Year: 2012 | PT id = 1163, Type = misc |
Itch: A Book & TV Drama About a Boy Who Collects The Chemical Elements
Meet Itch – an accidental, accident-prone hero. Science is his weapon. Elements are his gadgets.
All-action adventure perfect for fans of Alex Rider and Young Bond.
Itchingham Lofte – known as Itch – is fourteen, and loves science, especially chemistry. He's also an element-hunter: he's collecting all the elements in the periodic table. Which has some interesting and rather destructive results in his bedroom.
Then, Itch makes a discovery. A new element, never seen before. At first no one believes him – but soon someone hears about the strange new rock and wants it for himself. And Itch and his family are catapulted into a breathless adventure with terrifyingly high stakes...
The debut novel from BBC radio presenter Simon Mayo.
Children's book Itch by Simon Mayo is available from Amazon and all good bookshops, ISBN: 9780552565509. The book has been made into a TV Series, filmed in Western Australia, and is available on the BBC iPlayer.

 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
© Mark R. Leach Ph.D. 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com
This free, open access web book is an ongoing project and your input is appreciated.