Periodic Table |
 |
 |
 |
 |
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
The INTERNET Database of Periodic Tables
There are thousands of periodic tables in web space, but this is the only comprehensive database of periodic tables & periodic system formulations. If you know of an interesting periodic table that is missing, please contact the database curator: Mark R. Leach Ph.D. The database holds information on periodic tables, the discovery of the elements, the elucidation of atomic weights and the discovery of atomic structure (and much, much more).
Periodic Tables from the year 2018:
| Year: 2018 | PT id = 494, Type = data formulation |
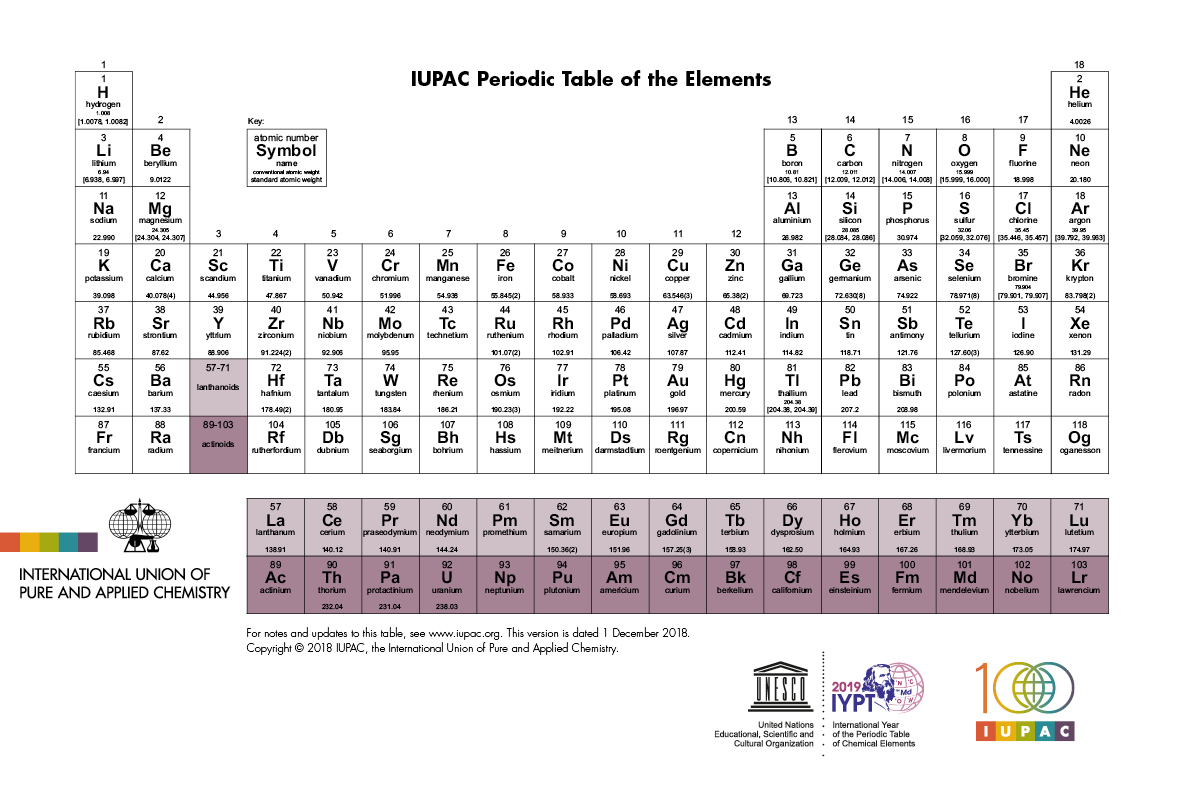
IUPAC Periodic Table of The Elements
The 1 Dec 2018 IUPAC (International Union of Pure and Applied Chemistry) Periodic Table of The Elements. For updates to this table click here.
By virtue of its work in relation with the chemical elements, IUPAC can dispense a periodic table that is up-to-date. IUPAC involvement covers various aspects of the table and data that it unveils, and several reports and recommendations, some quite recent, attest of that input. In particular, IUPAC is directly involved in the following:
- establishing the criteria for a new element discovery
- defining the structure of a temporary name and symbol
- assessing claims resulting in the validation and assignation of an element discovery
- coordinating the naming of a new element, involving the research laboratory and allowing for public comments
- setting up precise rules for how to name a new element
- defining Group 1-18 and collective names
- determining which elements belong to Group 3
- regularly reviewing standard atomic weights
| Year: 2018 | PT id = 766, Type = data |
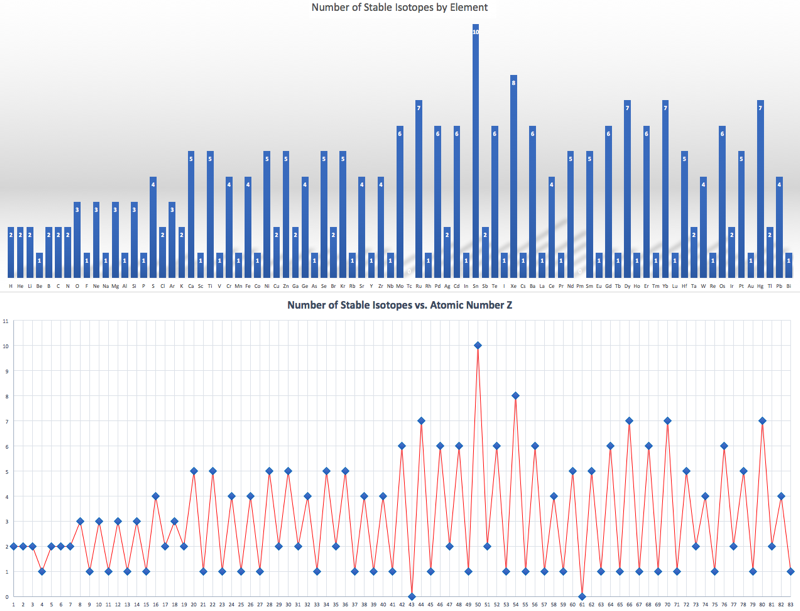
Number of Stable Isotopes by Element
When plotting the number of stable isotopes against element, and against atomic number Z, it is clear that elements with an even atomic number are likely to have more stable isotopes (average 4.9) than elements with an odd atomic number (average 1.3). Click here for the Excel file. There is a Wikipedia page here.
The effect is striking in graphical form:
By Mark Leach
| Year: 2018 | PT id = 774, Type = formulation data |
First Ionisation Energy to the Standard Form Periodic Table
There is debate amongst the cognoscenti about the 'best' representation of the periodic table, and how this 'best' formulation can be explained by [rationalized by] quantum mechanics (QM).
Many feel that the Janet PT formulation, the 'Left Step', is the ideal QM PT, but this formulation does not show periodicity very well, and there are issues with the placement of H, He, Be which spill over into questions about their placement in the standard form PT (the periodic table used in classrooms and textbooks around the world).
However, it is possible to get to the conventional standard form PT directly from the first ionisation energy data, where the 1st ionisation energy is the energy required to convert a gas phase atom (M) into its gas phase positive ion plus electron.
M(g) → M+(g) + e–
The process involves:
- taking the 1st ionisation data plot for the elements H to Xe (Z = 1 to 36)
- rotate 90° clockwise and stretch
- move the atoms horizontally into columns
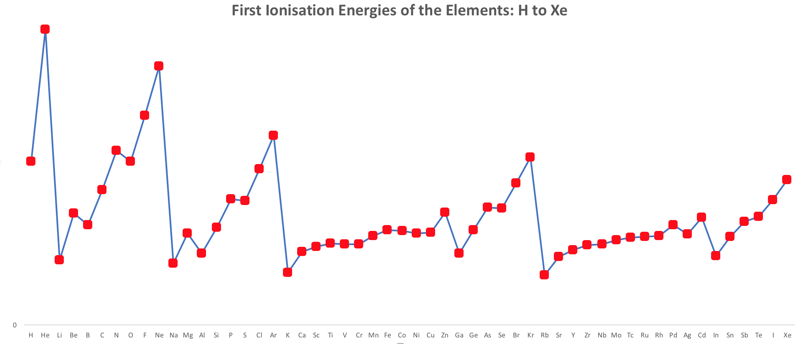
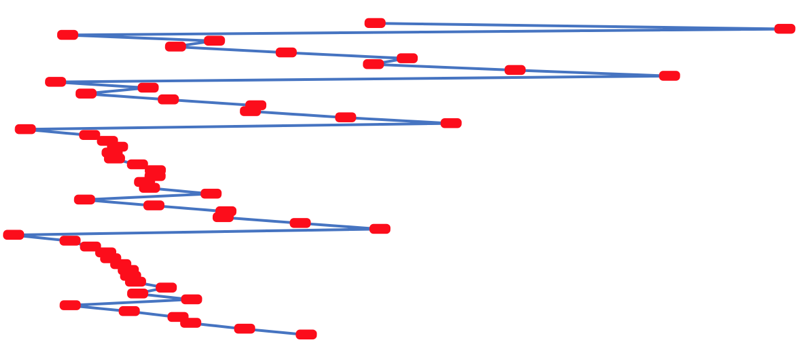
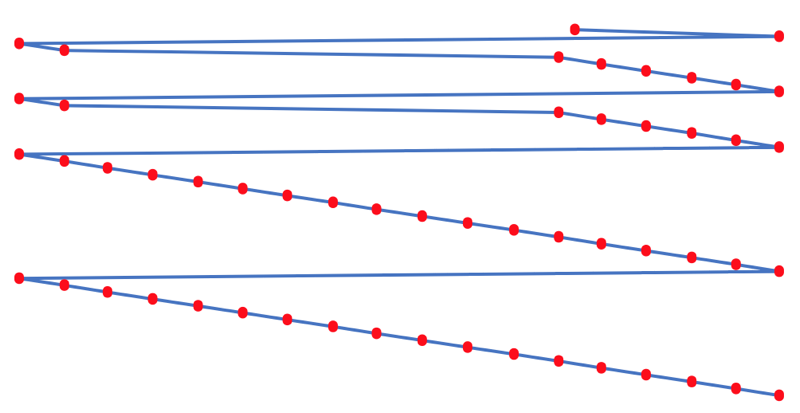
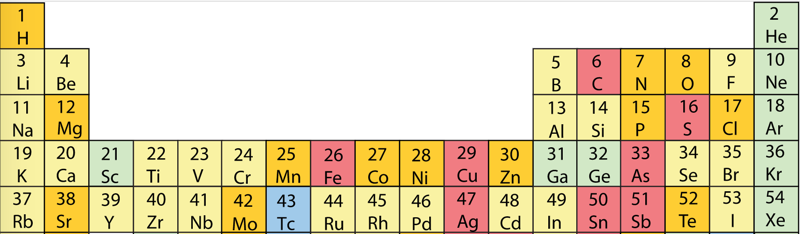
Note that a similar logic can be applied to atomic radius and electronegativity data.
However, there are issues about the measurement of atomic radius, because atoms are 'soft at their edges', and gas phase atomic radius is not precisely defined. And, electronegativity is a derived parameter.
By Mark Leach
| Year: 2018 | PT id = 779, Type = formulation 3D |
Stowe-Janet-Scerri Periodic Table (Extended)
Stowe's A Physicist's Periodic Table was published in 1989, and is a famous & well respected formulation of the periodic table.
Since 1989 quite a number of elements have been discovered and Jeries A. Rihani has produced an updated and extended version in 2017. This has been further updated, below. Click for the full size .pdf version:
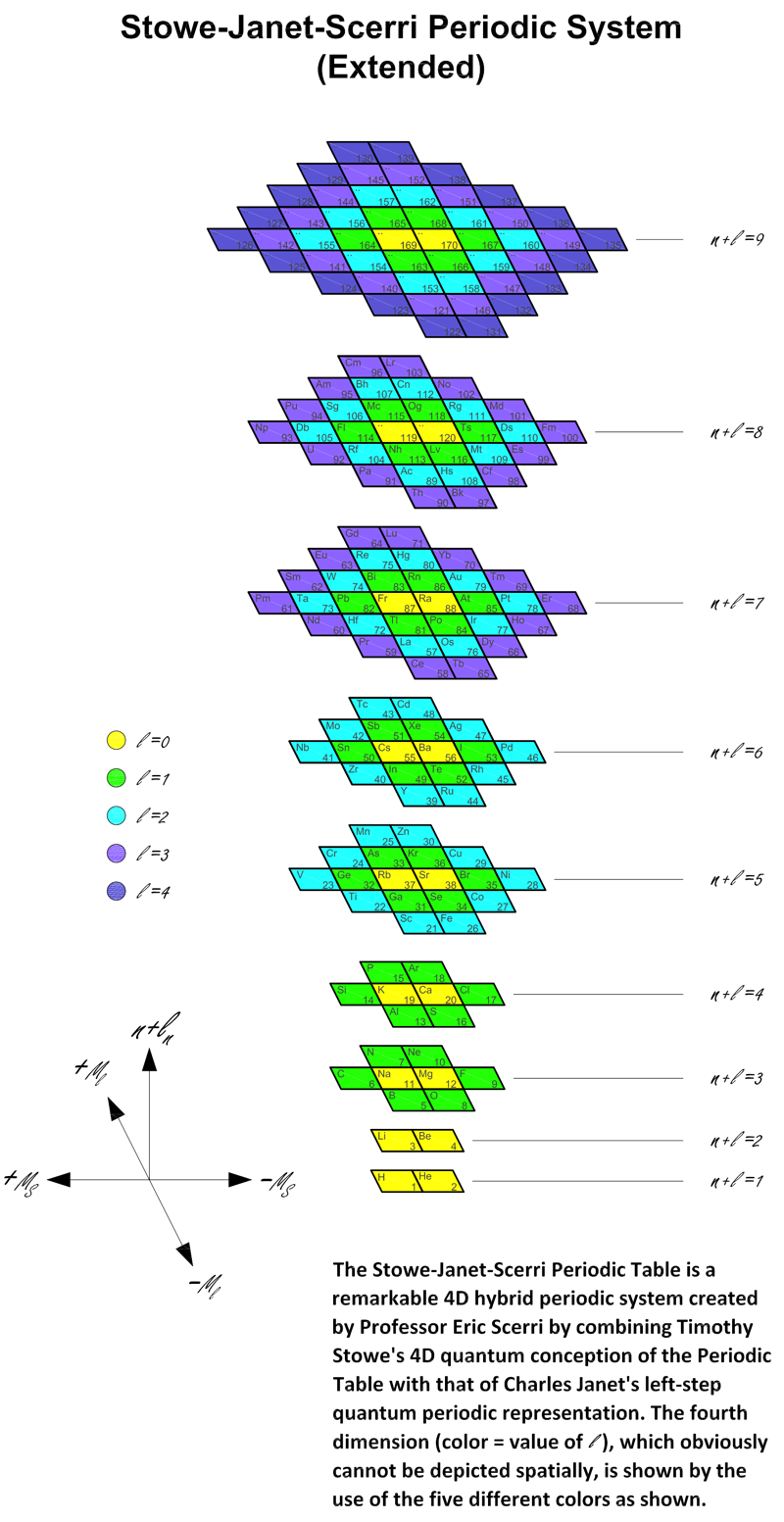
| Year: 2018 | PT id = 780, Type = formulation spiral |
Chemical Galaxy III
Updated from Philip Stewart's Chemical Galaxy II, version III shows all the recently discovered elements, including: 117 (Ts) and 118 (Og).
Click here for the full size .PDF version (which gives more data):

| Year: 2018 | PT id = 899, Type = formulation |
Hoyau's Periodic Table Formulations
From Davy Hoyau in France, a selection of periodic table formulations. Davy writes:
"I can explain how i came up to this solution, to better represent graphically the mathematics of the nature. The great advantage of that solution is to make visible how important are the subrings, and not the rings, of the electronics layers. That's why we can call this representation "the table of subrings".
"They seem to be responsible of element chemistry. However, most of time the atom is represented with the full layer, of 2, 8, 18, 32 available positions for determined quanta of energy. But the most important thing is to see how strange is the table when we only have to count the additional electrons needed to finish to fill the layers, following this suit ; 2, 6, 10, 14 (because 2+6=8, 8+10=18, and 18+14=32).
"The traditional representation make easier to follow, vertically, the type of element. For example, follow the column of copper, silver, gold & roentgenium. They have the most conductivity elements of their subrings.
"Also we can follow the column of the noble gases. And that is a surprise to find a noble gas at the only first subring of the first ring, but at all the second subrings of all other rings. That let imagine an extremely innovative way to fill the free locations of energy, not simple by adding marbles on a sequence of positions, but more like a type of musical chair game (i cant be more precise actually). That show how are possibles the "errors" of filling that this 3D representation can't show, if you watch attentively the known data on the filled layers. For example, vanadium 23 is 2-8-11-2, and chrome 24 is 2-8-13-1."
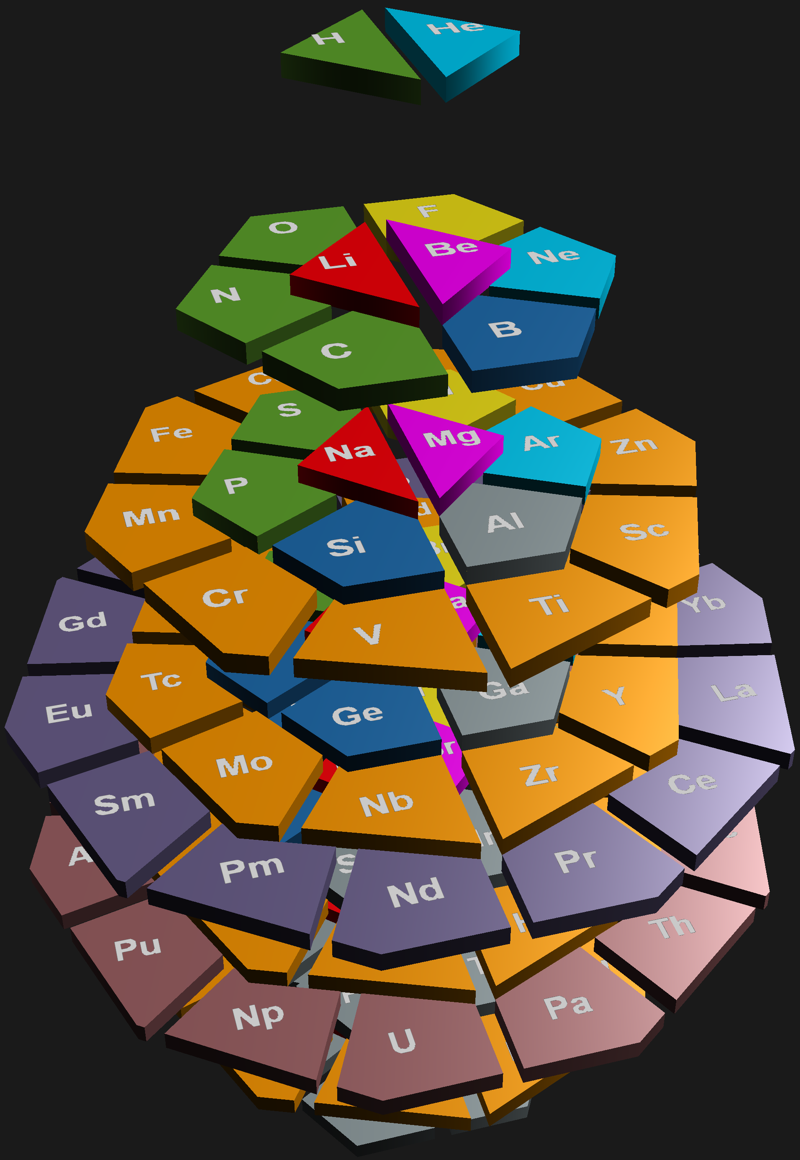
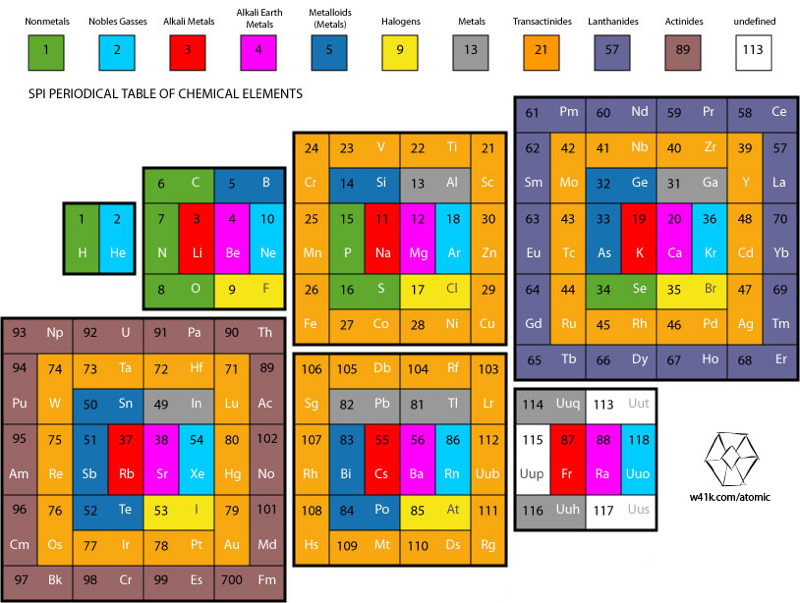
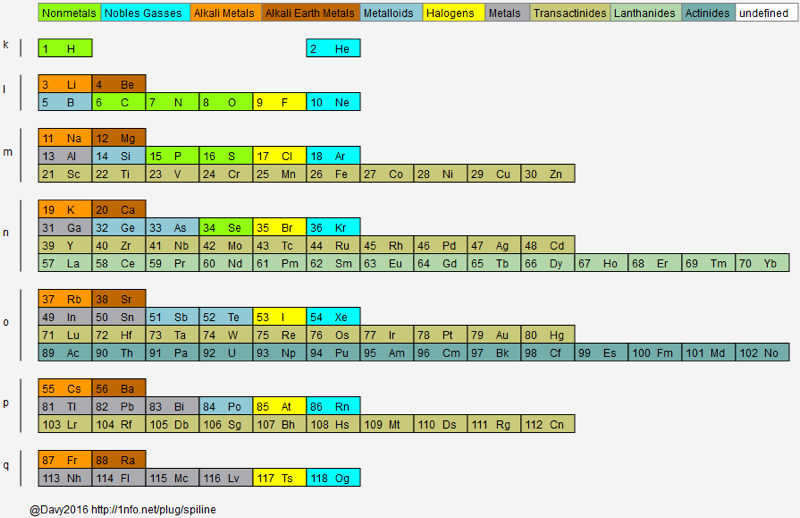
Davy has also provided some links to his ideas on the web:
- http://1nfo.net/plug/spitable
- http://1nfo.net/plug/spitablesvg
- http://1nfo.net/plug/spiline
- http://tlex.fr/frame/scene/17
- http://tlex.fr/frame/scene/17
| Year: 2018 | PT id = 900, Type = formulation review |
Race to Invent the Periodic Table
From PBS Digital Studios, a short-but-fast-moving video about the development of the periodic table during the 19th century, and a discussion about gallium:
Thanks to Eric Scerri for the tip!
See the website EricScerri.com and Eric's Twitter Feed
| Year: 2018 | PT id = 907, Type = review |
Alphabet of Chemistry
A BBC World Service radio program, first broadcast Tue 23 Jan 2018.
The Russian chemist Dmitri Mendeleev attempted nothing less than to pull apart the fabric of reality and expose the hidden patterns that lie beneath everything in existence, from shoes and ships and sealing wax to cabbages and kings. The result was something known to almost everyone who has ever been to school: the Periodic Table of the elements. But why this particular arrangement? And why is it still the foundation of chemistry?
The presenter Quentin Cooper is joined by:
- Hugh Aldersey-Williams, who since he was a teenager, has collected samples of elements and has drawn on his samples and knowledge to write Periodic Tales: The Curious Lives of the Elements.
- Michael Gordin, Professor of History at Princeton University and the author of A Well-Ordered Thing: Dmitri Mendeleev and the Shadow of the Periodic Table.
- Ann Robinson, Historian at the University of Massachusetts studying the development of the periodic table.
- Eugene Babaev, Professor of Chemistry at Moscow State University who maintains both Russian and English websites on Mendeleev and his work.

| Year: 2018 | PT id = 913, Type = formulation spiral |
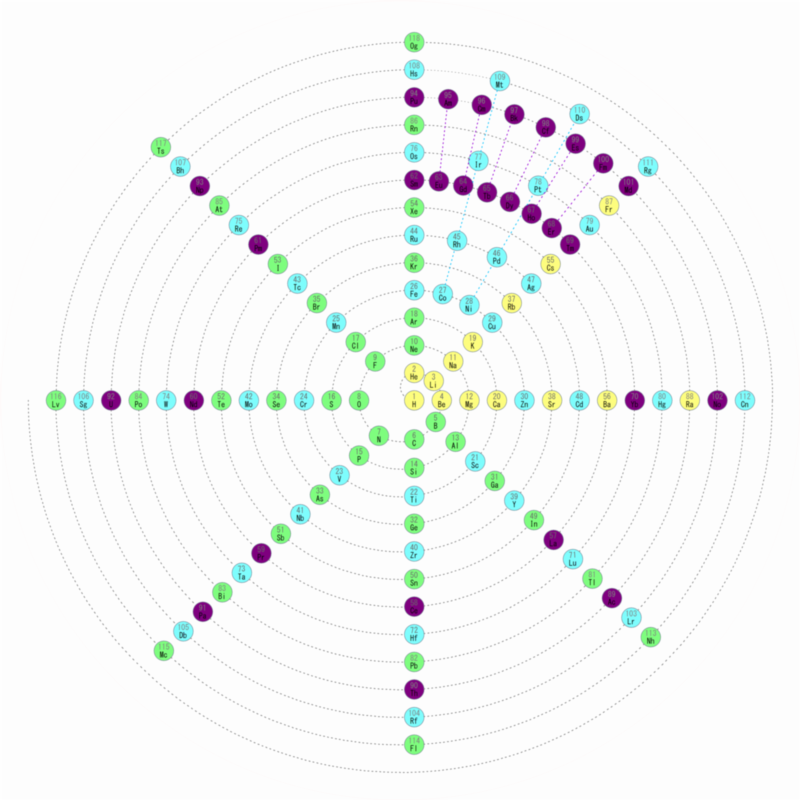
Nawa–Scerri Octagonal Periodic System
A spiral periodic table formulation by Nawa, called the Nawa–Scerri Octagonal Periodic System.
Click here for a larger version:
| Year: 2018 | PT id = 914, Type = formulation 3D |
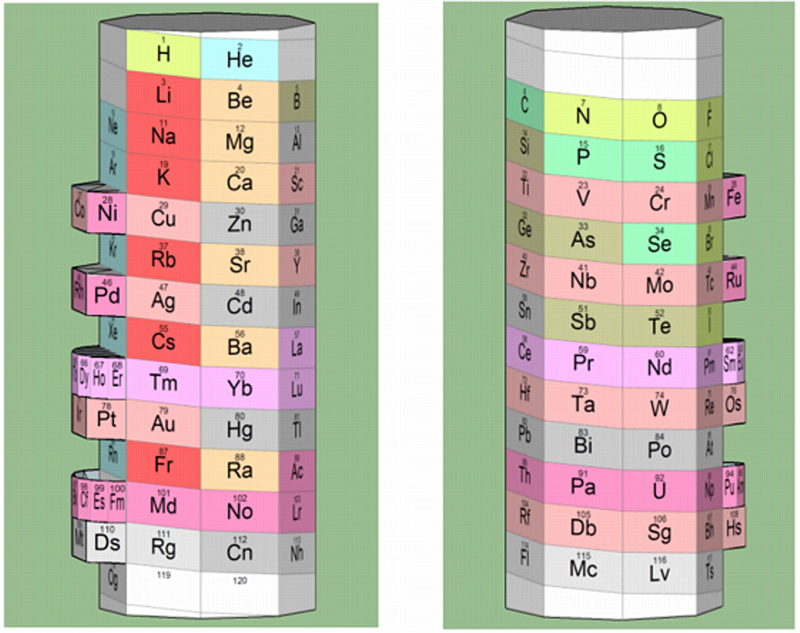
Nawa's 3-D Octagonal Pillar
A 3-D octagonal pillar periodic table model by Nawa, "acccording to Scerri's reverse engineering [of] Mendeleev's 8-column table":
| Year: 2018 | PT id = 915, Type = formulation |
Scerri's Reverse Engineered Version of Mendeleev's Eight Column Table
Eric Scerri has updated – reverse engineered – the classic Mendeleev Table, here, here & here:
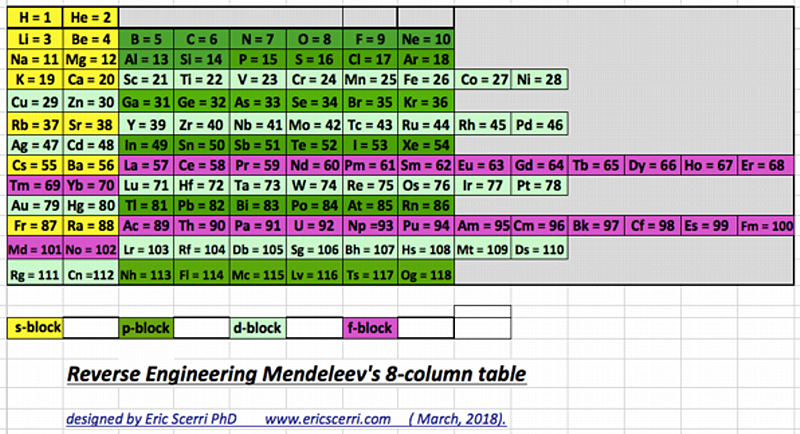
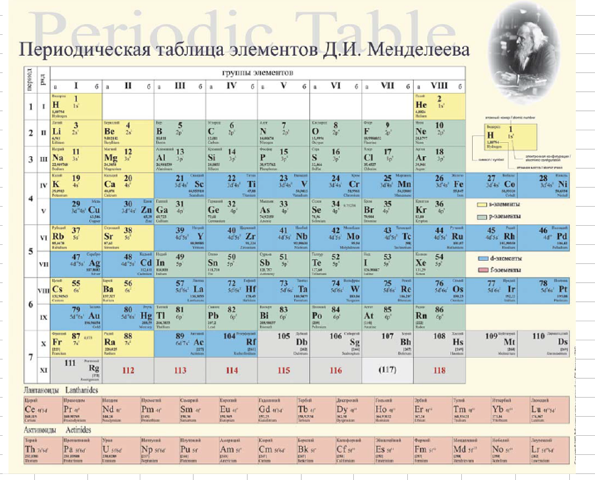
Thanks to Eric Scerri for the tip!
See the website EricScerri.com and Eric's Twitter Feed
| Year: 2018 | PT id = 918, Type = misc |
Lego® Periodic Table
Welcome to the Lego® Periodic Table of Elements.
Students and faculty at Spring Arbor University (MI), students and faculty at Hardin Valley Academy (TN), and members of the community surrounding Spring Arbor have worked together to construct a periodic table entirely out of Lego® blocks.
On each elemental square has been placed a small Lego® creation that somehow represents the element on which it is positioned.
You can learn a little bit about each element by clicking on its particular square on the table below.
| Year: 2018 | PT id = 919, Type = review |
Mendeleev to Oganesson: A Multidisciplinary Perspective on the Periodic Table
Since 1969, the international chemistry community has only held conferences on the topic of the Periodic Table three times, and the 2012 conference in Cusco, Peru was the first in almost a decade. The conference was highly interdisciplinary, featuring papers on geology, physics, mathematical and theoretical chemistry, the history and philosophy of chemistry, and chemical education, from the most reputable Periodic Table scholars across the world. Eric Scerri and Guillermo Restrepo have collected fifteen of the strongest papers presented at this conference, from the most notable Periodic Table scholars. The collected volume will contain pieces on chemistry, philosophy of science, applied mathematics, and science education.
Eric Scerri is a leading philosopher of science specializing in the history and philosophy of chemistry and especially the periodic table. He is the author of numerous OUP books including A Tale of Seven Scientists and a New Philosophy of Science (2016) and The Periodic Table: A Very Short Introduction (2012). Scerri has been a full-time lecturer at UCLA for the past eighteen years where he regularly teaches classes in history and philosophy of science.
Guillermo Restrepo is a chemist specializing in mathematical and philosophy of chemistry with more than sixty scientific papers and book chapters on these and related areas. Restrepo was a professor of chemistry at the Universidad de Pamplona (Colombia) between 2004 and 2017, and since 2014 has been in Germany as an Alexander von Humboldt Fellow at Leipzig University and more recently as researcher at the Max Planck Institute for Mathematics in the Sciences.
Preface
1. Heavy, Superheavy...Quo Vadis?
2. Nuclear Lattice Model and the Electronic Configuration of the Chemical Elements
3. Amateurs and Professionals in Chemistry: The Case of the Periodic System
4. The Periodic System: A Mathematical Approach
5. The "Chemical Mechanics" of the Periodic Table
6. The Grand Periodic Function
7. What Elements Belong in Group 3 of the Periodic Table?
8. The Periodic Table Retrieved from Density Functional Theory Based Concepts: The Electron Density, the Shape Function and the Linear Response Function
9. Resemioticization of Periodicity: A Social Semiotic Perspective
10. Organizing the Transition Metals
11. The Earth Scientist's Periodic Table of the Elements and Their Ions: A New Periodic Table Founded on Non-Traditional Concepts
12. The Origin of Mendeleev's Discovery of the Periodic System
13. Richard Abegg and the Periodic Table
14. The Chemist as Philosopher: D. I. Mendeleev's "The Unit" and "Worldview"
15. The Philosophical Importance of the Periodic Table
| Year: 2018 | PT id = 920, Type = formulation 3d |
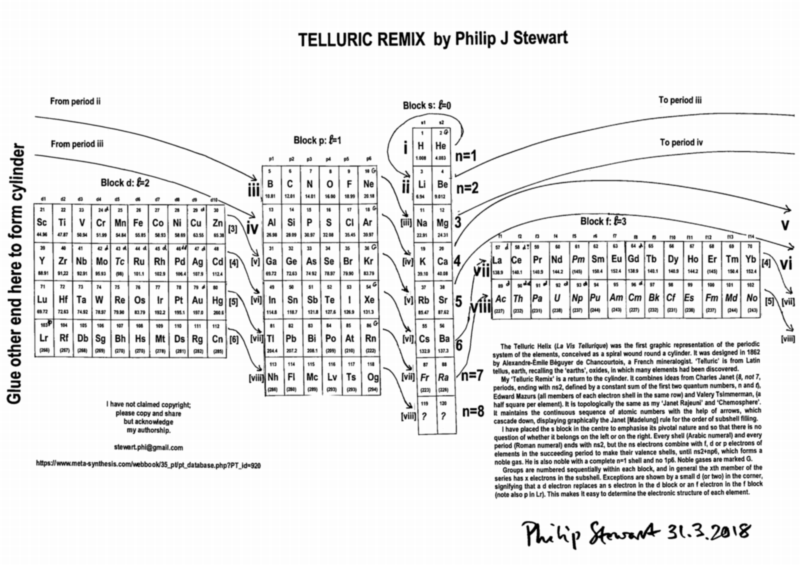
Telluric Remix
Philip Stewart writes:
The Telluric Helix (La Vis Tellurique) was the first graphic representation of the periodic system of the elements, conceived as a spiral wound round a cylinder. It was designed in 1862 by Alexandre-Émile Béguyer de Chancourtois, a French mineralogist. 'Telluric' is from Latin tellus, earth, recalling the 'earths', oxides, in which many elements had been discovered.
My 'Telluric Remix' is a return to the cylinder. It combines ideas from Charles Janet (8, not 7, periods, ending with ns2, defined by a constant sum of the first two quantum numbers, n and l), Edward Mazurs (all members of each electron shell in the same row) and Valery Tsimmerman, (a half square per element).
- The Telluric Remix is topologically the same as my 'Janet Rajeuni' and 'Chemosphere': it maintains the continuous sequence of atomic numbers with the help of arrows, which cascade down, displaying graphically the Janet [Madelung] rule for the order of subshell filling.
- I have placed the s block in the centre to emphasise its pivotal nature and so that there is no question of whether it belongs on the left or on the right. Every shell (Arabic numeral) and every period (Roman numeral) ends with ns2, but the ns electrons combine with f, d or p electrons of elements in the succeeding period to make their valence shells, until ns2+np6, which forms a noble gas. Helium, He, is also noble with a complete n=1 shell and no 1p6.
- Noble gases are marked G. Groups are numbered sequentially within each block, and in general the xth member of the series has x electrons in the subshell. Exceptions are shown by a small d (or two) in the corner, signifying that a d electron replaces an s electron in the d block or an f electron in the f block (note also p in Lr). This makes it easy to determine the electronic structure of each element.
- Click here for a larger version.
The printable version is available (click here for the full size version) to make your own:
I have not claimed copyright; please copy and share but acknowledge my authorship. stewart.phi@gmail.com
| Year: 2018 | PT id = 923, Type = review data |
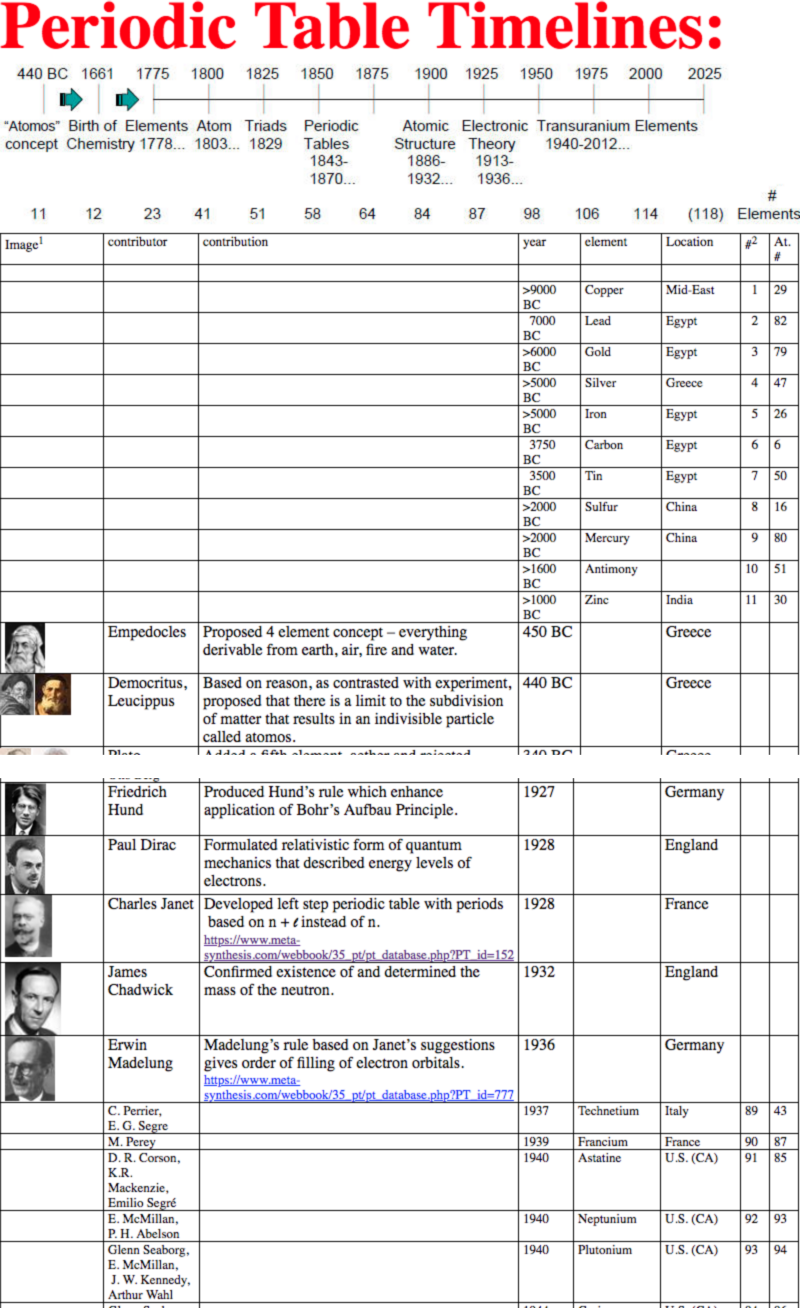
Timelines, of The Periodic Table
By Steven Murov, a chronology of the events that have resulted in our present periodic table of the elements and a celebration of the 150th anniversary of the Mendeleev (birthday, 02/08/1834) periodic table (1869).
Recursively, the Murov website has many links to this [Chemogenesis] website.
Thanks to Eric Scerri for the tip!
See the website EricScerri.com and Eric's Twitter Feed
| Year: 2018 | PT id = 929, Type = data |
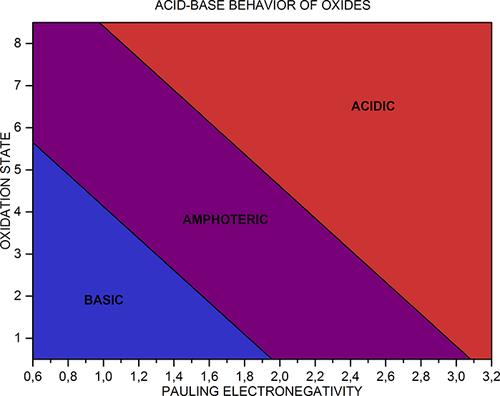
Acid-Base Behavior of 100 Element Oxides
Acid-Base Behavior of 100 Element Oxides: Visual and Mathematical Representations by Mikhail Kurushkin and Dmitry Kurushkin. J. Chem. Educ. 95, 4, 678-681.
A novel educational chart that represents the acid-base behavior of 100 s-, p-, d-, and f-element oxides depending on the element's electronegativity and oxidation state was designed. An updated periodic table of said oxides was developed. A mathematical criterion based on the chart was derived which allows prediction of the behavior of unfamiliar oxides:
| Year: 2018 | PT id = 930, Type = misc |
Stamps Commemorating Yuri Organeson
Stamps Commemorating Yuri Organeson, Issued by Armenia, Dec 28th, 2017

| Year: 2018 | PT id = 932, Type = data misc |
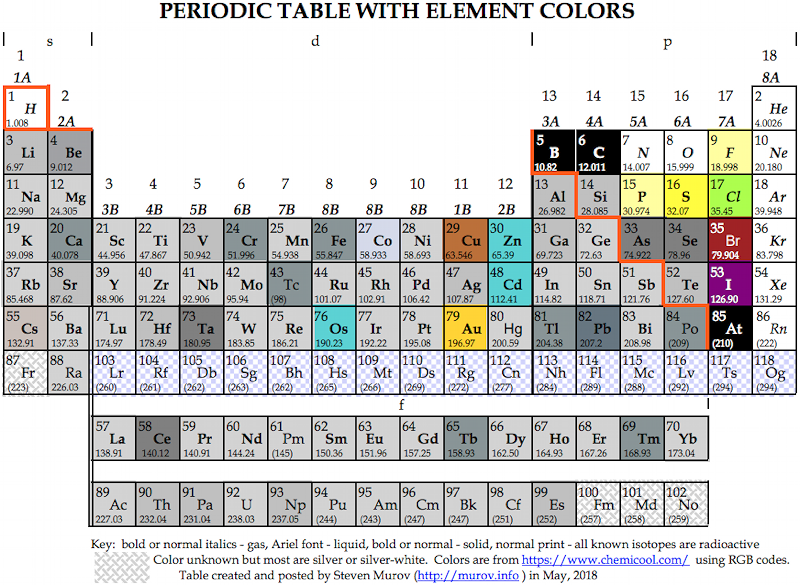
Murov's Colours of the Elements
Steven Murov writes :
"The element squares of this periodic table have colors resembling the actual colors of the elements. The table provides insight useful for helping to distinguish metals and non-metals as well as observations on elements of unusual color. The colors were taken from https://www.chemicool.com/ and applied with RGB codes."
The tables are available online at:
| Year: 2018 | PT id = 933, Type = data misc |
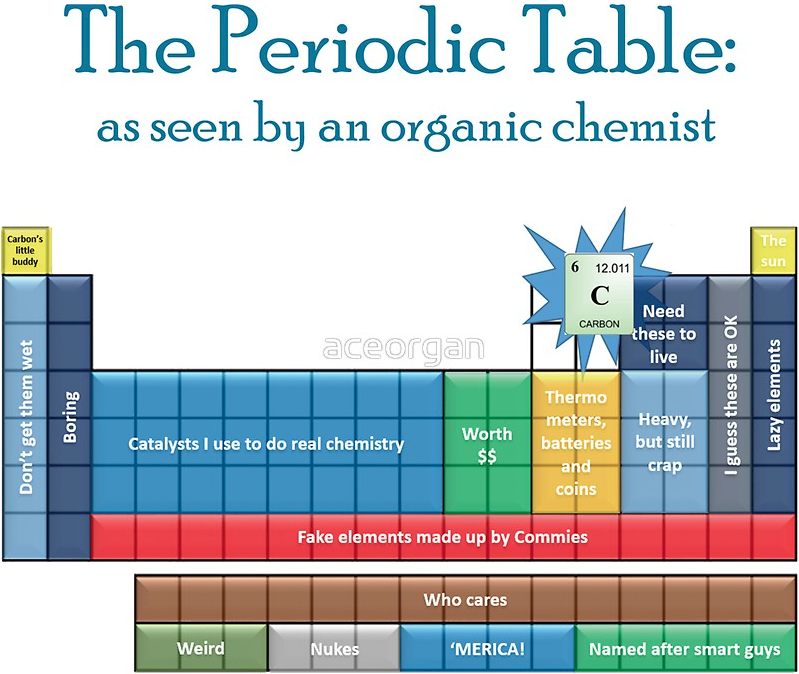
Organic Chemist's Periodic Table (another one)
The Periodic Table as seen by an Organic Chemist... a T-Shirt by REDBUBBLE:
Thanks to Marcus Lynch for the tip!
| Year: 2018 | PT id = 936, Type = formulation 3D |
Sistema Periódico Binodico
By Julio Antonio Gutiérrez Samanez, who writes:
"Sistema Periódico Binodico. Nuevo Paradigma Matematizado. I have followed the work of the wise Mendeleev, of Emil de Chancourtois, of Charles Janet; inspired by the work of my countryman Dr. Oswaldo Baca Mendoza. It is in Spanish but soon I will have the English version."
| Year: 2018 | PT id = 938, Type = formulation |
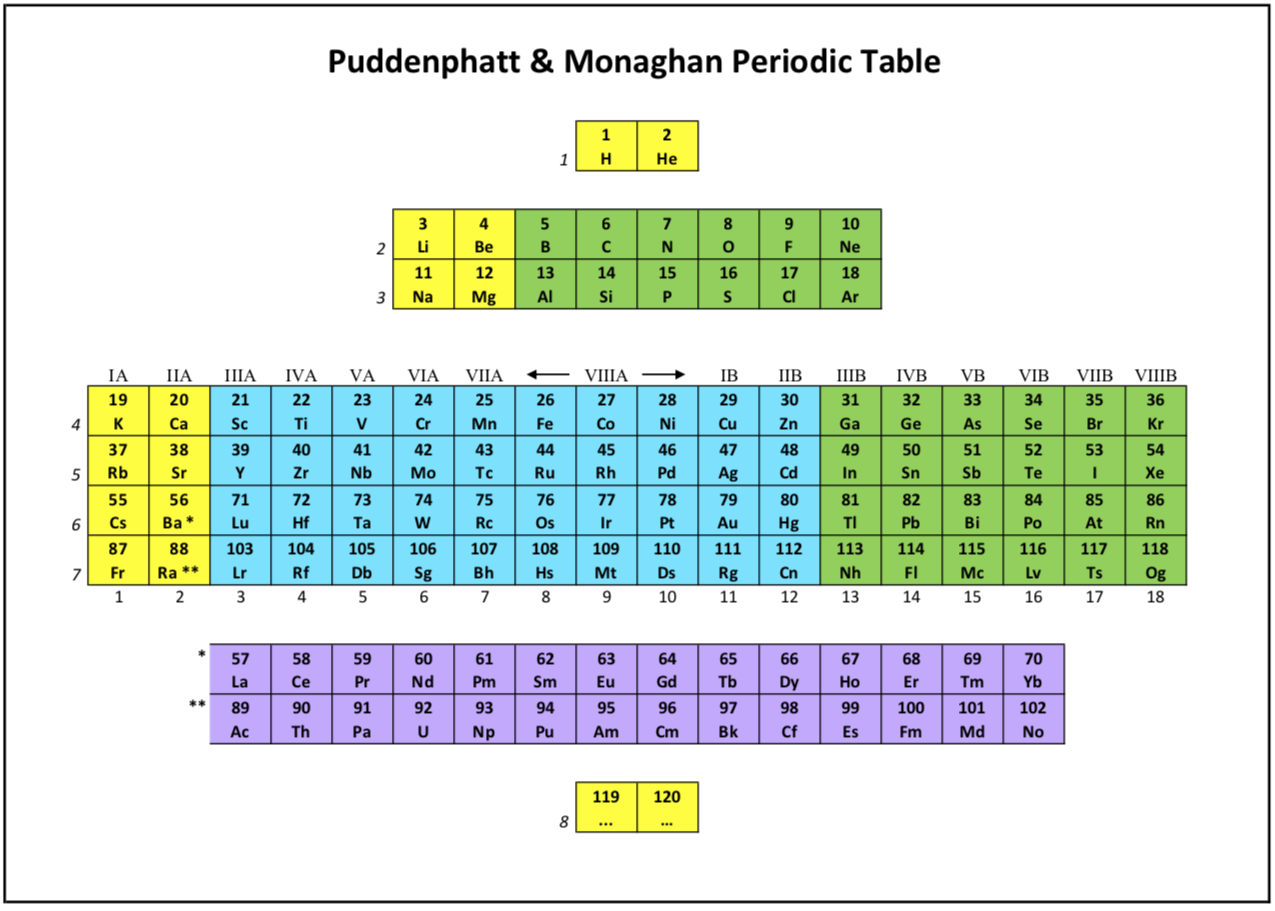
Puddenphatt & Monagham Periodic Table
Jeries Rihani's version of R. J. Puddenphatt and P. K. Monaghan, published in1989, but is not an exact copy. The differences are as follows:
- It adds color: YELLOW for the s-block, GREEN for the p-block, BLUE for the d-block and PURPLE for the f-block.
- It avoids being congested since it excludes the electronic configurations of the elements.
- It is updated and includes the atomic numbers 119 and 120.
- It shows that it is symmetrical around the vertical axis.
- The f-block, like all the other blocks, ends with even atomic numbers.
Puddephatt and Monaghan say "their table is after Philips and Williams":
Ref, Phillips CSG & Williams RJP 1965, Inorganic Chemistry, I: Principles and Non-metals, Clarendon Press, Oxford, p. 40.
| Year: 2018 | PT id = 942, Type = misc |
I Wear This Shirt Periodically T-Shirt
From Shared.com, a "I Wear This Shirt Periodically" T-Shirt:
Thanks to Clare Cheetham for the tip!
| Year: 2018 | PT id = 946, Type = formulation 3D |
Periodical System (Binodic Form): a new mathematical paradigm
By Julio Antonio Gutiérrez Samanez, who writes:
"System devised and prepared by the Peruvian chemical engineer, Julio Antonio Gutiérrez Samanez, deals with a new conception of Mendeleev's Law as a mathematical function and a new description of the process of forming the series of chemical elements according to mathematical laws and dialectical processes of changes quantitative and qualitative under a dynamic spiral architecture in 3D, which is postulated as a new scientific paradigm."
| Year: 2018 | PT id = 948, Type = formulation spiral |
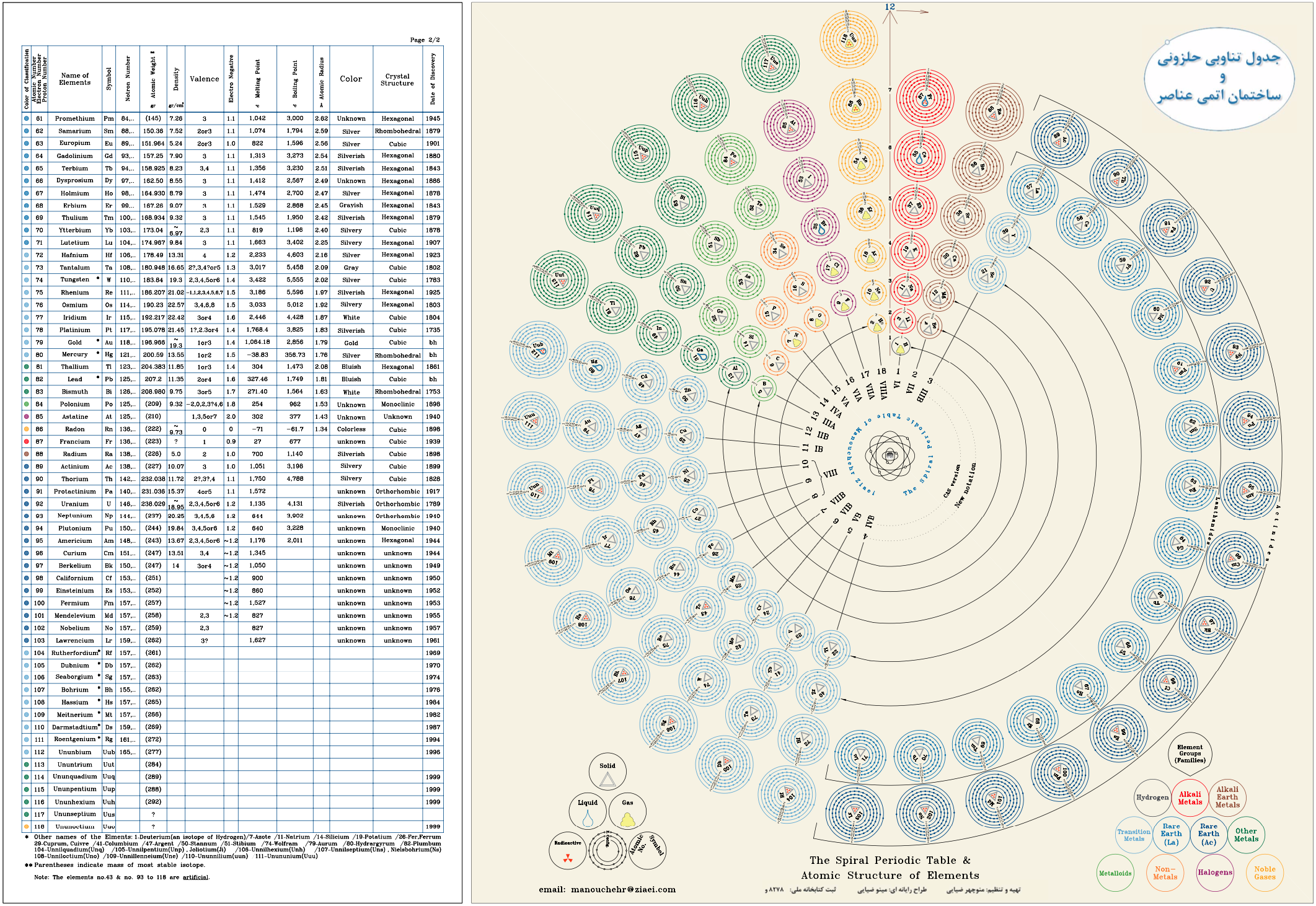
Ziaei's Circular Periodic Table
Minoo Ziaei writes:
"My father, Manouchehr Ziaei, has an interesting design of the periodic table, which I helped him draw using AutoCAD. He is very keen in introducing [this formulation] to potential interested viewers, he was recommended to visit [the Chemogenesis Database of Periodic Tables] website."
Click image to enlarge:
| Year: 2018 | PT id = 949, Type = formulation data misc |
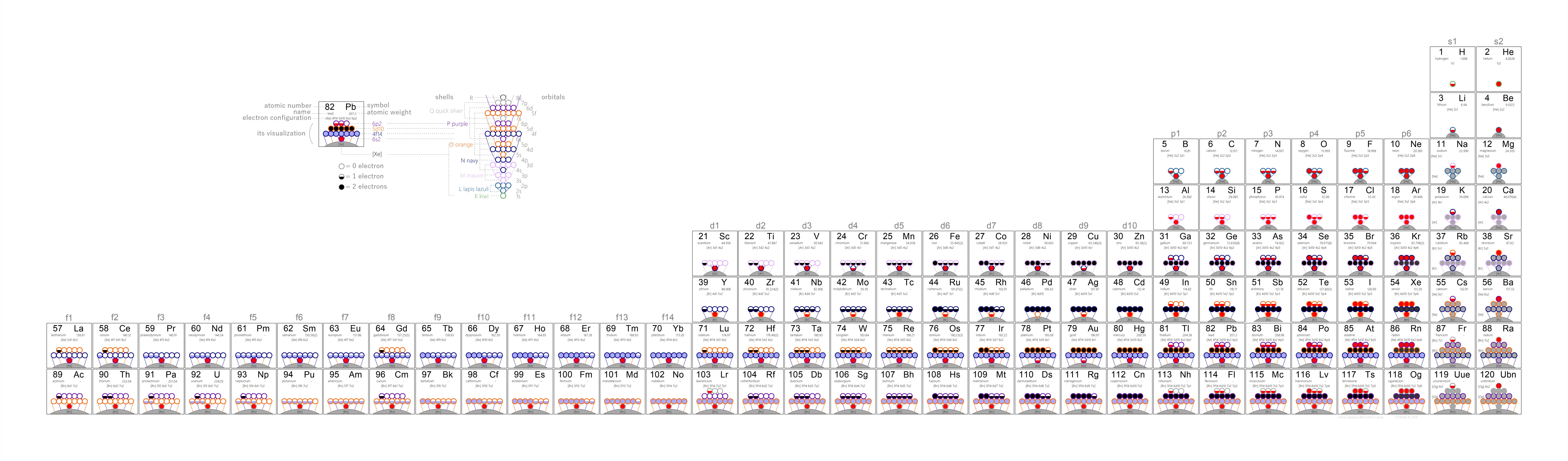
Nawa's V.E.T. Periodic Table & Hourglass
Nagayasu Nawa, the prolific designer of periodic tables, here and here, has come up with an orbital filling periodic table and a corresponding hourglass animation. Nawa writes:
"I have turned the v.e.c. PT into the GIF animation that I call the electron hourglass, 1 second for each element. It takes 120 seconds from 1H to 120 Ubn. I have coloured orbital with colour derived from each shell's name, such as:
- K kiwi
- L lapis lazuli
- M mauve
- N navy
- O orange
- P purple
- Q quick silver"
Click image to enlarge.
| Year: 2018 | PT id = 950, Type = data misc |
Waterloo Periodic Table Project/Projet Tableau Périodique
To celebrate the International Year of Chemistry (IYC), Chem 13 News magazine together with the University of Waterloo's Department of Chemistry and the Faculty of Science encouraged chemistry educators and enthusiasts worldwide to adopt an element and artistically interpret that element.
The project created a periodic table as a mosaic of science and art. Students from all Canadian provinces and territories, 20 U.S. states and 14 countries researched, created and designed the elemental tiles. We created a poster, wall mural and a mobile app. The app includes the creative process behind each tile along with basic atomic properties of the element. The free app work to truly highlight the artistic expression of the Periodic Table Project. Thank you to all the teachers and students who participated in the collaborative Periodic Table Project.
Read more on the University of Waterloo website.
Click here image to enlarge the PT below.
| Year: 2018 | PT id = 951, Type = formulation |
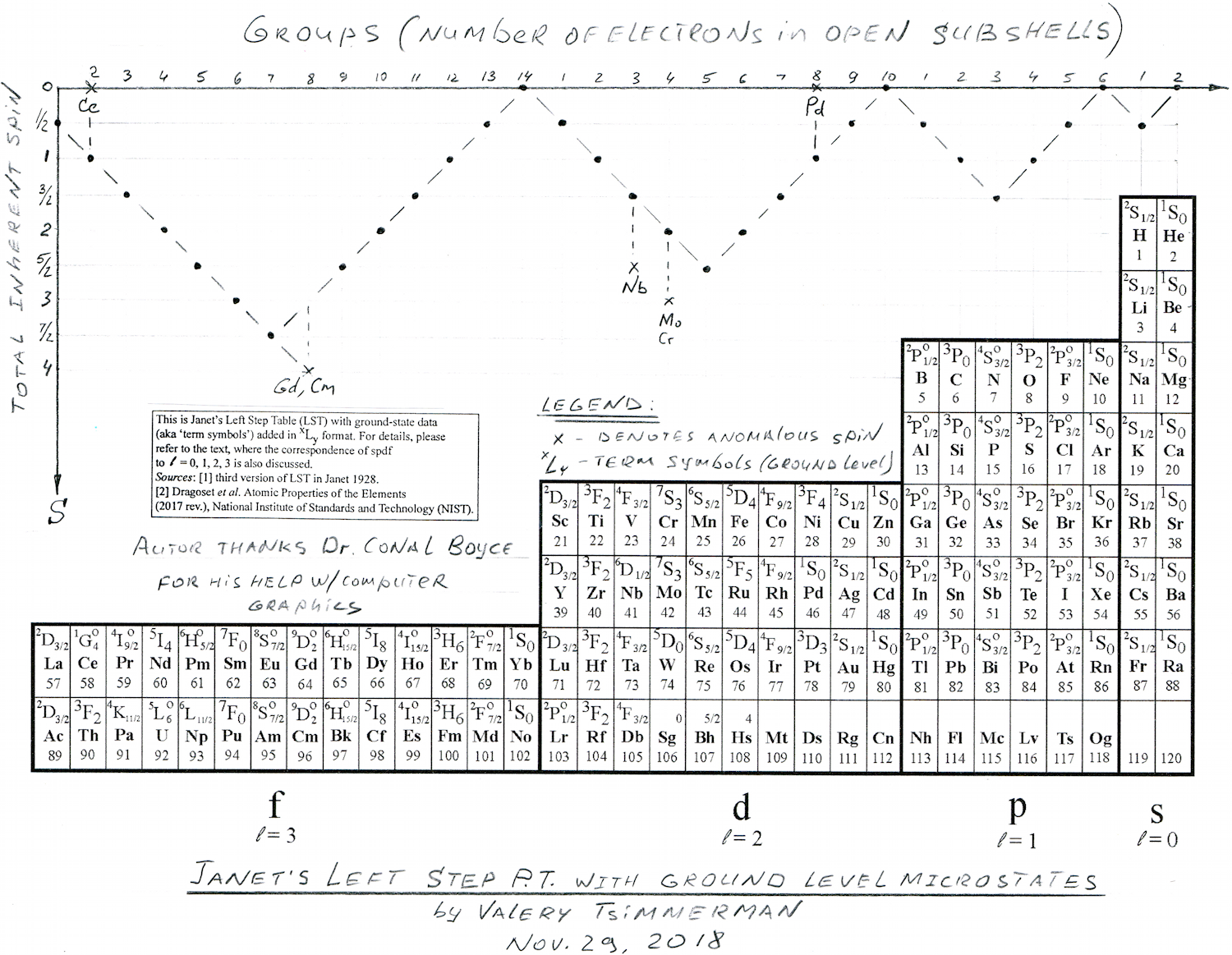
Janet's Left-Step with Ground Level Microstates
By Valery Tsimmerman, who writes:
Janet's LST with ground level microstate information and total spin graph shown for each group of elements. The top line represents number of electrons in open sub-shells (with exception of six anomalous elements). Information shows physical (spectroscopic) basis of the groups.
The zigzag line on top is a graphic representation of Hund's rule showing the total inherent spins of atoms and the total spin of Cu is 1/2, same as for Ag and Au. When it comes to ground level atomic microstates and Hund's rule Cu is not anomalous (2S1/2), despite its anomalous electron configuration.
The diagram represents Hund's Rule that states that "the lowest energy atomic state is the one that maximizes the total spin quantum number for the electrons in the open subshell" (Wikipedia). Y-axis is the total spin and x-axis is number of electrons in open shells (with exception of six anomalous elements).
First, I would like to make couple of general comments. When discussing periodicity, they typically talk about chemical properties and electron configurations/differentiating electrons, etc, but those are not specific enough. For each electron configuration there are multiple microstates. For example, for single electron configuration of carbon there are over 30 microstates and only one of them corresponds to ground level. So, microstates express combined physical/spectroscopic properties of whole atoms and, the most important, combined properties of electrons located in open subshells.
Now, look at ground level term symbols in each group. I see amazing consistency, especially in the main groups. It tells me that groups are not only chemical, but physical!
Looking at periods one can see that all periods in s, p & d blocks begin with elements that have multiplicity M=2 and end with M=1. This is also true for f-block if it starts with La and Ac and ends with Yb and No. This puts Lu and Lr firmly in group 3. Placing La and Ac in group three ruins spectroscopic consistency.
Click here image to enlarge the PT below.
| Year: 2018 | PT id = 952, Type = formulation |
ADOMAH Periodic Table Formulation with NIST Data
By Valery Tsimmerman, who writes:
I would like to share with you another variant of my ADOMAH periodic table formulation that holds additional spectroscopic information.
Click here image to enlarge the PT below.
| Year: 2018 | PT id = 956, Type = misc data |
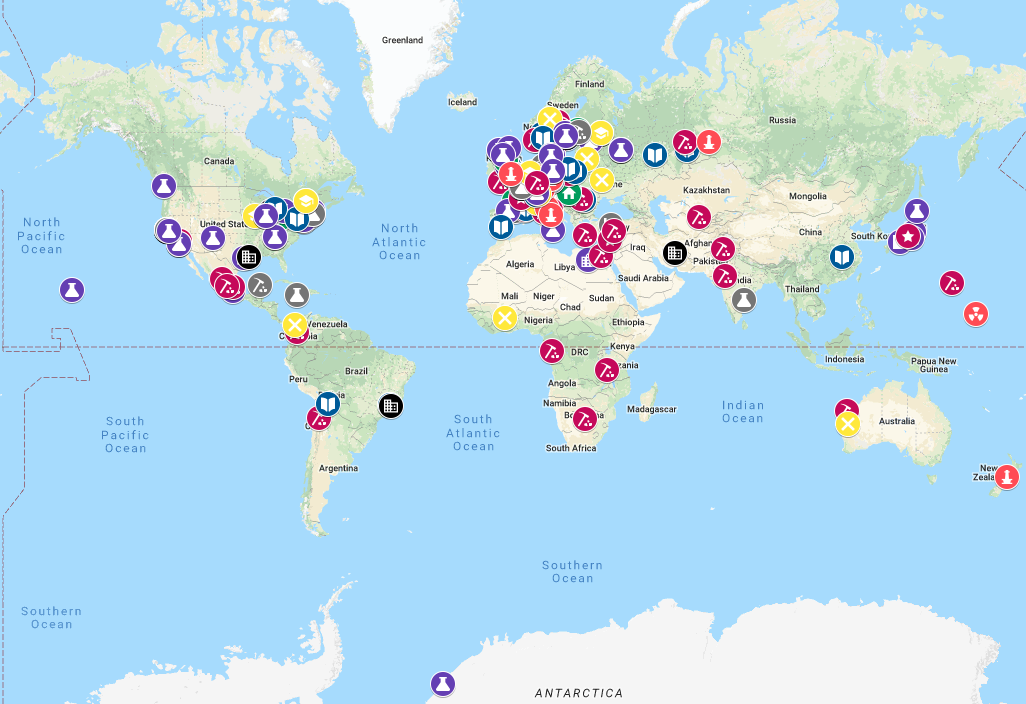
Places of the Periodic Table
An interactive, searchable Google map of places associated with the developers of the periodic table and with the chemical elements with links to further information brought to you by Carmen Giunta and James Marshall, with the encouragement of the ACS Division of the History of Chemistry (HIST), to mark the International Year of the Periodic Table (IYPT). This is an interactive searchable map of places associated with the developers of the periodic table and with the chemical elements with links to further information.
Examples include places where elements were discovered or synthesized, mineral sources of elements, places where discoverers of chemical periodicity worked, and places for which elements were named. Each entry contains links to further information about the person, place, or event described. The type of site is indicated (for example, lab, residence, mineral source, etc.), as well as whether (to the best of our knowledge) the historical site still exists at the location. For more information on the type of site, please consult this key to the map's fields. The map is intended for educational and informational purposes only, and is not meant as a travel guide. If you wish to visit a site on this map, please consult other resources to confirm access, and use common sense. (Read more here.)
Thanks to Eric Scerri for the tip! See the website EricScerri.com and Eric's Twitter Feed.
| Year: 2018 | PT id = 957, Type = misc |
Periodic Table Song (2018 UPDATE!)
An AsapSCIENCE song + video of the chemical elements. This is a nice alternative to the well known Tom Lehrer Elements song from 1959.
| Year: 2018 | PT id = 971, Type = data misc |
Superconductivity of Hydrides Periodic Table
Scientists from Moscow Institute of Physics and Technology and Skoltech have demonstrated the high-temperature superconductivity of actinium hydrides and discovered a general principle for calculating the superconductivity of hydrides based on the periodic table alone. The results of their study were published in The Journal of Physical Chemistry Letters.
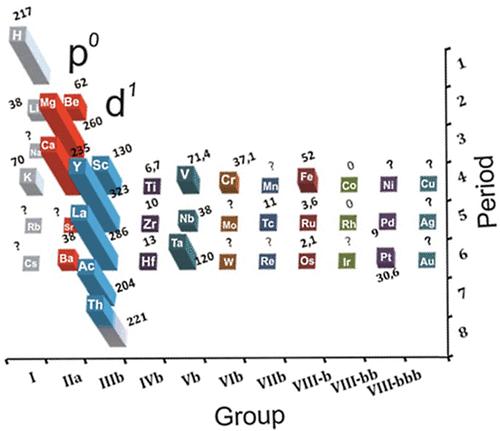
Thanks to Eric Scerri for the tip! See the website EricScerri.com and Eric's Twitter Feed.
| Year: 2018 | PT id = 977, Type = formulation 3D |
Simpson's 4-Dimensional Version of the ADOMAH Periodic Table
Doug Simpson writes:
"Valery Tsimmerman's ADOMAH table and website got me started as a periodic table hobbyist. The attached photos show what I've been up to. Valery's observation that n, l, & m conspire to generate a half-filled tetrahedral lattice inspired me to create a 4D periodic table using all four quantum numbers as coordinates."

| Year: 2018 | PT id = 978, Type = non-chem |
Snakes and Ladders of the Periodic Table
Gordon Marks has developed a game of Snakes & Ladders (Chutes and Ladders in the USA) using a Periodic Table, symbols as in Peter van der Krogt. Instructions here:
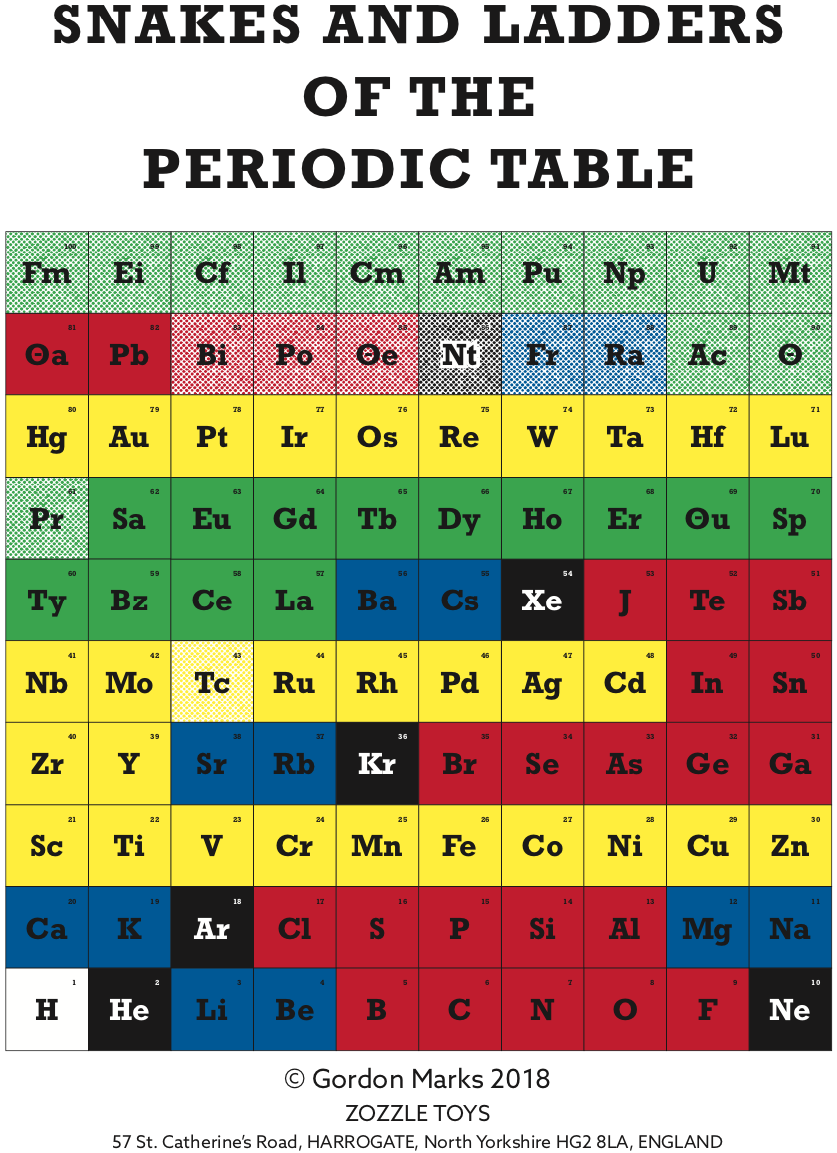
| Year: 2018 | PT id = 985, Type = misc |
Better Call Saul - Gale sings The Elements
The Better Call Saul Season 4 Episode 3 Clip starring David Costabile singing The Elements Song:
Thanks to Conal for the tip!
| Year: 2018 | PT id = 1103, Type = data |
Data Rich Periodic Table
Explore James L. Marshall's data rich periodic table.
Dr. Marsall provided the location data for Carmen Giunta's interactive, searchable Google map of places associated with the developers of the periodic table and the chemical elements.

| Year: 2018 | PT id = 1144, Type = misc non-chem |
Christmas Periodic Table of Substances
By Tom Gauld for New Scientist 22/29 December 2018 pp90:

| Year: 2018 | PT id = 1202, Type = formulation |
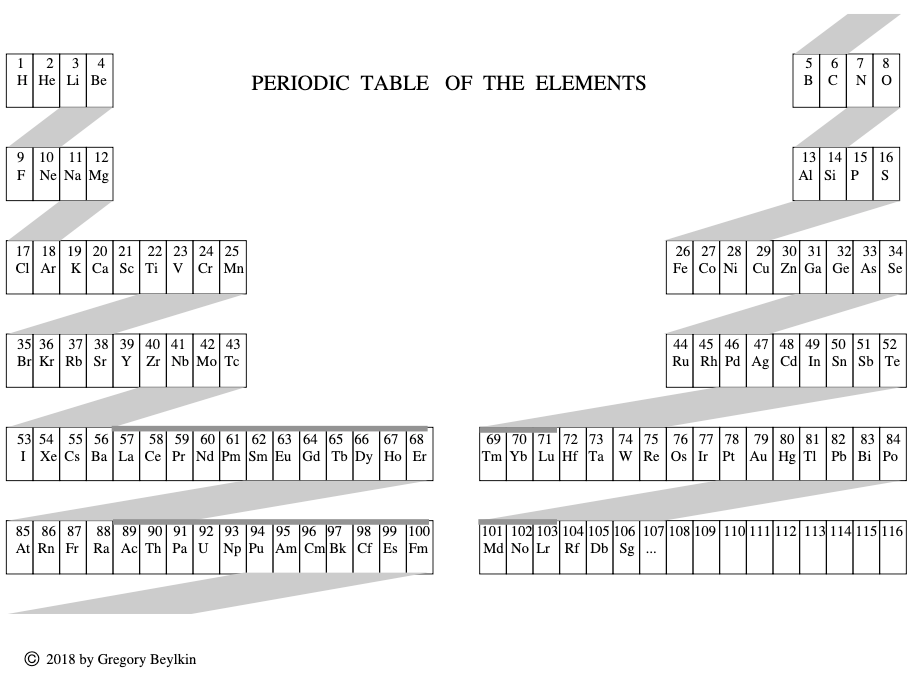
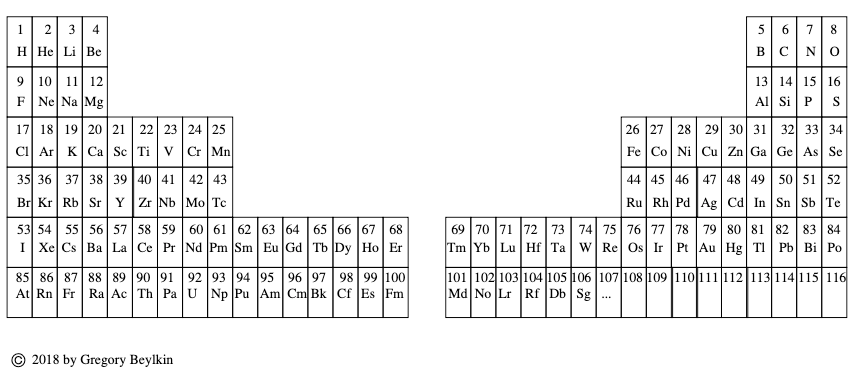
Beylkin's Periodic Table of The Elements
René Vernon writes: Beylkin's Periodic Table of The Elements has 4n2 periods, where n = 2,3..., and shows symmetry, regularity, and elegance, more so than Janet's left step table.
Beylkin (an applied mathematician) writes:
"Let us take a continuous strip of paper and, on one side of the strip, write all the elements in the order of their atomic numbers. We then form a spiral with the strip such that the two most chemically distinct groups, the group of halogens (in which we include hydrogen) and the group of noble gases, are properly aligned. By flattening the strip on a plane and folding it in the middle, we obtain the new periodic table..."
Other features:
- H is over F, which is a smoother fit in terms of physicochemical trends down the group
- He is over Ne, which is a smoother fit etc
- group 3 has lanthanum in it
- the modern relationships Ti-Zr-Hf, V-Nb-Ta, Cr-Mo-W, and Mn-Tc-Re can still be traced
- the lanthanides and actinides are integrated into the main body of the table
- 15 lanthanides and 15 actinides(!)
- the old school arrangement of B-Al-Sc-Y-La can still be traced, as can the less smooth alternative B-Al-Sc-Y-Lu
- the 1s "block" starts at H; the s block proper at Li; p at B; d at Sc; f at Ce
There are four new(ish) groups: Ti-Zr-Ce-Th, V-Nb-Pr-Pa, Cr-Mo-Nd-U and Mn-Tc-Pm-Np. For the actinide elements of these groups, the resemblance of the earlier actinides to their lighter transition metal congeners is well known. For the lanthanide elements, Johansson et al. (2014) wrote a nice article about Ce and its cross-road position. For Pr, Nd, and Pm, all of these are known in multiple oxidations states (+2, +3, +4 excl. Pm, and +5 for Pr only), just as the transitions metals are so known.
- Beylkin G 2018, The periodic table of the elements with 4n2 n = 2,3... periods, https://arxiv.org/pdf/1901.02337.pdf
- Eric 2006, https://www.meta-synthesis.com/webbook/35_pt/pt_database.php?PT_id=20
- Johansson, B., Luo, W., Li, S. et al. 2014, Cerium; crystal structure and position in the periodic table. Sci Rep 4, 6398. https://doi.org/10.1038/srep06398
- Gregory Beylkin: https://en.wikipedia.org/wiki/Gregory_Beylkin
| Year: 2018 | PT id = 1220, Type = formulation 3D |
Elements in Six Dimensions
The Elements in Six Dimensions, by Gerald Eadie, arranged by volume periods of nuclide mass averages:
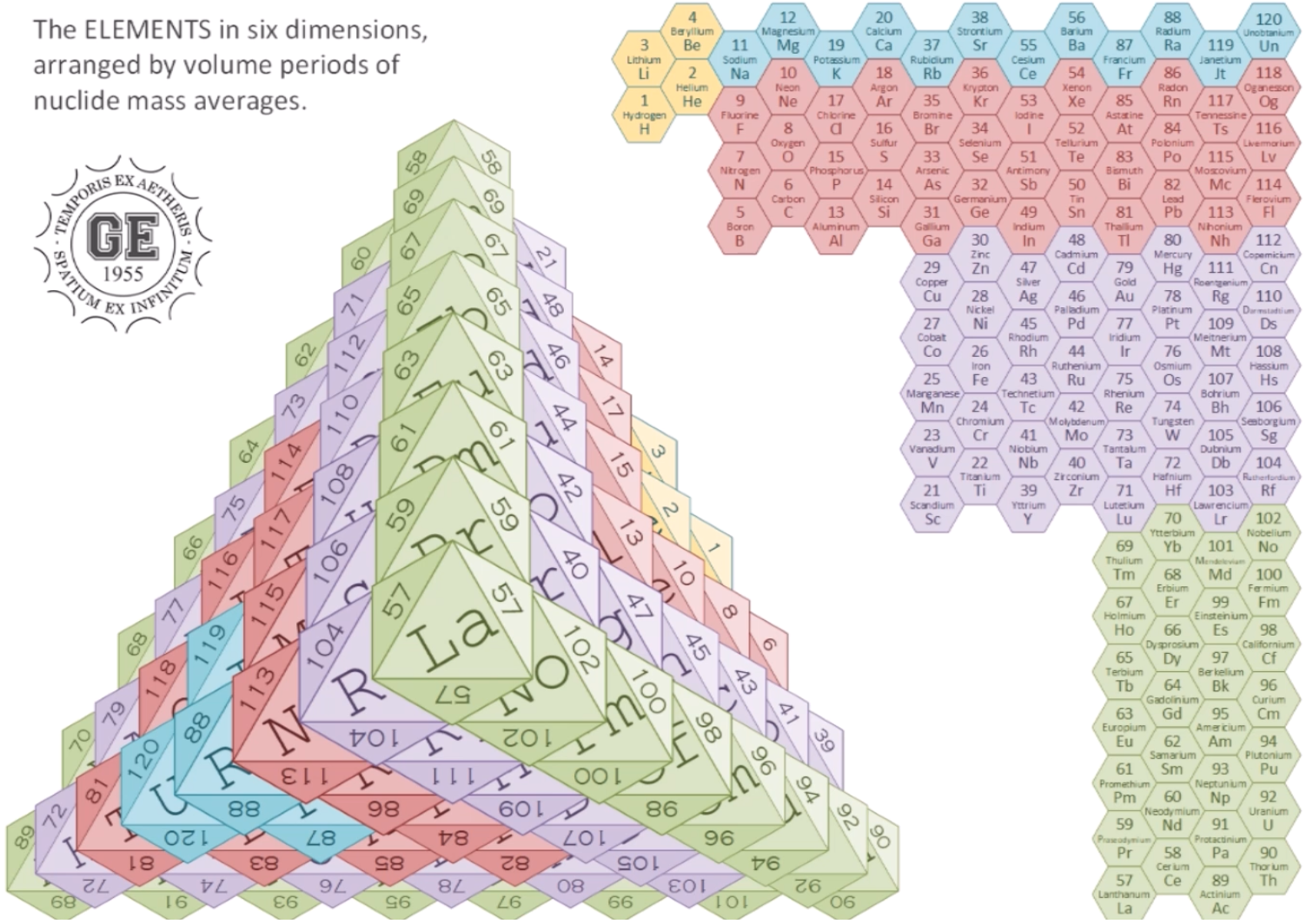
| Year: 2018 | PT id = 1261, Type = formulation |
Kurushkin's 32-Column Periodic Table & Left-step Periodic Table United
Dr Mikhail V. Kurushkin, 32-column Periodic Table & Left-step Periodic Table United: https://bernalinstitute.com/events/bernal-seminar-by-dr-mikhail-v-kurushkin-itmo-universityrussia/
ABSTRACT
The pursuit of optimal representation of the Periodic Table has been a central topic of interest for chemists, physicists, philosophers and historians of science for decades (Leigh, 2009; Scerri, 2009). Should the Periodic Table of Chemical Elements first and foremost serve the needs of chemists as implied by its name? Or should it start from considerations of before quantum mechanics and thus be more appealing to physicists (Scerri, 2010, 2012b)? Is there a representation which overcomes this problem? The Periodic Table is from a fundamental point of view a graphic representation of periodicity as a phenomenon of nature. While the 32-column Periodic Table, popularized by Glenn T. Seaborg, is considered by chemists the most scientifically correct representation (Scerri, 2012a), physicists apparently prefer the Left-step Periodic Table above all (Scerri, 2005; Stewart, 2010). Alternatively, it is suggested that a rigorously fundamental representation of periodicity could only take the form of a spiral as, evidently, the abrupt periods of 2-D Periodic Tables contradict the gradual increase of atomic number, and the spiral representation reconciles this debate (Imyanitov, 2016). An optimal representation is eagerly sought after both for the needs of philosophy of chemistry and chemical education as their never-ending dialogue secures a thorough methodology of chemistry. The aim of the present work is to show that the 32-column Periodic Table and the Left-step Periodic Table can co-exist in mutual tolerance in a form of what Philip Stewart has already called Kurushkin’s Periodic Table (Kurushkin, 2017), Figure 1 below.
René Vernon writes:
"Kurushkin reminds us that the Janet left step table (with Sc-Y-Lu-Lr, and He over Be), and the version of the table with the s-elements on the right (also with Sc-Y-Lu-Lr, and He over Be) are interchangeable.
"For an earlier paywall version which includes a short video see:
Kurushkin M 2018, Building the periodic table based on the atomic structure, Journal of Chemical Education, vol. 94, no. 7, pp. 976–979, https://pubs.acs.org/doi/10.1021/acs.jchemed.7b00242
"Kurushkin’s interchangeable approach extends to tables with group 3 as either Sc-Y-La-Ac or Sc-Y-Lu-Lr. See Vernon's Yin Yang of The Periodic Table https://www.meta-synthesis.com/webbook/35_pt/pt_database.php?PT_id=1252"
| Year: 2018 | PT id = 1278, Type = formulation |
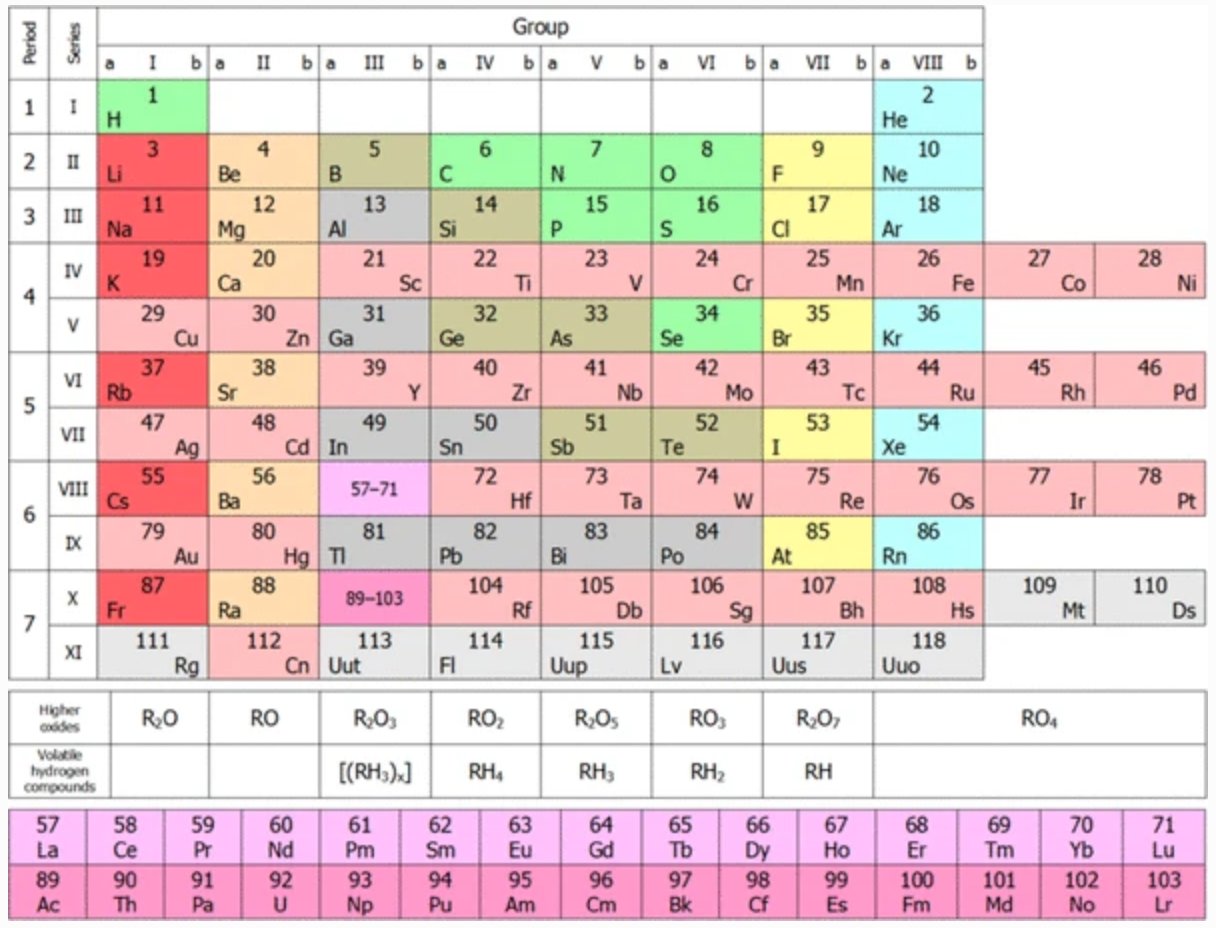
Short Form of Mendeleev’s Periodic Table of Chemical Elements
Andriiko, A.A., Lunk, HJ. The short form of Mendeleev’s Periodic Table of Chemical Elements: toolbox for learning the basics of inorganic chemistry. A contribution to celebrate 150 years of the Periodic Table in 2019. ChemTexts 4, 4 (2018). https://doi.org/10.1007/s40828-018-0059-y
| Year: 2018 | PT id = 1309, Type = review |
Creating a Symbol of Science: The Development of a Standard Periodic Table of the Elements
Robinson, Ann E., "Creating a Symbol of Science: The Development of a Standard Periodic Table of the Elements" (2018). Doctoral Dissertations. 1385.
https://doi.org/10.7275/12706048 https://scholarworks.umass.edu/dissertations_2/1385. Download and view the PDF.
See Ann E. Robinson's ORCID page.
Mark Leach writes:
"An excellent, comprehensive study that is full of details and references."

 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
© Mark R. Leach Ph.D. 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com
This free, open access web book is an ongoing project and your input is appreciated.